Research Article
Regeneration of plantlets from node-derived callus in Aegle marmelos Corr.
Rajshahi University, Bangladesh
S. Zaman
Rajshahi University, Bangladesh
K. Nasirujjaman Nasirujjaman
Rajshahi University, Bangladesh
Aegle marmelos Corr. locally called Bael, is a slow growing, medium spreading, highly heterozygous, deciduous spiny tree (Prain, 1963) belonging to the family Rutaceae. Among underutilized woody fruit plants of Bangladesh, Aegle marmelos is one of the most familiar ones. It is native to the Indo-Burma Subcontinent (Hooker, 1975) and now being cultivated as a backyard tree in India, Bangladesh, Pakistan, Srilanka, Burma and Thailand (Zaman, 1988). The plant has the capacity to adapt successfully to a wide range of habitats from arid, semi-arid to mesophytic condition (Arya, 1986) having considerable traditional socio-cultural values. It is one of the most important medicinal plants grown in India (Biswas and Ghosh, 1973) having properties of pulp of ripe fruit such as amoebocyte, astringent, digestive, tonic, stomatic and laxative and these have been intensively used in ayurvedic and folk medicine (Sharma, 1980; Hassan, 1988). A type of yellow dye is extracted from the pulp used in fabrication of cloth and leaf bark and root extracts are said to be effective in the treatment of different types of diseases (Biswas and Ghosh, 1973; Samad, 1966). For its heterozygous nature, maintenance of varietal purity is one of the important problems when improving through conventional breeding methods. Due to over exploitation and lack of organized plantation, the wild populations have been declined rapidly in their natural habitat. The loss of valuable genetic diversity may be mitigated by conservation of this species through in vitro methods. Aegle marmelos plants have been regenerated from hypocotyl (Arya et al., 1981); leaf (Islam, et al., 1993); nucellus, cotyledon and embryonic tissue (Hossain et al., 1993). Here we report organogenesis and plant regeneration from node-derived callus in Aegle marmelos.
In vitro grown cotyledon-derived shoots of Aegle marmelos Corr. were excised aseptically and after removal of leaves, 7 mm long node was excised from the shoot. The nodal explants were placed horizontally on MS medium (Murashige and Skoog, 1962) fortified with various concentrations and combinations of BA, 2, 4-D, NAA and IBA for callus induction. In all cases, callus proliferation began at the cut ends of the explants and the rate of growth was rapid. The 42 day-old proliferated calli were subcultured on MS medium containing BA alone and in combination with different composition of NAA and KIN for organogenesis and plantlet formation. For adventitious rooting half strength of MS medium without any growth regulator was used.
Explants were cultured separately in 25 x 150 mm culture tube for each combination of growth hormone. All cultures were maintained in a growth room under 16 h photoperiod with a light intensity 2000 to 3000 lux provided by warm-white fluorescent lights at 26±2°C. All media were solidified with 6 g L-1 agar (Carolina Biological Supply Co.) and supplemented with 30 g L-1 sucrose for callus formation, regeneration and root induction. The pH was adjusted to 5.6 prior to autoclaving at 15 lbs pressure per square inch at 121°C temperature for 20 min. All experiments were repeated for twice.
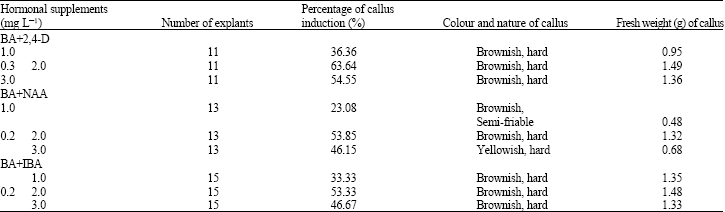
A number of reports indicate that callus initiation and proliferation from nodal explants strictly depended on exogenous growth regulator supplementation and no callus was induced on basal MS medium only. In vitro grown nodal explants were cultured on MS medium with different concentrations and combinations of BA with 2, 4-D, NAA and IBA for callus initiation (Table 1).
