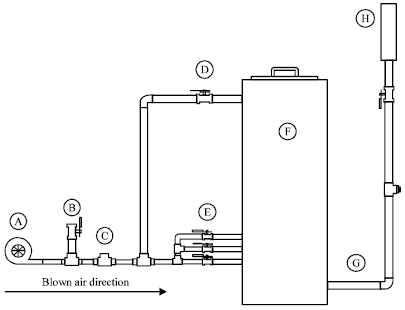
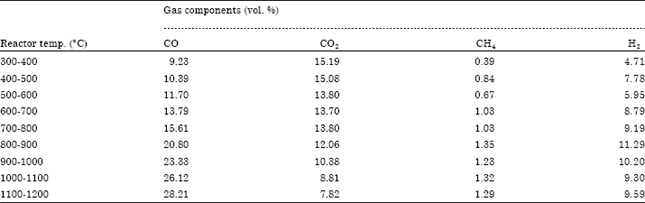
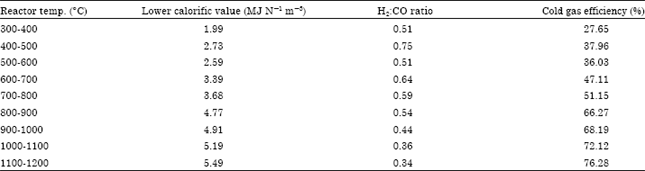
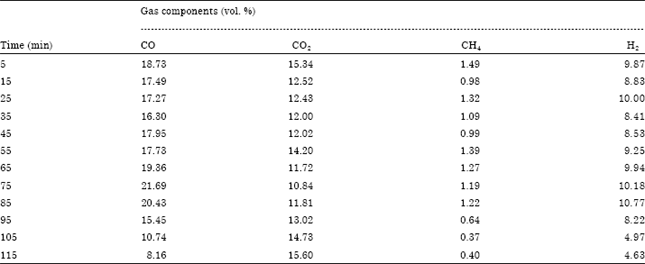
Research Article
Downdraft Gasification of Oil Palm Frond: Effects of Temperature and Operation Time
Department of Mechanical Engineering, Universiti Teknologi PETRONAS, Bandar Seri Iskandar, 31750 Tronoh, Perak, Malaysia
Shaharin A. Sulaiman
Department of Mechanical Engineering, Universiti Teknologi PETRONAS, Bandar Seri Iskandar, 31750 Tronoh, Perak, Malaysia