Research Article
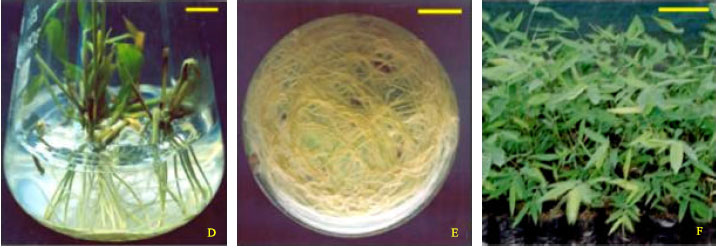
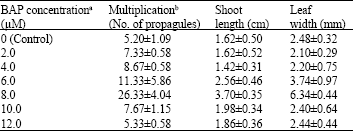
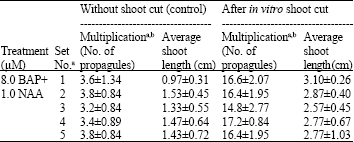
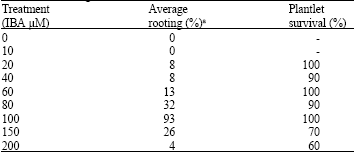
In vitro Shoot Cut: A High Frequency Multiplication and Rooting Method in the Bamboo Dendrocalamus hamiltonii
Biotechnology Applications, G.B. Pant Institute of Himalayan Environment and Development, Kosi-Katarmal, Almora, India
Shyamal K. Nandi
Biotechnology Applications, G.B. Pant Institute of Himalayan Environment and Development, Kosi-Katarmal, Almora, India










kanti priy bhardwajk Reply
This is a great artical