Research Article
Cellulase and Dairy Animal Feeding
Department of Dairy Science, National Research Center, Dokki, Cairo, Egypt
H.H. Azzaz
Department of Dairy Science, National Research Center, Dokki, Cairo, Egypt
The increasing demand for milk requires integrated strategies to develop the animal production sector. The hardest problems which facing the developing of animal production sector in many countries is the wide gap between animal's requirements and available feeds.
In addition, agricultural and agroindustrial activities produce thousand of tons of dry material by-products per year (Graminha et al., 2008). Although these residues are nutritious, a small portion is being used directly as feed or as components for industrially formulated cattle diets (Yang et al., 2001).
The problems of feeding agricultural by products (lignocellulosic materials) directly to farm animals are in general, low protein content, high crude fiber, low digestibility coefficients and containing some anti-nutrients factors such as tannins and alkaloids (Kholif et al., 2005). Thus, to increase digestibility of these agricultural residues, it is important to destroy the linkage between cellulose, hemicellulose and lignin.
Cellulose biodegradation by cellulases, produced by numerous microorganisms is very important in several agricultural and waste treatment processes (Hamer, 2003; Angenent et al., 2004; Das and Singh, 2004; Haight, 2005; Murad et al., 2009; Azzaz, 2009). Recent advances in fermentation technology and biotechnology have allowed for production of large quantities of biologically active enzymes such as cellulases that can also be used as livestock feed supplements (McAllister et al., 2001).
Supplementing diets of farm animals with cellulases can improve feed utilization and animal performance by enhancing fiber degradation in vitro (Gado et al., 2007; El-Adawy et al., 2008; Rodrigues et al., 2008; Murad et al., 2009; Azzaz, 2009), in situ (Lewis et al., 1996; Tricarico et al., 2005; Krueger et al., 2008), in vivo (Yang et al., 1999; Gado et al., 2007; Salem et al., 2007; Gado and Salem, 2008; Murad et al., 2009; Azzaz, 2009). and improve milk production by dairy cows (Lewis et al., 1995; Tricarico et al., 2005; Stella et al., 2007) and milk production by small ruminants (Titi and Lubbadeh, 2004; Stella et al., 2007; Murad et al., 2009; Azzaz, 2009).
This review provides an over view of the main variables to be considered for cellulase production from agricultural residues for ruminant feeding.
CELLULASE PRODUCTION
Cellulase is among the industrially important hydrolytic enzymes and is of great significance in present day biotechnology. Cellulase refers to a family of enzymes (Fiberolytic enzymes) which act in concert to hydrolyze fiber of plant cell wall to glucose, cellobiose or cellooligosaccharides. Microbial conversion of cellulosic/lignocellulosic biomass into useful products is a complex process involving combined action of three enzymes namely endoglucanase, exoglucanases and β-glucosidase (Knowles et al., 1987; Wood and Garica-Campayo, 1990; Henrissat, 1994; Teeri, 1997; Lynd et al., 2002; Zhang and Lynd, 2004).
Endoglucanases hydrolyze accessible intramolecular β-1, 4-glucosidic bonds of cellulose chains randomly to produce new chain ends; exoglucanases processively cleave cellulose chains at the ends to release soluble cellobiose or glucose; and β-glucosidases hydrolyze cellobiose to glucose in order to eliminate cellobiose inhibition. These three hydrolysis processes occur simultaneously as shown in Fig. 1.
Production of cellulases and their properties have been extensively studied during recent years (Rajoka and Malik, 1997; Azzaz, 2009). The development of microbial strains, media composition and process control have all contributed to achievements of high levels of extra cellular accumulation of cellulases for subsequent applications in industrial processes (Ghose, 1987). In this review we well focused on the cellulase production process including cellulase production microorganisms, substrates, techniques, optimization of culture condition and activity assay methods.
Cellulase production microorganisms: Cellulase enzyme produced by several microorganisms, commonly by bacteria and fungi, including aerobes, anaerobes, mesophiles, thermophiles and extremophiles (Bahkali, 1996; Magnelli and Forchiassin, 1999; Shin et al., 2000; Immanuel et al., 2006) Aerobic fungi and bacteria generally produce extracellular cellulases. Bacterial cellulases are constitutively produced, whereas fungal cellulase is produced only in the presence of cellulose (Suto and Tomito, 2001).
Interestingly, anaerobic bacteria (Clostridium thermocellum, C. cellulovorans, Ruminococcus albus, R. flavefaciens, Fibrobacter succinogenes and Acetivibrio cellulolyticus) and anaerobic fungi (Neocallimastix frontalis, N. patriciarum and Piromyces equi) produce cellulases in the form of a multienzyme aggregated complex (Groleau and Forsberg, 1981; Lamed et al., 1987; Wood, 1992; Gilbert and Hazlewood, 1993; Beguin and Lemaire, 1996; Bhat and Bhat, 1997)
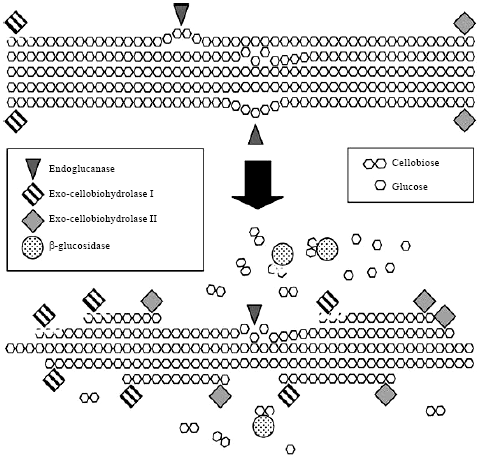 | |
| Fig. 1: | A simplified schematic representation of the process involved in complete enzymatic hydrolysis of a cellulose microfibril. Endoglucases attack amorphous regions within the crystalline microstructure, thereby creating new foci for attack by exo-cellobiohydrolases. Cellobiose dimers are cleaved by β-glucosidases to yield glucose monomers, which can now be transported across the membrane to participate in energy generating metabolic reactions (Malherb and Cloete, 2003) |
Most of the early studies were carried out on the biochemistry and enzymology of cellulases from aerobic mesophilic fungi, Trichoderma viride, T. reesei, Penicillium pinophilum, Sporotrichum pulverulentum, Fusarium solani, Talaromyces emersonii and Trichoderma koningii (Grigelmo-Migeul; Martin-Belloso, 1998).
