ABSTRACT
The effects of sucrose and polyethylene glycol (PEG) 8000 on production of somatic embryogenesis of date palm cv. Sukary were investigated. Treatments of the experiment were composed of in vitro media that included, 0, 30 and 60 g L-1 sucrose alone or combined with PEG-8000 at 5% level. The results indicated that sucrose at a high level of 60 g L-1 improved production of embryos. Furthermore, the production of embryos, however, was greatly enhanced by the addition of PEG-8000 at 5% with 60 g L-1 sucrose. On this medium, the embryo germination and maturation was also enhanced. Adding PEG-8000 to 60 g L-1 sucrose media induce stress condition during embryos formation and development. This is the first report on the use of sucrose and polyethylene glycol when date palm cv. Sukary are used as explant.
PDF Abstract XML References
How to cite this article
DOI: 10.3923/biotech.2006.466.470
URL: https://scialert.net/abstract/?doi=biotech.2006.466.470
INTRODUCTION
The increasing demand for date palms requires massive multiplication of selected plants. This could be achieved through tissue culture. Somatic embryogenesis is the development of embryos from somatic cells. This is achieved via a series of developmental stages, most of which are similar to zygotic embryogenesis (Svobodova et al., 1999).
The production of well developed somatic embryos in tissue cultured date palm (Phoenix dactylifera L.) is affected by many interrelated factors, among which are the nature of carbon source and osmotic adjusting agents (Litz, 1986; Ramarosandratana et al., 1999). Sucrose has been used as a carbon source for the successful induction of somatic embryogenesis in plants (Custers et al., 1988; Chee, 1990; Cade et al., 1991). Lou and Kako (1994) had improved somatic embryogenesis almost 100% by increasing the sucrose concentration of the initiation medium to 0.26 or 0.35 M. The elevated sucrose concentrations normally affect osmotic potential of medium resulting in improved somatic embryogenesis (May and Trigiano, 1991). Furthermore, it was also noted that somatic embryo-derived seedlings from elevated sucrose media had better chances for acclimatization and successful growth vigor (Saranga et al., 1992). Depending on the plant species, other carbon sources may also promote somatic embryogenesis (Swedlund and Locy, 1993).
Increased medium osmolarity and hence the frequency of somatic embryogenesis was also obtained by the addition of polyethylene glycol (Brown et al., 1989; Kavi Kishor, 1987; Ramarosandratana et al., 2001; Steiner et al., 2005; LuShou et al., 2005). Ramarosandratana et al. (1999) reported that addition of polyethylene glycol (PEG) 4000 at 6% greatly enhanced the production of mature embryos by 2 to 4 times. Furthermore, the embryo length, depending on the sugar was significantly increased by 10 to 20%. The survival of PEG treated embryos was greatly improved and the resulting plantlets were successfully acclimatized with better growth vigor. In many studies showed that only non-penetrating osmotica are able to promote the maturation of embryos (Ramarosandratana et al., 1999; Svobodova et al., 1999). This kind of osmotica restricts water uptake and, hence, provides a natural drought stress during embryos development. The best results were obtained with polyethylene glycol 4000, a non-plasmolyzing osmoticum which cannot penetrate into the plant cell but can induce water stress. Azpeitia Morales et al. (2003) found that in the induction of somatic embryos of Cocus nucifera the best treatment was 90 μM ABA+15 g PEG L-1 with the formation of 10.6 somatic embryos per embryogenic callus at 150 days of culture and formation of three well developed plantlets per embryogenic callus after 210 days of culture. Chen and Dribnenki (2004) reported that replacing 15% sucrose with 6% sucrose and 9% polyethylene glycol (PEG) or 3% sucrose and 12% PEG in pre-culture medium did not significantly affect callus induction and shoot regeneration of flax. Their results indicate that sucrose may act as carbon/energy source as well as an osmatic regulator in flax anther culture. Sucrose as an osmotic regulator may be replaced by a non-metabolizable osmoticum: PEG.
The objectives of the present study, were to determine the effects of PEG and different sucrose concentrations on somatic embryogenesis and also to determine whether the improvement of somatic embryogenesis is supported by osmotic potential resulted from sugar concentrations and PEG as well as combined action of the two.
