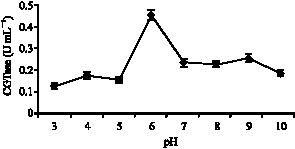
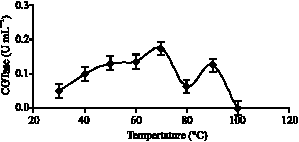
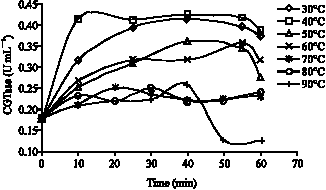
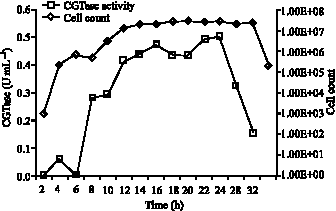
Research Article
Optimization of Cyclodextrin Glycosyltransferase Production by Response Surface Methodology Approach
Faculty of Biotechnology and Biomolecular Sciences, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
Suraini Abd-Aziz
Faculty of Biotechnology and Biomolecular Sciences, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
Norjahan Alitheen
Faculty of Biotechnology and Biomolecular Sciences, Universiti Putra Malaysia, 43400 UPM Serdang, Selangor, Malaysia
Osman Hassan
Faculty of Science and Technology, School of Chemical Sciences and Food Technology, Universiti Kebangsaan Malaysia, 43600 Bangi, Selangor, Malaysia
Mohamed Ismail Abdul Karim
Department of Biotechnology Engineering, Faculty of Engineering, International Islamic University, Jalan Gombak, 53100 Kuala Lumpur, Malaysia