In the past two decades, it has been recognized that other microorganisms such as thermophilic fungi (Sporotrichum thermophile, Thermoascus aurantiacus, Chaetomium thermophile and Humicola insolens), mesophilic anaerobic fungi (N. frontalis, N. patriciarum P. communis, Sphaeromonas communis, P. equi and Orpinomyces sp.), mesophilic and thermophilic aerobic bacteria (e.g., Cellulomonas fimi, Pseudomonas fluorescens subsp. cellulosa, Cellvibrio sp., Microbispora bispora, Clostridium cellulolyticum and C.cellulovorans) mesophilic and thermophilic anaerobic bacteria (A. cellulolyticus, Bacteroides cellulosolvens, F. succinogens, R. albus, R. flavefaciens, C. thermocellum and C. stercorarium), as well as actinomycetes (Thermomonospora fusca), produce highly active cellulase systems (Bhat and Maheswari, 1987; Aubert et al., 1988; Beguin and Lemaire, 1996; Claeyssens et al., 1998).
In addition, hyperthermophilic microorganisms-namely, Thermotoga sp., Pyrococcus furiosus and Thermofilum sp., which grow between 85 and 110°C-produce extremely stable cellulolytic and enzymes (Simpson et al., 1991; Winterhalter and Liebl, 1995). Fungal genera like Aspergillus, Cladosporium, Fusarium, Geotrichum, Myrothecium, Paecilomyces, Penicillium and Trichoderma have received considerable study with respect to their cellulolytic enzymes capability (Lynd et al., 2002).
A capacity to degrade cellulose is a character distributed among a wide variety of aerobic, facultative aerobic, anaerobic bacteria and fungi. A fundamental difference exists in the mechanism of cellulose hydrolysis between aerobic and anaerobic fungi and bacteria reviewed by Leschine (1995) and Tomme et al. (1995). Aerobic fungi and bacteria characteristically comprise non-complexed cellulase systems, which entail the secretion of the cellulose hydrolysis enzymes into the culture medium. However, anaerobic bacteria especially (Clostridium spp.) and fungi of the genera (Neocallimastix, Piromonas and Sphaeromonas) contain complexed cellulase systems where the cellulose hydrolyzing enzymes are contained in membrane-bound enzyme complexes.
Complexed cellulase systems allow greater coordination between the different cellulose hydrolyzing enzymes. In aerobic systems, where active aeration and agitation is required, loss of the secreted enzymes and their degradation intermediates might prove detrimental to overall process efficiency. This apparent contradiction might be offset when the energetics of aerobic and anaerobic microorganisms is compared. In general, aerobic microorganisms gain far more energy from glucose than anaerobic microorganisms (38 mole ATP vs. 2-4 mole ATP per mole of glucose). Therefore, the apparently aggressive cellulose hydrolyzing strategy utilized by aerobes might be beneficial given the potential enormous gain in metabolic energy.
Cellulase production substrates: Since the production of cellulase enzyme is a major process and economically viable, major attention has been given to use lignocellulosics as substrate for cellulase production. The major component of lignocellulosic materials is cellulose, followed by hemicellulose and lignin (Fig. 2) Cellulose is the primary product of photosynthesis in terrestrial environments and the most abundant renewable bioresource produced in the biosphere (~100 billion dry tons/year) (Macrae et al., 1993; Jarvis, 2003; Zhang and Lynd, 2004). Cellulose and hemicellulose are macromolecules constructed from different sugars; whereas lignin is an aromatic polymer synthesized from phenylpropanoid precursors. The composition and proportions of these compounds vary between plants (Prassad et al., 2007; McKendry, 2002; Malherbe and Cloete, 2003; John et al., 2006; Stewart et al., 1997; Reguant and Rinaudo, 2000; Perez-Diaz et al., 2005).
Chemically, cellulose is a linear polymer that is composed of D-glucose subunits linked by β-1, 4 glycosidic bonds forming the dimer cellobiose. These form long chains (or elemental fibrils) linked together by hydrogen bonds and van der Waals forces. Cellulose usually is present as a crystalline form and a small amount of no organized cellulose chains forms amorphous cellulose. In the latter conformation, cellulose is more susceptible to enzymatic degradation (Perez et al., 2002). Cellulose appears in nature to be associated with other plant compounds and this association may affect its biodegradation.
Hemicellulose is a polysaccharide with a lower molecular weight than cellulose. It is formed from D-xylose, D-mannose, Dgalactose, D-glucose, L-arabinose, 4-O-methyl-glucuronic, D-galacturonic and D-glucuronic acids. Sugars are linked together by β-1, 4- and sometimes by β-1, 3-glycosidic bonds. The main difference between cellulose and hemicellulose is that hemicellulose has branches with short lateral chains consisting of different sugars and cellulose consists of easily hydrolyzable oligomers.
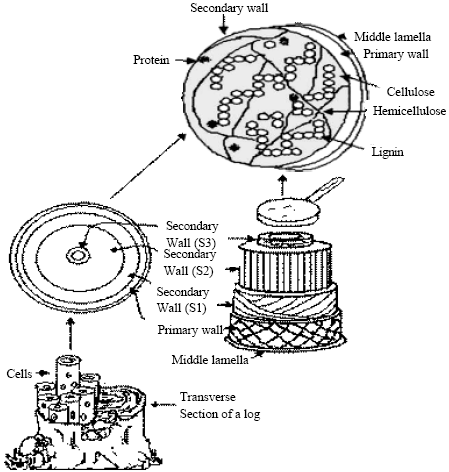 | |
| Fig. 2: | Composition of lignocellulosic residues (Sanchez, 2009) |
Lignin is linked to both hemicellulose and cellulose, forming a physical seal that is an impenetrable barrier in the plant cell wall. It is present in the cellular wall to give structural support, impermeability and resistance against microbial attack and oxidative stress. It is an amorphous heteropolymer, non-water soluble and optically inactive that is formed from phenylpropane units joined together by non-hydrolyzable linkages.
