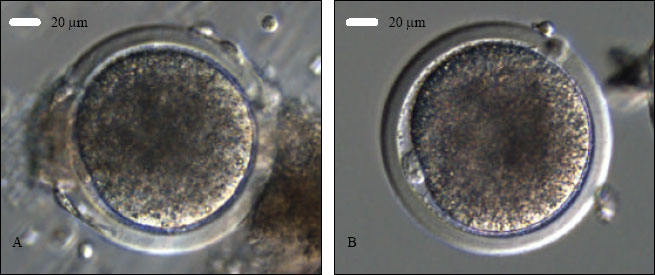
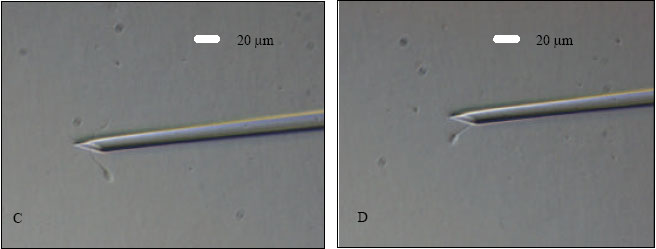
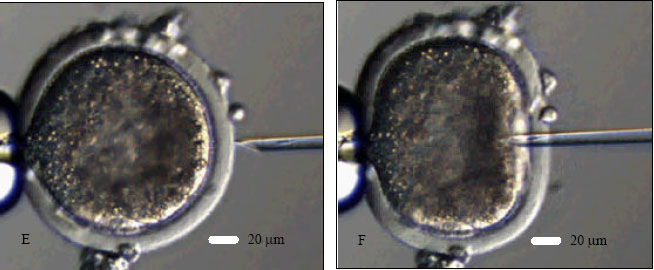
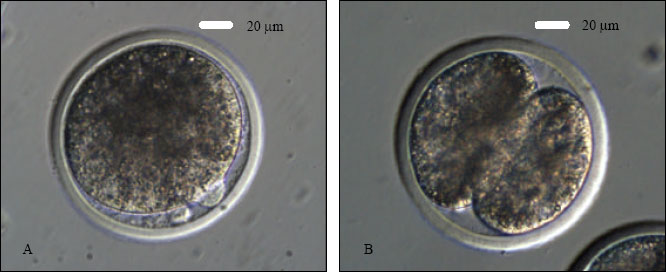
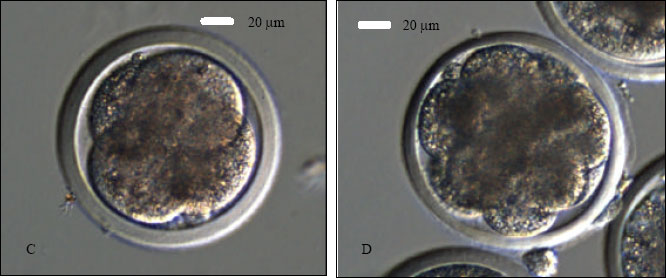
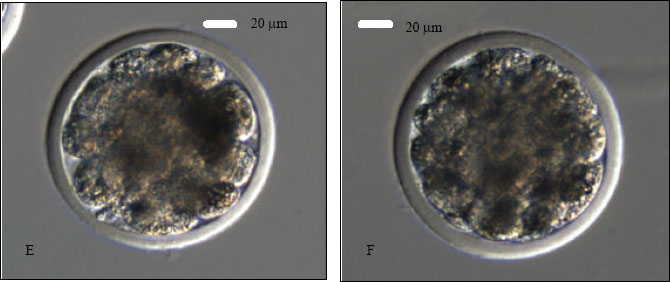
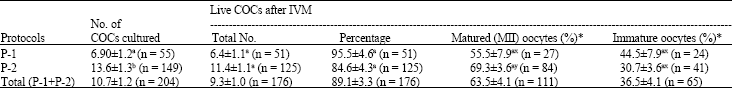Research Article
Goat Embryo Development from in vitro Matured Oocytes of Heterogeneous Quality Through Intracytoplasmic Sperm Injection Technique
School of Agriculture and Rural Development, Bangladesh Open University, Gazipur-1705, Bangladesh
Ramli Bin Abdullah
Animal Biotechnology-Embryo Laboratory (ABEL), Institute of Biological Sciences, University of Malaya, Lembah Pantai, 50603 Kuala Lumpur, Malaysia
Wan Khadijah Wan Embong
Animal Biotechnology-Embryo Laboratory (ABEL), Institute of Biological Sciences, University of Malaya, Lembah Pantai, 50603 Kuala Lumpur, Malaysia