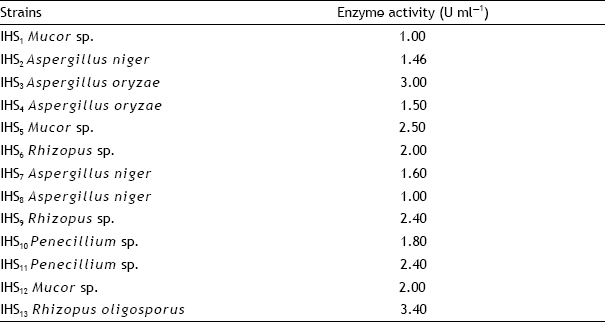
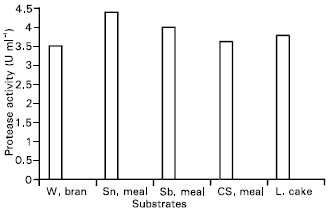
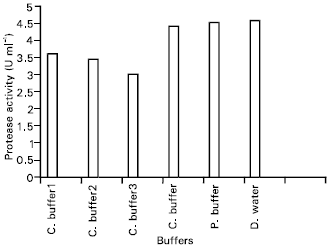
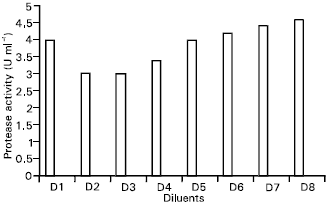
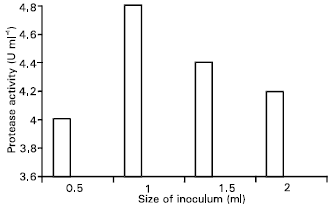
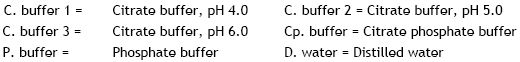Research Article
Production of Proteases by a Locally Isolated Mould Culture under Lab Conditions
Government College University, Lahore, Pakistan
Hamid Mukhtar
Government College University, Lahore, Pakistan
Sunila Daudi
Government College University, Lahore, Pakistan
Sikander Ali
Government College University, Lahore, Pakistan
M. A. Qadeer
Government College University, Lahore, Pakistan