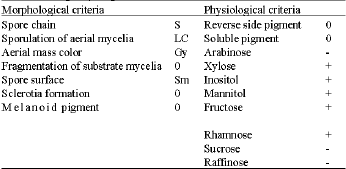
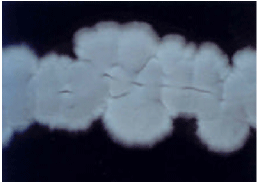
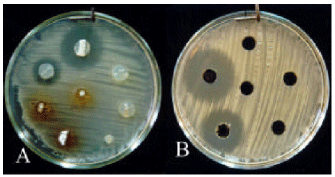
Research Article
First Report of Antibacterial Properties of a New Strain of Streptomyces plicatus (Strain 101) Against Erwinia carotovora subsp. Carotovora from Iran
Department of Plant Pathology, College of Agricultural Sciences, Bahonar University of Kerman, Iran
G. H. Shahidi Bonjar
Department of Plant Pathology, College of Agricultural Sciences, Bahonar University of Kerman, Iran
I. Saadoun
Department of Applied Biology, Jordan University of Science and Technology, P.O. Box 3030, Irbid 22110, Jordan