Within 28 days of culture, nodal explants showed callus initiation in all the compositions of hormones applied. Among various media compositions, BA+2 ,4-D combination was proved to be the best for callus induction. Rao and Chopra (1987) also found 100% callus induction by using 2, 4-D along with BA. The maximum percentage of callusing (63.64) was found in media having 0.3 mg L-1 BA+2.0 mg L-1 2,4-D (Fig. 1 and Table 1). The highest fresh weight of callus (1.49 g) was also observed in that media composition (Table 1). Lower concentration of cytokinin with higher concentration of auxin showed efficient callus induction. In most of the cases, the colour of the induced callus was brownish and hard in nature but yellowish and semi-friable callus was also found. Early reports of Islam et al. (1992; 1993) have proven the applicability of leaf segment as explant in case of Aegle marmelos tissue culture and they developed an efficient plant regeneration system through organogenesis.
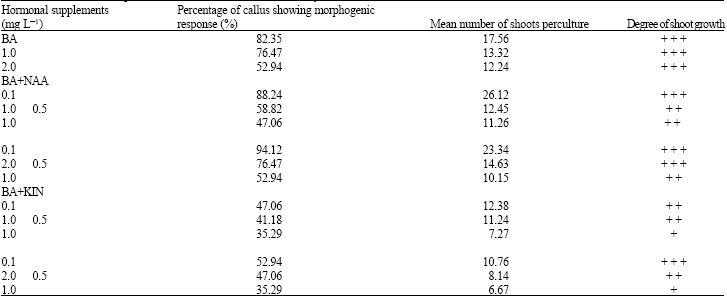
The 42 day-old hard, compact, irregular shaped callus obtained from each explant were cut into smaller pieces and subcultured onto fresh medium containing BA (1-3 mg L-1) alone and in combination with different composition of NAA and KIN for organogenesis (Table 2). Under appropriate conditions of auxin and cytokinin, 42 day-old calli showed the formation of a number of adventitious shoot buds in compact masses. Later, many of these buds developed into small shoots. The best percentage of callus (94.12) showing morphogenic response was obtained using 2.0 mgL-1 BA+0.1 mgL-1 NAA (Fig. 2) with 23.34 shoots per culture and the degree of shoot growth was very good (Table 2).
| Table 1: | Effect of different concentrations and combinations of BA with 2,4-D, NAA and IBA on callus induction from in vitro grown nodal explants of Aegle marmelos [Data collected 42 days after culture initiation] |
 | |
| Table 2: | Effect of BA singly or in combination with KIN and NAA on regeneration of plantlets from node-derived callus of Aegle marmelos [17 explants were used for each experiment. Data were collected after 42 days of subculture of callus] |
 | |
| + = Poor; + + = Good; + + + = Very good | |
Successful morphogenic response has also been achieved using nodal explants of D. caryophyllus (Ghosh, 1986). However, the maximum (26.12) shoots per culture was observed in the media having 1.0 mg L-1 BA+0.1 mg L-1 NAA (Table 2). The frequency of shoot differentiation was directly related with higher concentration of BA.
After excision of the elongated shoots, the remaining explants with small shoots attached to it could be recultured on the same media for further elongation. About 4 cm long elongated shoots were excised and cultured on hormone free half strength of MS medium for root induction and satisfactory root initiation was observed (Fig. 3). The rooted plantlets were successfully transplanted to soil.
 | |
| Fig. 1: | Induction of callus from nodal segment on MS+0.3 mg L-1 BA+2.0 mg L-1 2,4-D after 5 weeks of culture |
 | |
| Fig. 2: | Regeneration of shoots from callus on MS+2.0 mg L-1 BA+0.1 mg L-1 NAA after 7 weeks of culture |
 | |
| Fig. 3: | Development of adventitious roots on micropropagated shoot in hormone free ½ MS medium, 5 weeks after subculture |
The present study has demonstrated that plant regeneration could be achieved using callus cultures from nodal tissues of Aegle marmelos. Earlier, it has been shown that complete plant regeneration is possible from hypocotyls and cotyledons (Arya et al., 1981) and leaf segments (Islam et al., 1992) in this woody plant. Successful microprpagation of A. vasica using nodal explant culture has been reported by Jaiswal et al. (1989). The use of in vitro grown nodal explants has several benefits. Microbial contamination has never been a serious problem.
Efficient recovery of Aegle marmelos plants through the process mentioned will facilitate storage and international exchange of germplasm and can be exploited for selecting valuable genotypes.