Cellulase production techniques: There were two fermentation techniques we can use for cellulase production, as many other enzymes (Murad and Foda, 1992) these techniques are Solid State Fermentation (SSF) and submerged fermentation (SmF).
Solid state fermentation is defined as the cultivation of microorganisms on moist solid supports, either on inert carriers or on insoluble substrates that can be used as carbon and energy source. This process occurs in the absence or near absence of free water in the space between substrate particles. In this system, water is present in the solid substrate whose capacity for liquid retention varies with the type of material (Lonsane et al., 1985; Pandey et al., 2000). In contrast, in submerged fermentation (SmF) the nutrients and microorganisms are both submerged in water.
Approximately 90% of all industrial enzymes are produced in SmF, frequently using specifically optimized, genetically manipulated microorganisms. In this respect SmF processing offers an insurmountable advantage over SSF. On the other hand, almost all these enzymes could also be produced in SSF using wild-type microorganisms (Filer, 2001; Pandey et al., 2001). Interestingly, fungi, yeasts and bacteria that were tested in SSF in recent decades exhibited different metabolic strategies under conditions of solid state and submerged fermentation.
The aim of SSF is to bring the cultivated fungi or bacteria into tight contact with the insoluble substrate and thus to achieve the highest substrate concentrations for fermentation. This technology results, only on a small scale, in several processing advantages of significant potential economic and ecological importance as compared with SmF (Table 1).
| Table 1: | Biotechnological advantages of solid state fermentation (SSF) against submerged technology (SmF) (Holker et al., 2004) |
 | |
However, there are also several disadvantages of SSF, which have discouraged use of this technique for industrial production. The main obstructions are due mainly to the build-up of gradients of temperature, pH, moisture, substrate concentration or CO2 during cultivation, which are difficult to control under limited water availability. It has become clear (as mentioned in nearly every review cited) that the cost-factor for the production of bulk-ware enzymes in most cases favors SSF over SmF. The low estimated costs of SSF are due to the rather traditional preferential claim of SSF, viz. SSF utilises complex, heterogenous agricultural wastes as substrates and uses low-cost technology regarding sterility and regulation demands. However, attempts to reduce costs progress in SSF because of the strongly increased diversity in SSF research.
There is no consensus on the methods, the microorganisms or the substrates used, that would allow comparison with other cultivation technologies. The broad spectrum of substrates used represents an especially severe problem. As already mentioned, one great advantage of SSF has always been the possibility of using substrates that are abundant, cheap and not applicable to SmF. However, regardless of the differences in process up-scaling, the scientific and technological impact of research data is difficult to compare when results are obtained with different microorganisms producing different products and using a vast variety of substrates (Rafae et al., 2006; Abd-El-salam et al., 1994). There are many substrates could be used as, pineapple, mixed fruit, maosmi waste, wheat straw with raspberry seed powder, broiler matter, corn stover, almond meal, apple pomace, molasses, permeate, corncob, barley husk, banana waste, soybean cake, cacao jelly, sweet lime rind, cassava, soybean, amaranth grain, eucalyptus kraft pulp, coffee residues, hardened chickpeas, lignite, rubber or orange bagasse and some food industry wastes (Holker et al., 2004; Murad, 1998).
To facilitate comparison of results, the use of inert substrates as solid supports is becoming increasingly important (Gautam et al., 2002; Ooijkaas et al., 2000). Surprisingly, biological parameters, such as the stability of the produced enzymes at high temperature or extreme pH, have also been reported to be better in SSF (Deschamps and Huet 1985; Acuna-Arguelles et al., 1995). Capability repression or protein degradation by proteases severe problems in SmF were often reduced or absent in SSF (Pereira et al., 1993; Aguilar et al., 2001). In contrast, much less research has been carried out to evaluate the metabolic differences of microorganisms when cultivated in SSF or SmF.
Optimization of culture conditions for cellulase production: The optimization of fermentation conditions is an important problem in the development of economically cellulase production, so this condition ex: initial pH of growth medium, incubation period, inoculum size, nitrogen source and carbon source…etc. must be optimum for getting maximum production of cellulase.
Initial pH of growth medium: According to Shoichi et al. (1985) the initial pH of the medium has a great effect on the growth of the organism, on the membrane permeability, also on the biosynthesis and stability of the enzymes (Murad, 1998; Murad and Salem, 2001). Fadel and Foda (1993) reported that most fungal cellulases are produced optimally at initial pH range between 4.0-5.5 Thus, the optimum pH for cellulase formation was reported to be 4.0 for a mutant of A.terreus (Garg and Nieellakantan, 1982) pH ranging between 4.0-4.8 for T. viride, T. reesi, A. terreus, A. phoenicis and p.decumbens (Shewale and Sandana, 1978; Aleksidze and Kvachadze, 1984; Deschamps and Huet, 1984; Qu et al., 1986; Duff et al., 1986), pH 5.0 for T. viride, T. pseudokoningi, T. harzianum, A. terreus and A. fumigatus (Peitersen, 1977; Leena, 1979; Kalra and Sandhu, 1986) and pH 5.5 for Scytalidium lignicola and P. purpurogenum (Desai et al., 1982; Shoichi et al., 1985). It was reported that optimal pH for CMCase from A. niger and A. flavus NRRL 5521 was found to be 6.0 to 7.0 (Parry et al., 1983; Azzaz, 2009). Also Immanuel et al. (2007) found that pH 5-7 is optimum in case of coir waste as substrate, but pH 6 is optimum when sawdust used as substrate for cellulase production by A. niger and A. fumigatus. But Akiba et al. (1995) reported that the production was high at pH 4 and 4.5 by A. niger. While Azzaz (2009) reported that cellulase production by A.niger and A.flavus NRRL 5521 on cellulose powder medium showed highest values of cellulase activity at pH 6.0 (0.094 U mL-1) and pH 7.0 (0.042 U mL-1), respectively, On other hand, Krishna (1999) found that initial pH 7.0 is optimum in case of banana waste used as substrate for cellulase production by Bacillus subtilis and it varied with slight changes in the pH of the medium. Coral et al. (2002) observed that the enzyme activity has a broad pH range between 3 and 9.
