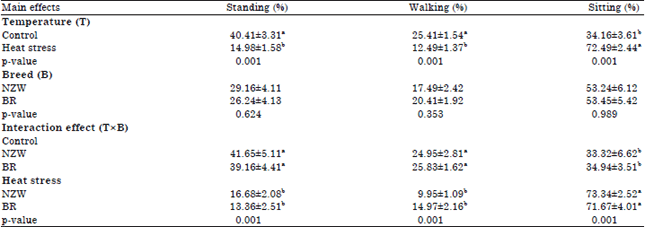
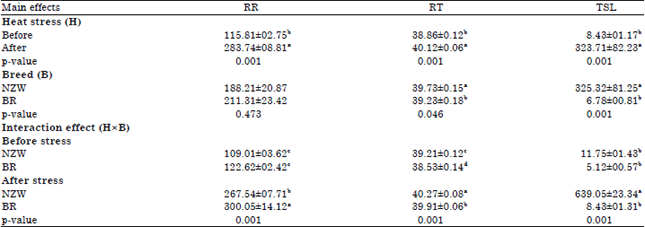
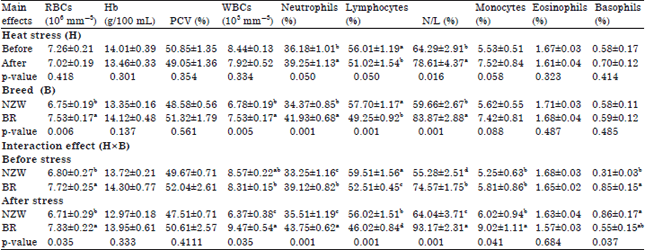
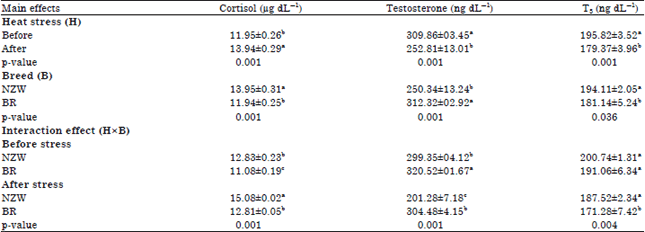
Research Article
Behavioral Activities, Physiological Body Reactions, Hematological Parameters and Hormonal Profiles for Bucks of New Zealand White and Baladi Red Rabbits Exposed to Short Term of High Temperature
Department of Animal Production, Faculty of Agriculture, Suez Canal University, 41522, Ismailia, Egypt
Mohamed A. Yaseen
Department of Animal Production, Faculty of Agriculture, Suez Canal University, 41522, Ismailia, Egypt
Akrum M.M. Hamdy
Department of Animal Production, Faculty of Agriculture, Minia University, Minia, Egypt