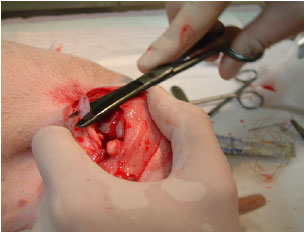
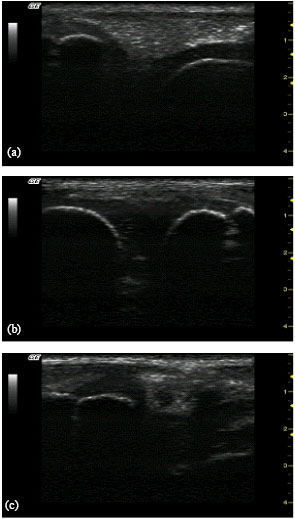
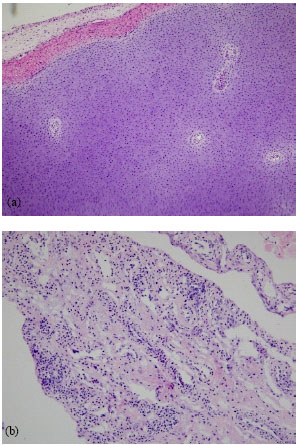
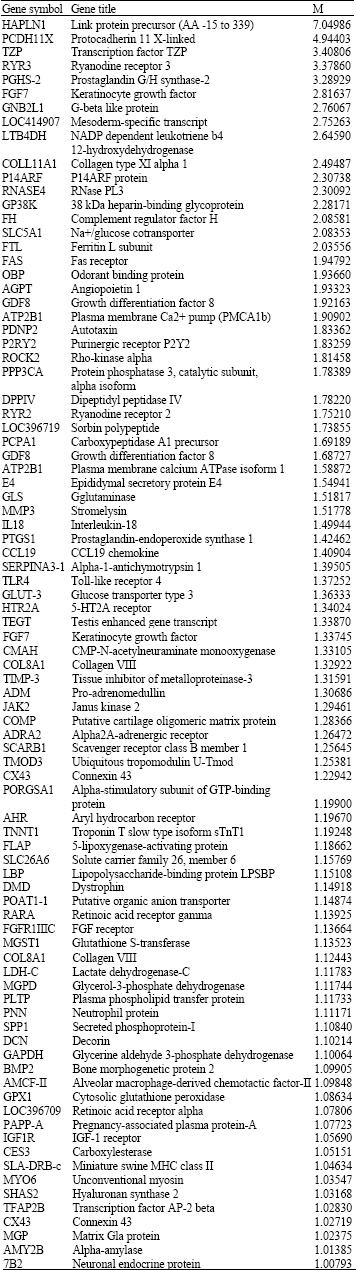
Research Article
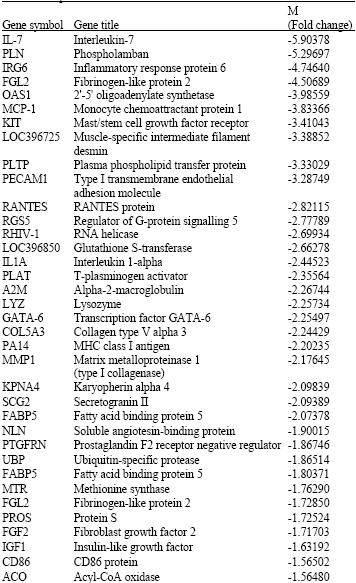
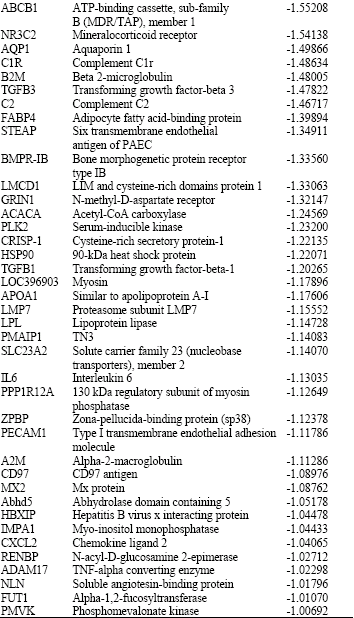
Gene Expression Profile of Synovial Cells in Experimental Post-Traumatic Arthritis of Knee in Swine
School of Biotechnology, Hankyong National University, Ansung-City, 456 749, Korea
K. Choi
School of Biotechnology, Hankyong National University, Ansung-City, 456 749, Korea
P.K. Mandal
School of Biotechnology, Hankyong National University, Ansung-City, 456 749, Korea
Hee-kyong Lim
School of Biotechnology, Hankyong National University, Ansung-City, 456 749, Korea
Guan-Hao Li
Department of Food Science, Agricultural College of Yanbian University, Jilin Province, China
Hong-Sik Kong
School of Biotechnology, Hankyong National University, Ansung-City, 456 749, Korea
Dae-Hyun Hahm
Acupuncture and Meridian Science Research Center, Kyung-Hee University, Seoul, Korea
Chi-Ho Lee
Department of Animal Food Science and Biotechnology, Konkuk University, Seoul, Korea
Jun-Heon Lee
Division of Animal Science and Resources, College of Agriculture and Life Sciences, Chungnam National University, Daejeon 305-764, Korea