Anustrup (1979) reported that there were no arrangements regarding enzyme data results. Such different results may appear because of differences within the same genus. In addition, no comparative investigations have been published on the enzymes from these organisms but the difference appears to be small as difference in morphology between the species.
Incubation period: The time of fermentation had a profound effect on microbial product formation (Murad and Foda, 1992; Murad, 1998; Murad and Salem, 2001). Thus cellulase production reached the maximum cellulase activity by Bacillus subtilis after 72 h of fermentation with banana waste (Krishna, 1999). Allen and Roche (1989) and Muniswaran and Charyulu (1994) have reported similar trend in cellulase production using Trichoderma viride. Also, Chandra et al. (2007) found that maximum cellulase activity was recorded on 72 h of incubation with groundnut fodder, wheat bran and rice bran fermented by A. niger. Milala et al. (2005) reported that cellulase show maximum activity after 72 hr of fermentation by A. niger grow on maize straw and rice husk. But Kang et al. (2004) found that the highest cellulase activity was obtained after 5-6 days of fermentation by A. niger grow on rice straw, while Ojumu et al. (2003) stated that A. flavus grown on sawdust, bagasse and corncob gave the highest cellulase activity at 12 h of fermentation. While, Azzaz (2009) found that cellulase production by A. niger and A. flavus NRRL 5521 on cellulose powder medium showed highest values of cellulase activity after 48 hr of incubation. However, the amount of cellulase activity is decreasing with increasing period of incubation, this might be due to denaturation of the enzyme, resulting from variation in pH during fermentation as reported by Krishna (1999), or may be due to cumulative effect of cellobiose, a dimer of glucose which is known to inhibit both endoglucanase and β-glucosidase (Howell and Mangat, 1978). Also Hattaka (1983) suggested that delignification produces aromatic water-soluble products which can repress the cellulolytic action of the enzyme.
Inoculum size: Zhang et al. (2001) investigated the effect of inoculum size on cellulase synthesis by Trichoderma viride they reported that the impact of the amount of inoculants on cellulase production was small and 5% inoculum was the most suitable, also Alam et al. (2005) revealed that the higher cellulase activity of 0.0413 unit was achieved with 5% (v/w) of inoculum size when fermented oil palm biomass by Trichoderma harzianum. In addition, Azzaz (2009) reported that production of cellulase on cellulose powder medium by A.niger and A.flavus NRRL 5521 was increased significantly by increasing inoculum size (v/v) up to 4% (0.077 U mL-1) and 7% (0.060 U mL-1), respectively. Omojasola et al. (2008) found that amount of cellulase activity was decreased at inoculum sizes above 6 and 8% for pineapple peel and pineapple pulp fermentation by A. niger. The decrease in cellulase production with further increase in inoculum might be due to clumping of cells which could have reduced sugar and oxygen uptake rate and also, enzyme release (Omojasola et al., 2008).
Nitrogen source: Prescott and Dunn (1959) reported that molds in general, may utilize a large number of nitrogen containing compounds. They added that ammonium salts, nitrates, proteins, amino acids and urea are considered satisfactory nitrogen sources. Linko et al. (1978) reported that inorganic nitrogen source in the form of ammonium salts, seemed to be an excellent source of nitrogen for T. viride. Zeltins (1970) found that the presences of NaNO3 in A. terreus growth medium enhanced the synthesis of cellulases by 7.13%. Xavier and Lonsane (1994) also observed a similar increase in carbohydrate utilization and reduction in fermentation time when enriching a sugarcane- press-mud medium with 1.8% ammonium sulphate but further increase in concentration did not improve total soluble carbohydrate degradation.
Krishna (1999) reported that additional supply of N-sources to medium contains banana waste fermented with Bacillus subtilis influenced the cellulase activity to a certain extent, whereas the rate of cellulase production increased with increase in the concentration of ammonium sulphate or sodium nitrate in the medium up to 1% (w/w).
In contrast, Sternberg (1976) found that Trichoderma can not use nitrate as source of nitrogen. Reese and Meguire (1971) found that addition of organic nitrogen source such as peptone or protease peptone at one-tenth the cellulose concentration tended to decrease the lag phase in growth and enhanced the cellulase yield. Azzaz (2009) found that meat extract was the best nitrogen source producing the highest level of cellulase activity on cellulose powder medium by A. niger (0.097 U mL-1). While yeast extract gave the highest level of cellulase activity by A. flavus NRRL 5521 (0.11 U mL-1). This data indicating that the source of nitrogen should be organic for better results. Enari and Markenan (1977) reported that good cellulase production can be obtained with peptone as the organic nitrogen source and presence of certain levels of organic nitrogenous compounds was essential for high levels of cellulase production.
