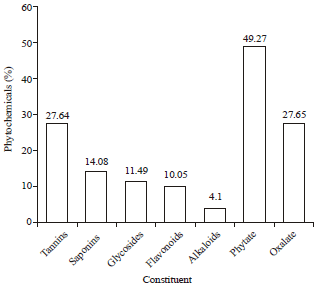
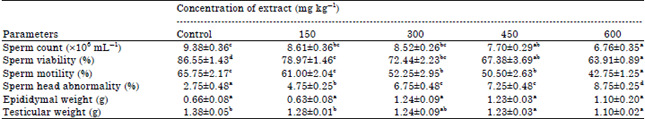
Research Article
Phytochemistry and Reproductive Activities of Male Albino Rats Treated with Crude Leaf Extract of Great Bougainvillea (Bougainvillea spectabilis)
Department of Genetics and Biotechnology, University of Calabar, PMB 1115, Calabar, Nigeria
U.B. Ekaluo
Department of Genetics and Biotechnology, University of Calabar, PMB 1115, Calabar, Nigeria
O.U. Udensi
Department of Genetics and Biotechnology, University of Calabar, PMB 1115, Calabar, Nigeria
E.E. Ekerette
Department of Genetics and Biotechnology, University of Calabar, PMB 1115, Calabar, Nigeria
M. Pius
Department of Genetics and Biotechnology, University of Calabar, PMB 1115, Calabar, Nigeria