MATERIALS AND METHODS
Offshoots taken from adult date palm (Phoenix dactylifera L.) cv. Sukary were used as plant material. Terminal shoot tip and surrounding leaf primordia excised from offshoots were cultured on MS salts (Murashige and Skoog, 1962) supplemented with 170 mg-1 NaH2PO4.2H2O, Inositol 125 mg-1, Glutamine 200 mg-1, Thiamine 5 mg-1, Pyridoxine HCl 1 mg-1, Nicotinic acid 1 mg-1, Glycine 2 mg-1, Sucrose 30 g-1, Activated charcoal 1.5 g-1 and Purified agar 6.5 g-1. In order to form embryogenic callus culture were transferred in 3 media described below:
| • | MS media supplemented with 100 mg-1 2,4-D and 3 mg-1 2 ip for culture initiation medium. Culture were incubated at 25±2°C in darkness for 9 weeks. |
| • | MS media supplemented with 10 mg-1 NAA and 30 mg-1 2 ip for culture swelling medium. Culture were incubated at 25±2°C in a 16 h photo period provided from cool white florescent lamps for 3 weeks only. |
| • | MS media supplemented with 10 mg-1 NAA and 6 mg-1 2 ip for embryogenic callus formation. Culture were incubated at 25±2°C in a 16 h photo period provided from cool white florescent lamps for 16 weeks only. |
Media pH was adjusted to 5.7 and sterilized by autoclaving for 15 min at 121°C. Cultures were transferred to fresh media at 4 week intervals. The resultant callus used for the experiment. Treatments to increase osmolarity of the media include the addition of 0, 3 and 6% sucrose alone or combined to 5% PEG MW 8000 (Sigma). Treatments were replicated 10 times and the cultures were incubated at 25±2°C in 16 h of light daily supplied by 65/80 Warm White Weisse 3500 fluorescent tubes.
Data were taken after 5 weeks of culture as followed:
| • | Total number of somatic embryos. |
| • | Total number of germinating embryos. |
| • | Length of embryo (cm). |
| • | Fresh and dry weights of culture (g). |
Data subjected to statistical analysis as a randomized complete were design according to Gomez and Gomez (1984). The treatment means were compared using Least Significant Difference (LSD) at 5% level of probability according to Waller and Duncan (1969). All statistical analysis was performed using the facility of computer and SAS software package (SAS, 2001).
RESULTS AND DISCUSSION
The results indicated that addition of sucrose or polyethylene glycol (PEG) enhanced all studied characters (Fig. 1 and 2). Sucrose at 30 or 60 g L-1 improved production of embryos and their germination especially at high concentration. However, addition of PEG 8000 to sucrose treatments substantially increased embryos production, germination, fresh and dry weights. The corresponding increments in embryo No. with adding PEG were 82.1, 93.2 and 34.1% as compared with medium containing 0, 30 or 60 g L-1 sucrose per se, respectively.
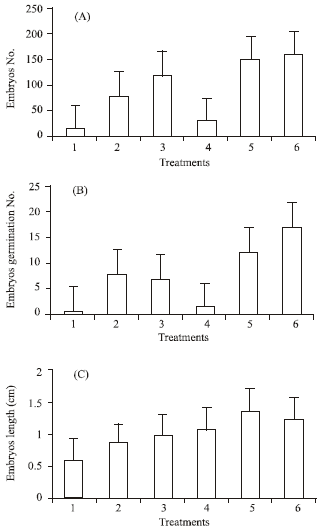 | |
| Fig. 1: | Effect of sucrose and PEG concentration on number of embryos (A), number of embryos germinated (B) and it length (cm) (C) of date palm cv. Sukary, 1 = without sucrose and PEG, 2 = 30 g L-1 sucrose, 3 = 60 g L-1 sucrose, 4 = 0 sucrose+5% PEG, 5 = 30 g L-1 sucrose+5% PEG and 6 = 60 g L-1 sucrose+5% PEG |
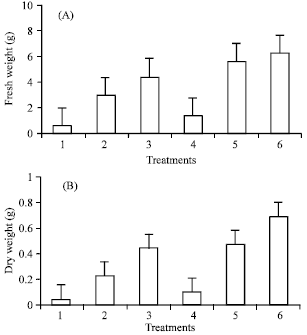 | |
| Fig. 2: | Effect of sucrose and PEG concentration on fresh (A) and dry weights (B) of date palm cv. Sukary 1 = without sucrose and PEG, 2 = 30 g L-1 sucrose, 3 = 60 g L-1 sucrose, 4 = 0 sucrose+5% PEG, 5 = 30 g L-1 sucrose+5% PEG and 6 = 60 g L-1 sucrose+5% PEG |