Carbon source: Major impediments to exploit the commercial potential of cellulases are the yield stability and cost of cellulase production. The use of available lignocellulosic wastes as carbon source in the growth medium would reduce the costs of enzyme production; also use of these agricultural wastes in bioprocesses may helps to solve environmental problems, which are otherwise caused by their disposal. Ojumu et al. (2003) studied effect of several carbon sources including bagasse, sawdust and corncob on cellulase production by A. flavus; they found that A. flavus grown on sawdust gave the highest cellulase activity. Kang et al. (2004) reported that the mixture of rice straw and wheat bran showed better results in submerged fermentation for the production of cellulases and hemicellulases by A. niger. Likewise coir and sawdust are suitable lignocellulosic bio wastes for the production of cellulase enzyme. It could understand that sawdust is most suitable substrate for cellulase production when compared to that of baggase or corncob (Ojumu et al., 2003) as it gives highest yield of enzyme (Immanuel et al., 2007). Chandra et al. (2007) studied effect of several carbon sources including groundnut fodder, wheat bran, rice bran and sawdust on cellulase production by A. niger. They found that titres of cellulolytic enzymes at peak production time interval in solid state fermentation were higher on wheat bran than on other lignocelluloses substrates in this study. Also Azzaz (2009) studied effect of several carbon sources including banana wastes, rice straw, wheat straw, corn stalks and pure cellulose powder on cellulase production by A. niger and A. flavus NRRL 5521, wheat straw gave the highest cellulase production when fermented with A.niger (0.177 U mL-1), while rice straw gave the highest cellulase production when fermented with A. flavus NRRL 5521(0.046 U mL-1). Krishna (1999) reported that banana fruit stalk gave the maximum cellulase production when fermented with Bacillus subtilis under solid state fermentation. The difference in enzyme production could be attributed to individual enzymes of the total cellulase enzyme system. Chandra et al. (2007) reported that differences in titres of cellulase yields in different studies can be attributed to use of different materials as solid matrix, different cultural practices and different organisms.
Cellulase activity assay: The measurement of cellulase activity is hampered by the nature of substrates used and the complexity of the enzyme systems produced by different microorganisms. In order to overcome these problems, numerous assays have been developed (Wood and Bhat, 1988; Biely et al., 1992).
Cellulase quantitative assays: There are two basic approaches to quantitative measuring cellulase activity are: (1) measuring the individual cellulase (endoglucanases, exoglucanases and β-glucosidases) activities and (2) measuring the total cellulase activity.
Endoglucanase activity is generally determined by measuring the reducing sugars released from either carboxymethyl (CM-) or hydroxyethyl (HE-) cellulose (Wood and Bhat, 1988). This activity can also be measured by determining either the decrease in viscosity of CM-cellulose, the swelling of cotton fiber in alkali or the decrease in turbidity of amorphous cellulose. In addition, substituted, unsubstituted, radio- and reduced end-labelled cello-oligosaccharides have been used to characterize endoglucanases (Bhat et al., 1990). Interestingly, some endoglucanase catalyse transferase reactions and act synergistically with cellobiohydrolase during the solubilization of crystalline cellulose (Wood et al., 1988; Claeyssens et al., 1990).
Cellobiohydrolase (CBH; exoglucanase) activity is determined by measuring the reducing sugars released from either Avicel or H3PO4-swollen cellulose (Wood and Bhat, 1988). Besides, CBH activity can be measured by determining either the release of dyed cellobiose from dyed Avicel or the decrease in turbidity of amorphous cellulose. Substituted and unsubstituted cello-oligosaccharides have been used to characterize CBHs (Claeyssens et al., 1998). B-glucosidase activity is generally determined by measuring the release of glucose and o-/p-nitrophenol from cellobiose and o-/p-nitrophenyl B-D-glucoside, respectively (Wood and Bhat, 1988). Also, the increase in reducing power of cello- oligosaccharides can be used as a measure of B-glucosidase activity.
Major reducing sugar assays depend on the reduction of inorganic oxidants such as cupric ions (Cu2+) or ferricyanide, which accepts electrons from the donating aldehyde groups of reducing cellulose chain ends. Their detection ranges vary from less than 1μg per sample to >2500 μg per sample. The most common reducing sugar assays include the dinitrosalicyclic acid (DNS) method (Ghose, 1987; Miller, 1959), the Nelson- Somogyi method (Nelson, 1944; Somogyi, 1952), the 2,2'-bicinchroninate (BCA) method (Waffenschmidt and Janeicke, 1987; Zhang and Lynd, 2005), the 4-hydroxybenzoylhydrazine (PAHBAH) method (Lever, 1972; Lever et al., 1973) and the ferricyanide methods (Kidby and Davidson, 1973; Park and Johnson, 1949).
The DNS and Nelson- Somogyi methods are two of the most common assays for measuring reducing sugars for cellulase activity assays because of their relatively high sugar detection range (i.e., no sample dilution required) and low interference from cellulase (i.e., no protein removal required). Detection ranges of many sugar assays can be modified using two strategies: (1) a further dilution after the color reaction and (2) varying sugar volume per sample prior to the reaction. For example, the DNS method was originally designed for 20-600 μg reducing sugar per sample (Miller, 1959), but its detection range can be expanded to samples of 100-2500 μg, followed by water dilution (Ghose, 1987). The same is true for the Nelson-Somogyi method.
Total cellulase activity, comprising endoglucanase, exoglucanase and B-glucosidase, is measured by determining the solubilization of cotton fiber, filter paper or Avicel. The most common total cellulase activity assay is the Filter Paper Assay (FPA) using Whatman No. 1 filter paper as the substrate, which was established and published by the International Union of Pure and Applied Chemistry (IUPAC) (Ghose, 1987). This assay requires a fixed amount (2 mg) of glucose released from a 50 mg sample of filter paper (i.e., 3.6% hydrolysis of the substrate), which ensures that both amorphous and crystalline fractions of the substrate are hydrolyzed. A series of enzyme dilution solutions is required to achieve the fixed degree of hydrolysis.
The strong points of filter paper assay are (1) it is based on a widely available substrate (2) it uses a substrate that is moderately susceptible to cellulases and (3) it is based on a simple procedure (the removal of residual substrate is not necessary prior to the addition of the DNS reagent). However, the FPA is reproduced in most laboratories with some considerable effort and it has long been recognized for its complexity and susceptibility to operators' errors (Coward-Kelly et al., 2003; Decker et al., 2003).
