Research Article
An Evaluation of Influence of Citrus paradisi Seed Extract on Doxorubicin-Induced Testicular Oxidative Stress and Impaired Spermatogenesis
Department of Anatomy, Lagos State University College of Medicine (LASUCOM), Ikeja, Lagos, Nigeria
A.A. Osinubi
Department of Anatomy, College of Medicine, University of Lagos, Lagos, Nigeria
P.I. Jewo
Department of Anatomy, Lagos State University College of Medicine (LASUCOM), Ikeja, Lagos, Nigeria
A.O. Oyewopo
Department of Anatomy, College of Medicine, University of Ilorin, Ilorin, Nigeria
G.O. Ajayi
Department of Medical Biochemistry, Lagos State University College of Medicine (LASUCOM), Ikeja, Lagos, Nigeria









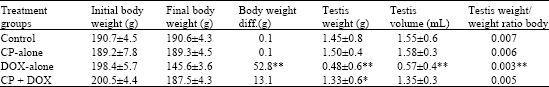

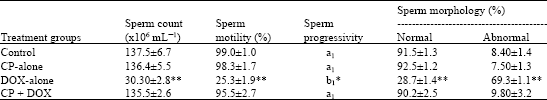
emmanuel ogundimu. Reply
the authors of this journal are successful in making their research. therefore, this work is practicable.