Research Article
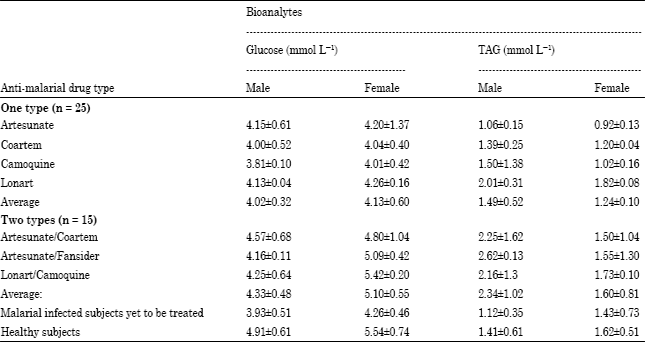
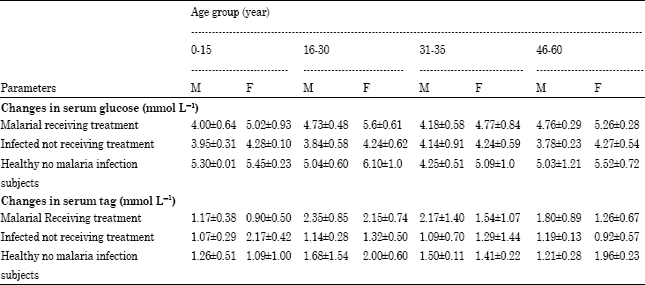
Changes in Serum Glucose and Triacylglycerol Levels Induced by the Co-administration of Two Different Types of Antimalarial Drugs among Some Plasmodium falciparum Malarial Patients in Edo-delta Region of Nigeria
Department of Medical Biochemistry, Delta State University, Abraka, Nigeria
J. E. Agho
Department of Medical Biochemistry, Delta State University, Abraka, Nigeria