Reliability of results could be influenced by (1) the β-D-glucosidase level present in the cellulase mixture (Breuil and Saddler, 1985a, b; Schwarz et al., 1988; Sharrock, 1988), because the DNS readings are strongly influenced by the reducing end ratio of glucose, cellobiose and longer cellodextrins (Ghose, 1987; Kongruang et al., 2004; Wood and Bhat, 1988; Zhang and Lynd, 2005); (2) the freshness of the DNS reagent, which is often ignored (Miller, 1959); (3) the DNS reaction conditions, such as boiling severity, heat transfer and reaction time (Coward-Kelly et al., 2003); (4) the variations in substrate weight based on the area size (1x6 cm a strip), because this method does not require substrate excess (i.e., substrate amounts strongly influence enzyme activity) (Griffin, 1973) and (5) filter paper cutting methods, because the different paper cutting methods such as paper punching, razoring, or scissoring could lead to different accessible reducing ends of the substrate (Zhang and Lynd, 2005).
Cellulase qualitative assays: Qualitative assays have been developed to select microbial strains producing high levels of cellulases or to identify and/or characterize these enzymes in a given sample. Similarly, methods using CM-cellulose stained with Congo red can be used to select microorganisms producing endoglucanase activity. The capacity of Congo red to complex with CM- cellulose, but not with small oligosaccharide products, is conveniently used to detect endoglucanase activity after the fractionation of protein mixtures (Coughlan, 1988).
The advantages of the latter method are: (1) that the dyed fragments released from CM-cellulose diffuse from the detection gel into the separating gel and further help to identify the position of endoglucanase, which can subsequently be eluted and (2) the hydrolysis of the dyed substrate can be visually followed and the reaction terminated when necessary. The use of these substrates facilitates the identification of multiple forms of endoglucanase produced by different microorganisms. (Bhat and Hazlewood, 2001).
CELLULASE AND ITS APPLICATION IN RUMINANTS FEEDING
Slow or incomplete digestion of fibrous substrates often limits the overall digestive process in the rumen and can significantly influence animal performance in livestock production systems that use forages as a major component of the diet. As a result, many strategies have been developed to stimulate the digestion of the fibrous components in ruminant feeds. These have included the use of fiberolytic enzymes such as cellulase which stimulate fiber digestion and processing feeds to increase the rate and extent of fiber digestion. Cellulase preparations can be used to drive specific metabolic and digestive processes in the gastrointestinal tract and may augment natural digestive processes to increase nutrient availability and feed intake (McAllister et al., 2001).
In the last decade, fiberolytic enzymes preparations have become valuable tools for economically improving digestive processes in the ruminant (Yang et al., 2000; Bowman et al., 2002; Titi and Tabbaa, 2004; Abdel-Gawad et al., 2007; Gado et al., 2007; Knowlton et al., 2007; Gado et al., 2009; Murad et al., 2009; Azzaz, 2009) To date, little is known about the way that exogenous fiberolytic enzymes improve feed by rumen microorganisms. Several potential modes of action have been proposed. These include: (a) increase in microbial colonization of feed particles (Yang et al., 1999), (b) enhancing attachment and/or improve access to the cell wall matrix by ruminal microorganisms and by doing so, accelerate the rate of digestion (Nsereko et al., 2000) and (c) enhancing the hydrolytic capacity of the rumen due to added enzyme activities and /or synergy with rumen microbial enzymes (Newbold, 1997; Morgavi et al., 2000). In this part of paper we will review some of the recent data demonstrating the important effects of cellulase enzyme preparations as feed additives on feed digestibility and as well as milk production by ruminants.
Effect of cellulase enzyme preparations on feed digestibility: Adding cellulase enzyme preparations to the diets of ruminant animals has been the topic of many recent studies. A number of in vitro studies have demonstrated that it is possible to use cellulase enzyme preparations to enhance the processes associated with fiber digestion in the rumen (Hristov et al., 1996; Gado et al., 2007; El-Adawy et al., 2008; Rodrigues et al., 2008; Murad et al., 2009; Azzaz, 2009). The response is generally measured as an increase in the initial rate of dry matter and organic matter disappearance, increase in the rate of Neutral Detergent Fiber (NDF) and Acid Detergent Fiber (ADF) disappearance, altered ruminal pH, increase in VFA production, reduced the lag phase and improved efficiency of fermentation, increase ruminal microbial growth and increase in microbial protein synthesis in batch cultures of ruminal bacteria that have been supplemented with cellulase preparations (Lewis et al., 1996; Mohamed et al., 2005; Colombatto et al., 2007; Abdel-Gawad et al., 2007; Giraldo et al., 2008; Krueger and Adesogan, 2008; Murad et al., 2009; Azzaz, 2009). In addition, Positive effects of cellulase enzyme preparations on nutrients digestibility have been reported in different in vivo studies (Feng et al., 1996; Dong et al., 1999 Yang et al., 1999; Gado et al., 2007; Salem et al., 2007; Gado and Salem, 2008; Gado et al., 2009; Murad et al., 2009; Azzaz, 2009).
Dong et al. (1999) demonstrated that the effects of cellulase might start when the enzyme is in contact with the substrate, so enzyme-feed interaction appears as important. Giraldo et al. (2004) confirmed that a pre-ingestive enzyme-feed interaction is necessary for any significant beneficial effects on ruminal digestion. Others have also noted that a pre-feeding enzyme-feed interaction period is necessary for cellulase enzyme-mediated increases in digestion (Lewis et al., 1996; McAllister et al., 1999; Wang et al., 2001; Krueger and Adesogan, 2008). The enzyme addition onto feeds may create a stable enzyme-feed complex that protects free enzymes from proteolysis in the rumen as reported by Kung et al. (2000).