Production of embryos (158.4), however, was greatly enhanced by the addition of PEG at 5% with 60 g L-1 sucrose media. The same media had also effectively improved germination, length, fresh and dry weights of embryos (Fig. 1 and 2). In 30 g L-1 sucrose treatments, supplementation of PEG to the media seemed to provide to a lesser extent, relatively better quantity and quality of embryos. It is quite probable that addition of PEG decreases the osmotic potential of the media and consequently improves somatic embryogenesis production. However, results of the experiment revealed that addition of PEG 8000 at 5% with 60 g L-1 sucrose media seemed to be the optimum levels of both variables to adequately improve somatic embryogenesis production in date palm cv. ‘Sukary’ (Fig. 3). Roots of date palm have been developed readily after transfer of somatic embryos to half-strength MS medium supplemented with 0.1 mg L-1 NAA (Fig. 4). The formation of roots was differ among the treatments (Data not shown)
The results reported herein are in line with those obtained by Steiner et al. (2005) in Araucaria angustifolia, LuShou et al. (2005) in Larix kaempferi, Agarwal et al. (2004) in Morus alba and Capuana and Debergh (1997) in Aesculus hippocastanum.
 | |
| Fig. 3: | Effect of sucrose and PEG on somatic embryogenesis in date palm cv. Sukary 1 = without sucrose and PEG, 2 = 30 g L-1 sucrose, 3 = 60 g L-1 sucrose, 4 = 0 sucrose+5% PEG, 5 = 30 g L-1 sucrose+5% PEG and 6 = 60 g L-1 sucrose+5% PEG |
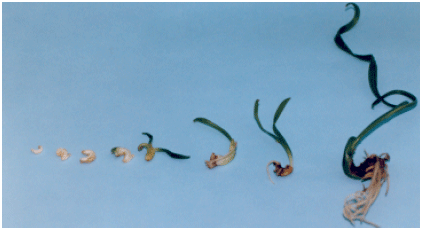 | |
| Fig. 4: | The sequence development of somatic embryo date palm |
However, Korbes and Droste (2005) found that in soybean plants the addition of PEG did not affects the number of embryos converted into plants compared to sucrose.
The possible explanation for increasing dry weight and the number of embryos was that the osmotic potential of media was more negative due to high sugar concentration plus adding PEG at 5%. Therefore, the tissues were forced to accumulate more organic solutes and sugar for osmotic adjustment, this accumulation of sugar may be used in production of more embryos. These results are in agreement with those obtained by Handa et al. (1982) who found that when PEG added to the medium the cells accumulate more sugars and amino acids which cause the enhancement of embryos production and maturation. PEG application also caused the accumulation of total free amino acids, proline, total soluble phenols, sugars (reducing, non-reducing and total) as well as Na and Cl concentrations (Harb et al., 2005) on banana cv. Williams.
Brown and Thorpe (1980) found that in Nicotiana, during shoot initiation processes, exogenously supplied sucrose is used in the ration one-thirds to two-thirds. Also, in Nicotiana, Cousson and Van (1983) reported that flowering processes require some soluble sugar acting as an osmoregulator. In Potato, Khuri and Moorby (1995) found that sucrose acts primarily as a suitable carbon source for uptake and utilization by the plantlets, but at 80 g L-1, it also provides a favorable osmolarity for the development of microtubers. Furthermore, Lou and Kako (1995) demonstrated that the specific carbon effect is a critical stimulus for cucumber somatic embryogenesis and also that osmotic potential provided by sugars has a promotive effect but of a lesser magnitude than the specific carbon effect. Therefore, it seem that in date palm embryos formation depend on the sugar concentration and may used in the ratio of 1/3 to 2/3 for osmoregulation and as carbon energy source, respectively since adding PEG to this medium synergistically enhances embryos formation as shown in the results (Fig. 3).
