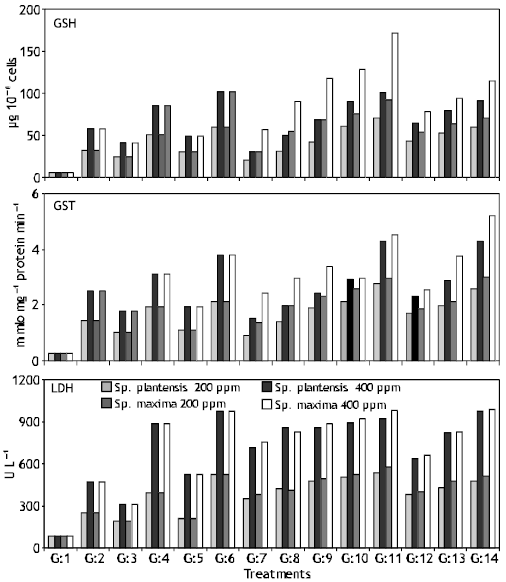
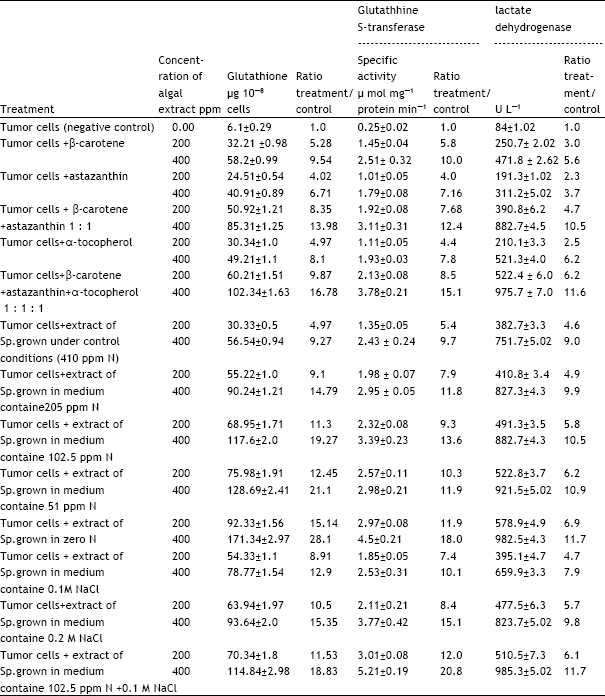
Research Article
Spirulina Species as a Source of Carotenoids and a-Tocopherol and its Anticarcinoma Factors
National Research Center, Cairo, Egypt
Farouk K. El Baz
National Research Center, Cairo, Egypt
Gamal S. El-Baroty
Faculty of Agriculture, Cairo University, Pakistan