Measurements of total tract digestibility in ruminants have generally shown positive responses to fiberolytic enzymes with variable but often significant increases in the digestion of Dry Matter (DM), Organic Matter (OM), NDF, ADF and nitrogen (Yang et al., 1999, 2000; Rode et al., 1999; Beauchemin et al., 1999, 2000). Feng et al. (1996) concluded that the in vivo improvements in digestibility by enzymes are resulted from the enhanced colonization and digestion of degradable fiber fraction by ruminal microorganisms and, consequently, increase in degradation and particle size reduction.
Effect of cellulase enzyme preparations on milk production: Positive effects of adding fiberolytic enzyme to ruminant diets have been reported for lactating dairy cows. Dairy cows fed forage treated with a cellulase enzyme preparations ate more feed and produced 5-25% more milk (Lewis et al., 1995; Tricarico et al., 2005; Stella et al., 2007), improved the energy balance of transition dairy cows (De Frain et al., 2005) and increased milk production in small ruminants (Titi and Lubbadeh, 2004; Stella et al., 2007; Murad et al., 2009; Azzaz, 2009). Increased milk production has been observed in some studies (Beauchemin et al., 1999; Schingoethe et al., 1999; Yang et al., 1999; Murad et al., 2009; Azzaz, 2009) when the enzymes were applied at feeding (direct fed) or several hours before feeding. This response may be attributed to improved nutrient digestion after cellulase preparation supplementation (Beauchemin et al., 1997; Beauchemin and Rode, 1996). Milk fat and protein yields were higher for cows fed cellulase-treated diets. (Zheng et al., 2000).
Why the fat and protein content of milk was higher when cows were fed cellulases-treated diets is not clear, but it is likely indirectly related to changes in energy and protein digestion (Beauchemin et al., 1997). The use of enzyme additives has been associated with an improved efficiency of synthesis of microbial protein in the rumen (Jacobs et al., 1992). Therefore, it is probable that improved efficiency of microbial protein synthesis is a result of enzyme action on the forage structural polysaccharides altering the rate of ruminal degradation of structural carbohydrates (Lewis et al., 1996) and the provision of a suitable ruminally degradable nitrogen source (Beauchemin et al., 1999). Likewise, yields of total solids tended to be higher when cows were fed cellulase-treated diets (Yang et al., 1999; Zheng et al., 2000) this may be reflecting the higher milk yields or may be due to the generation of more nutrients which become available as a result of improvements in feed digestibility. Specifically, the increase in ruminally fermented OM, which resulted in a numerical downward shift in the ratio of acetate to propionate, would have increased delivery of glucogenic precursors to the mammary gland (Yang et al., 1999). Over all, the results of these studies provide more evidence that cellulase enzyme preparation can be used to improve milk production by lactating ruminants.
Factors affecting cellulase action as animal feed additive: It is evident from studies, that there is a wide range in responses to supplementation with direct fed-enzymes. Some of the reasons for the variation are given below:
Mode and time of enzyme delivery: Previous calls for more research on pre-feeding storage times of cellulase enzyme- treated dietary components (Wallace et al., 2001), led to in vitro and in vivo studies in which enzymes were added immediately or 24 h prior to feeding. However since such studies showed no differences due to time of enzyme treatment it has been suggested that there is little or no requirement for a reaction phase for enzymes added to diets (Beauchemin et al., 2003). However, more research is required in this area since many studies now involve enzyme addition to concentrates at milling and entail enzyme-diet interaction periods of up to one month. Depending on storage conditions, enzyme activity may be reduced by such protracted periods. Intraruminal dosing of exogenous enzymes did not affect apparent digestibility of DM, Crude Protein (CP) or Neutral Detergent Fiber (NDF) but reduced rumen pH and the activity of key endogenous fiberolytic enzymes and also increased the soluble DM fraction and effective DM degradability (Hristov et al., 2000). Earlier work by these authors (Hristov et al., 1998) showed that abomasal infusion or dietary supplementation with exogenous enzymes did not increase DM intake, in situ degradation or total tract digestion in cattle. No differences were also found between dietary concentrate or TMR supplementation or rumen infusion with enzymes on DM intake digestibility or milk yield in dairy cows (Sutton et al., 2003). These studies suggest that post-ingestive supply of fiberolytic enzymes is no more effective than dietary supplementation for increasing feed intake, digestion and milk yield in cattle. It is not clear why dietary treatment was not effective in the studies above, since this mode of delivery is the key to harnessing the potential of exogenous enzymes in ruminant nutrition (Wallace et al., 2001).
Ruminal activity and stability of direct-fed cellulase: Cellulase activity is dictated by several factors including presence of inhibitors and co-factors, prevailing pH, moisture, temperature and concentration of enzyme and substrate. A common error is the determination of enzyme activity under conditions that optimize enzyme action but differ considerably from the ruminal environment, such that measured enzyme activity is overestimated. Clearly, if the enzyme is expected to exert most of it’s’ effect in the rumen, the enzyme activity should be measured under conditions that mimic the ruminal environment. Adoption of recently proposed methods for standardizing fiberolytic enzyme activity measurement (Colombatto and Beauchemin, 2003) should help in this regard. Dawson and Tricarico (1999) suggested that the most active period for enzyme effects is in the first 6-12 h of the digestive process, though they also speculated that such action occurs prior to bacterial colonization of feed substrates or action of endogenous enzymes. In support, Newbold (1997) noted that enzymes must function within a few hours of feeding before being degraded by the proteolytic activity of rumen microbes. The likelihood of ruminal proteolysis limited the use of enzymes in ruminant feeds for decades. However, Morgavi et al. (2001) found that four commercial enzymes were stable when incubated in rumen fluid, pepsin or pancreatin and adduced this to carriers and stabilizers, manufacturing processes and enzyme-substrate interactions. Host proteases and the acid pH of the abomasum are more likely to degrade exogenous enzymes than ruminal proteases (Hristov et al., 1998; Morgavi et al., 2001). Sustained enzyme stability in the rumen can result from natural or artificially induced enzyme glycolysation, which involves covalent bonding of monosaccharides to specific aminoacid side chains in enzymes (Van de Vyver et al., 2004). Glycolysation has been shown to confer resistance to proteolysis in monogastrics and ruminal fluid (Van de Vyver et al., 2004), but non glycosylated enzymes may also resist ruminal proteolysis due to adaptation over time and their genetic composition (Fontes et al., 1995).
