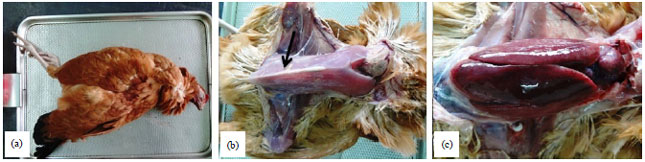
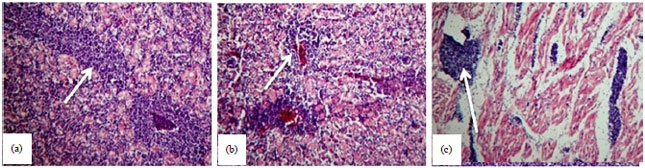
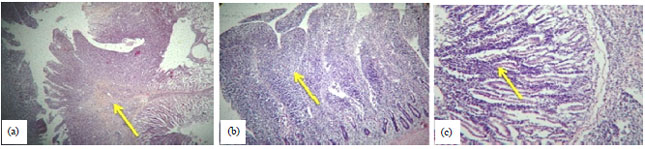
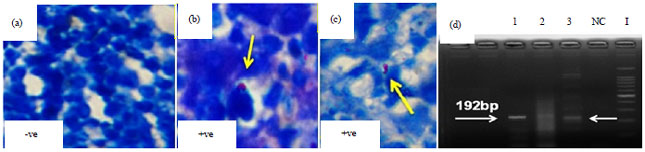
Research Article
Standardize Polymerase Chain Reaction (PCR) Technique for the Detection of Pathogenic Serovars of Mycobacterium a. avium Infection in Layer Chicken
Department of Pathology, Faculty of Veterinary Science, Bangladesh Agricultural University, 2202 Mymensingh, Bangladesh
U.K. Rima
Department of Pathology, Faculty of Veterinary Science, Bangladesh Agricultural University, 2202 Mymensingh, Bangladesh
M.Z. Hossain
Department of Pathology, Faculty of Veterinary Science, Bangladesh Agricultural University, 2202 Mymensingh, Bangladesh
M.S. Islam
Department of Pathology, Faculty of Veterinary Science, Bangladesh Agricultural University, 2202 Mymensingh, Bangladesh
S.M.Z.H. Chowdhury
Department of Pathology, Faculty of Veterinary Science, Bangladesh Agricultural University, 2202 Mymensingh, Bangladesh
M.M. Hossain
Department of Pathology, Faculty of Veterinary Science, Bangladesh Agricultural University, 2202 Mymensingh, Bangladesh
LiveDNA: 880.1453
M.A.H.N.A. Khan
Department of Pathology, Faculty of Veterinary Science, Bangladesh Agricultural University, 2202 Mymensingh, Bangladesh