Research Article
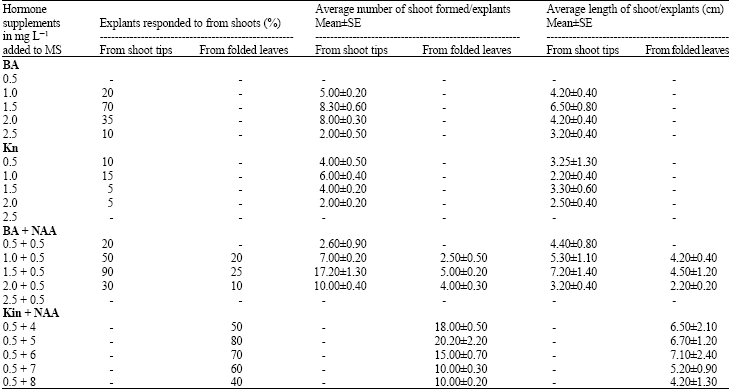
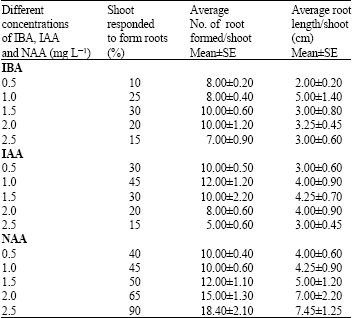
In vitro Mass Propagation of Sugarcane (Saccharum officinarum L.) var. Isd 32 through Shoot tips and Folded Leaves Culture
Atomic Energy Research Establishment, Ganakbari, Savar, Dhaka, Bangladesh
M. H. Kabir
Atomic Energy Research Establishment, Ganakbari, Savar, Dhaka, Bangladesh