However several cellulase enzyme preparations are commercially available and lack of response to enzyme treatment in some of the studies may be attributed to ruminal enzyme instability. For instance (Vicini et al., 2003) attributed the lack of response to enzyme treatment in their study to higher ruminal pH and lower ruminal temperature than the optima for the cellulase activities in their enzyme preparation. Therefore there are notable variations in the stability of commercially-available enzyme preparations and their rumen stability should be verified before they are used in practice.
Cellulase- feed specificity and the portion of the diet to which cellulase preparations are applied: The following studies reveal the importance of matching cellulase preparations to specific substrates: Beauchemin et al. (1997) reported greater responses when cellulase preparation was applied to dry forages instead of wet forages. Feng et al. (1996) showed that direct-fed enzymes were more effective when applied to dried grass at feeding than to freshly cut, dried grass at harvest or wilted dried grass after harvest. When the same enzyme was applied to hay and corn silage, it increased the NDF digestion of corn silage but not hay (Siciliano-Jones, 1999).
Also application of the same cellulase preparation to alfalfa and ryegrass increased the digestibility of alfalfa but not ryegrass (Pinos-Rodriguez et al., 2002). Further evidence for enzyme-feed specificity is apparent from studies in which enzymes were added a specific dietary component. Bowman et al. (2002) found that enzyme application to the concentrate (45% of total mixed ration, TMR) instead of a pelleted supplement (4% of TMR) or a premix (0.4% of TMR) did not affect intake, salivation or rumen function but numerically increased fat-corrected milk yield compared to control cows. They therefore concluded that the proportion of the diet to which the enzyme is applied must be maximized to ensure a beneficial response. In contrast (Yang et al., 2000) showed that applying enzymes to the concentrate was more effective than applying them to the total mixed ration in terms of the response in milk yield and digestibility of DM, Organic Matter (OM) and CP. However other studies found no differences in milk yield and intake when enzymes were applied to TMR or forage (Vicini et al., 2003) or to TMR or concentrate (Phipps et al., 2000; Sutton et al., 2003) or to alfalfa cubes and the concentrate (Yang et al., 1999). Since concentrates are ruminally readily fermented and contain low fiber concentrations, the beneficial effects of enzyme addition to this dietary fraction may be due to synergistic effects on microbial populations and endogenous enzyme secretion, than to direct cell wall hydrolysis. Also, the study in which enzyme application to concentrate proved more effective (Yang et al., 2000) had a lower forage to concentrate ratio (38:62) than those (57:43, 57:43, 55:45 and 60:40) in which it did not (Yang et al., 1999; Phipps et al., 2000; Sutton et al., 2003; Vicini et al., 2003). Therefore the effect of the dietary component to which the enzyme is added may depend on the forage to concentrate ratio and the uniformity of enzyme application to that component.
Level of cellulase application: Several studies have shown that applications of high levels of cellulase to forages or diets produce less desirable responses than low levels. For instance Lewis et al. (1999) noted that a medium level of enzyme supplementation produced more milk than a low or high level of application and Beauchemin et al. (2000) found that a high level of cellulase enzyme application was less effective than a low level at increasing total tract digestibility. The reason for the poor response to the low enzyme level is obvious, but that for the higher level is less apparent. It may be partly attributed to negative feedback inhibition which is one of the classical modes of regulation of enzyme action. This feedback mechanism occurs when enzyme action is inhibited by production of a critical concentration of a product of the enzyme-substrate interaction. For instance fermentation of sugars produced by cell wall hydrolysis may reduce ruminal pH to levels that inhibit cell wall digestion. An alternative hypothesis is that excessive enzyme application blocks binding sites for enzymes or may prevent substrate colonization (Beauchemin et al., 2000; Beauchemin et al., 2003). The fact that enzymes can be overfed or underfed makes their application complex (Dawson and Tricarico, 1999) and underscores the need for determining the optimal level of application for each enzyme preparation. A more disconcerting observation is that in vitro evaluation of the activities of two cellulase enzyme revealed that when added at the rates recommended by their manufacturers, the enzymes would not increase significantly glycanase and polysaccharidase activities in rumen fluid unless much higher application rates are used (Wallace et al., 2001). This highlights the need for further in vivo studies to verify the application rates and activities of some commercially available enzymes.
Stage of lactation of dairy animals: Theoretically direct-fed cellulase enzyme supplementation should be most effective when ruminal fiber digestion is compromised due to factors like acidosis, or when dietary glucose supply is inadequate to meet the needs of the ruminants such as in early lactation. In support, direct-fed cellulase enzyme supplementation has increased milk production from cows in early lactation, but not from cows in mid lactation (Schingoethe et al., 1999) and has increased weight gain, milk production and feed intake in early lactation, but not in late lactation (Knowlton et al., 2002). Also when cows in positive energy balance were fed cellulase supplemented diets, increased intake of digestible energy due to enzyme supplementation did not increase milk yield (Beauchemin et al., 2000). In contrast, Lewis et al. (1999) showed that cellulase supplementation increased milk yield in early or mid lactation in two separate experiments. Also Zheng et al. (2000) found that stage of lactation did not affect the increase in milk production due to cellulase supplementation, but concluded that delaying cellulase supplementation till 6 weeks postpartum resulted in a loss of 280 kg of milk in the first 18 week of lactation and therefore recommended starting to feed enzyme-supplemented diets soon after parturition. The discrepancies between the studies cited above are due to factors such as differences in dietary components, forage to concentrate ratio and enzyme composition and activity.