The results show that somatic embryos could be induced in embryogenic callus of date palm cv. Sukary. This was achieved by culturing embryogenic callus in media supplemented with 6% sucrose or in combination with PEG-8000. This regeneration system could be used for: (i) continuous regeneration of somatic embryos for production of date palm in large scale; (ii) genetic transformation studies.
In conclusion, this study has demonstrated the possibility of increasing the number of somatic embryos of date palm cv. Sukary by adding PEG at 5% to the 60 g L-1 sucrose. Therefore, further study on the specific role of high sugar concentration and PEG during the process of somatic embryo formation seems imperative. The study may examine the demand for energy or/and the stress caused by such high concentration.
REFERENCES
- Agarwal, S., K. Kanwar and D.R. Sharma, 2004. Factors affecting secondary somatic embryogenesis and embryo maturation in Morus alba L. Sci. Hortic., 102: 359-368.
Direct Link - Brown, C., F.J. Brooks, D. Pearson and R.J. Mathias, 1989. Control of embryogenesis and organogenesis in immature wheat embryo callus using increased medium osmolarity and abscisic acid. J. Plant Physiol., 133: 727-733.
CrossRefDirect Link - Cade, R.M., T.C. Wehner and F.A. Blazich, 1991. Somatic embryos derived from cotyledons of cucumber. J. Am. Soc. 115: 691-696.
Direct Link - Capuana, M. and P.C. Debergh, 1997. Improvement of the maturation and germination of horse chestnut somatic embryos. Plant Cell Tissue Org. Cult., 48: 23-29.
CrossRef - Chen, Y.R. and P. Dribnenki, 2004. Effect of medium osmotic potential on callus induction and shoot regeneration in flax anther culture. Plant Cell Rep., 23: 272-276.
CrossRef - Cousson, A. and K.T.T. Van, 1983. Light-and sugar-mediated control of direct de novo flower differentiation from tobacco thin cell layers. Plant Physiol., 72: 33-36.
PubMed - Gomez, K.A. and A.A. Gomez, 1984. Statistical Procedures for Agricultural Research. 2nd Edn., John Wiley and Sons Inc., Hoboken, New Jersey, ISBN: 978-0-471-87092-0, Pages: 704.
Direct Link - Handa, A.K., R.A. Bressan, S. Handa and P.M. Hasegawa, 1982. Characteristics of cultured tomato cells after prolonged exposure to medium containing polyethylene glycol peg. Plant Physiol., 69: 514-521.
PubMed - Kavi Kishor, P.B., 1987. Energy and osmotic requirement for high frequency regeneration of rice plants from long-term cultures. Plant Sci., 48: 189-194.
Direct Link - Khuri, S. and J. Moorby, 1995. Investigations into the role of sucrose in potato CV. Estima microtuber production in vitro. Ann. Bot., 75: 295-303.
Direct Link - Korbes, A.P. and A. Droste, 2005. Carbon sources and polyethylene glycol on soybean somatic embryo conversion. Pesq. Agropec. Bras., 40: 211-216.
Direct Link - Lou, H. and S. Kako, 1994. Somatic embryogenesis and plant regeneration in cucumber. HortScience, 29: 906-909.
Direct Link - Murashige, T. and F. Skoog, 1962. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant., 15: 473-497.
CrossRefDirect Link - Svobodova, H., J. Allbrechtova, L. Kumstyrova, H. Lipavska, M. Vagner and Z. Vondrakova, 1999. Somatic embryogenesis in norway spruce anatomical study of embryo development and influence of polyethylene glycol on maturation process. Plant Physiol. Biochem., 37: 209-221.
Direct Link - Swedlund, B. and R.D. Locy, 1993. Sorbitol as the primary carbon source for the growth of embryogenic callus of maize. Plant Physiol., 103: 1339-1346.
Direct Link - Waller, R.A. and D.B. Duncan, 1969. A bayes rule for the symmetric multiple comparisons problem. J. Am. Stat. Assoc., 64: 1484-1503.
CrossRefDirect Link - Ramarosandratana, A., L. Harvengt, E. Garin, F. Canlet, M. Paques and R. Calvayrac, 1999. Importance of carbohydrate source and peg on maturation of Pinus Pinaster somatic embryos yield morphology and conversion rate. Proceedings of the 4th Biotechnology Conference Advances in Tissue Culture and Transformation.








