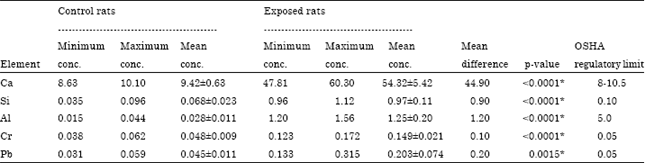
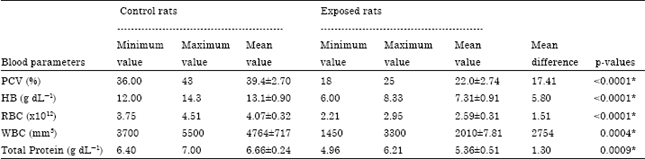
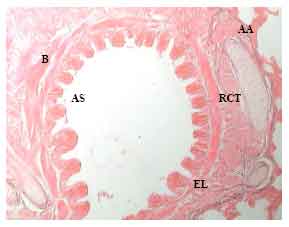
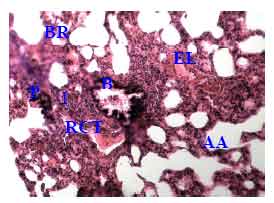
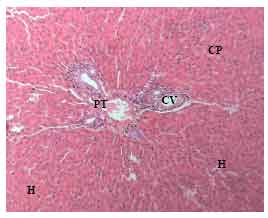
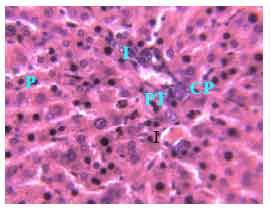
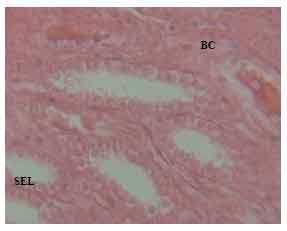
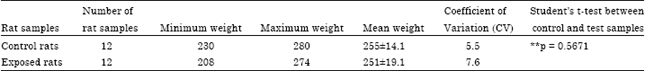Research Article
Investigation of General Effects of Cement Dust to Clear the Controversy Surrounding its Toxicity
Department of Cell Biology and Genetics, University of Lagos, Nigeria
Joy Okpuzor
Department of Cell Biology and Genetics, University of Lagos, Nigeria
Adedayo Titilola Fausat
Department of Cell Biology and Genetics, University of Lagos, Nigeria