ABSTRACT
The natural distribution of two essential fatty acids for animal, namely linoleic and alpha linolenic acid, is not in same order. While the distribution of linoleic acid is cosmopolitan, alpha linolenic acid distribution is very much restricted more confined to marine environment. The seven days old chicks of Gallus domesticus, broiler strain, were fed a diet supplemented with Staphylococcus, Streptococcus and Pseudomonas bacterial strains, collected from the west coast of India, for a period of 30 days. These bacterial strains contained about 15-20% of alpha linolenic acid (as essential fatty acid) when grown on sodium acetate medium. Dietary supplementation of these bacteria increases the net weight of the birds in comparison to the control birds. This net weight gain of the bird was also reflected in total protein, triglycerides, cholesterol and phospholipid concentrations of various tissues. Supplementation of these bacteria as a source of alpha linolenic acid in birds enhanced the tissue level eciosapentaenoic and docosahexaenoic fatty acids at the cost of palmitic, oleic, linoleic and arachidonic acid. A reduction in the serum total cholesterol, total triglycerides along with an increase of HDL cholesterol was noticed in the birds whose diet was supplemented with Streptococcus strain of bacteria only. The cardiac and liver function tests confirmed the well being status of the birds supplemented with Streptococcus strain. The involvement of dietary alpha linolenic acid towards the metabolism of cholesterol in birds was noticed in the Gallus.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajpsaj.2014.64.74
URL: https://scialert.net/abstract/?doi=ajpsaj.2014.64.74
INTRODUCTION
Even though essential fatty acid requirement for poultry birds as well as in other animals is known, little information is made available to the practical nutritionist regarding the metabolic and physiological importance of alpha linolenic acid, one of the two essential fatty acids in animals. Linoleic acid (9,12, octadecaenoic acid) and alpha linolenic acid (9,12,15, ocatdecatrienoic acid) are accepted as essential fatty acids for all animals including the human beings. Dietary linoleic acid may protect chicks from nutritional encephalomalacia induced by vitamin E deficiency (Budowski and Crawford, 1986), an absolute requirement for alpha linolenic acid in poultry has not been demonstrated. Dietary fatty acids including linoleic and linolenic acids are readily absorbed through the intestinal wall where resynthesis of triglycerides and the packaging of lipid into proto microns occur for transport to the liver. Metabolic machinery of birds is altered during post hatching development depending upon the type and proportion of metabolite consumed along with the diet (Halle, 1999). The metabolic utilization of dietary fat affects the carcass composition and meat quality in farmed animals (Doreau and Chilliard, 1997) including the poultry birds (Mieczkowska et al., 1999). Sven et al. (2000) and Perez-Bonilla et al. (2012) showed that by manipulating hen’s diet the quality of the eggs can be altered for better human consumption along with increased reproductive performance of the bird. The dietary fatty acid influence the fatty acid composition of egg yolk and the developing chick embryo utilized the essential fatty acid. Rinogi et al. (2000) showed that, the daily heat production of cocks fed the diet with fish oil, rich in PUFA, was found to be higher than that in those fed with control diet containing soya bean oil and linseed oil. This result also suggests that in poultry long chain PUFA induces thermo genesis, particularly during daylight. Watkins (1995) reviewed the dietary and hormonal control of PUFA formation and the role of PUFA in eicosanoids production during cell differentiation, ovi-position and bone cell metabolism of poultry birds. There is an absolute dearth of knowledge about the exact quantity of linoleic and alpha linolenic acid required in the diet in order to maintain animals in a “well being condition” (with proper growth and sound health).
Polyunsaturated fatty acids cannot be synthesized de novo in animals. They are involved in temperature, salinity adaptation and production of other secondary metabolites. These metabolites in turn maintain the well being state of the animals. We have demonstrated that supplementation of omega 3 enriched fish oil to the growing birds enhanced net weight of bird by 35% more (Roy et al., 2008). Perhaps linolenic acid, the precursor of omega 3 polyunsaturated fatty acid can also enhance the growth rate of the bird. The natural distribution of linoleic and alpha linolenic acids is not in a same order. Synthesis of linoleic acid from oleic acid is very common among the terrestrial, freshwater and marine microbes and plants. Alpha linolenic acid which is a desaturated product of linoleic acid, is very restricted in terrestrial ecosystem but abundant in marine ecosystem. El Razak et al. (2013) isolated new Polyunsaturated Fatty Acids (PUFAs) bacterial producers especially Eicosapentaenoic Acid (EPA) and Docosahexaenoic Acid (DHA). We have isolated some bacterial strains from the marine sediment samples of the west coast of India. These strains showed quite higher concentration of alpha linolenic acid when grown on sodium acetate medium (Pujari et al., 2004). Hence, in the present study an emphasis is being taken to find out whether some of these marine bacteria can be used as an alternative source of alpha linolenic acid (omega-3 fatty acid) and to study its effect on the growth and health of poultry birds, Gallus domesticus, during post hatching development period.
Marine bacteria are known to produce wide range of compounds which have potential applications as bioactive compounds (Devi et al., 2011), probiotics and nutritional supplements (Jothi et al., 2012). The live microorganisms are being used as probiotics in fish feed (Banerjee et al., 2000), of poultry (Patterson and Burkholder, 2003) and of other animals. The use of living microbial supplementation in diet as an additional ingredient for enhancing growth of the animal has been a thrust area for nutritionist in the recent past.
MATERIALS AND METHODS
After obtaining the approval of animal ethics committee of Goa University (registered under Committee for the Purpose of Control and Supervision of Experiments on Animals, Ministry of Social Justice and Empowerment, Govt. of India), the day old poultry chicks, Gallus domesticus (Broiler strain) were obtained from a local hatchery (Mandovi Hatcheries, Goa, India). The birds were acclimatized to laboratory conditions for 7 days. The birds were maintained till 35 days in the aviary room and utmost care was taken with respect to their cleanliness, food and water. The 7 day-old birds were divided into four groups. There were six cages (60x60x60 cm) for each group with two birds in each.
| Table 1: | Proximate composition of feeds used in the experiment |
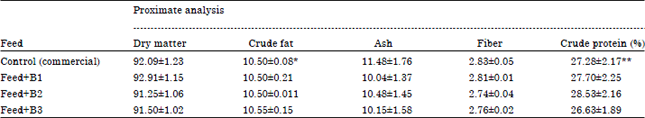 | |
| *Feed contain 5.0 g/100 g fat, **Feed contain 22.5 g/100 g protein. By adding sunflower oil and casein the lipid and protein content were increased. The control feed was made iso-lipidous and iso-proteinous with the experimental feed. B1: Staphylococcus sp., B2: Streptococcus sp. and B3: Pseudomonas sp. | |
| Table 2: | Relative composition of fatty acid profiles of feed supplemented with different lipid sources |
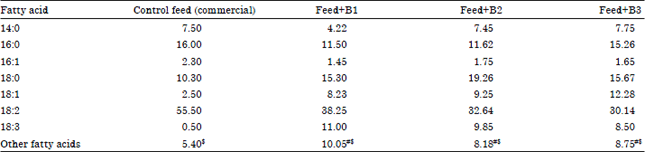 | |
| #Short chained fatty acids of C-10 and C-12 series, $Unidentified fatty acids of C-16 and C-14 series, B1: Staphylococcus sp., B2: Streptococcus sp. and B3: Pseudomonas sp. Values are means of six estimates | |
Group 1 served as control and was fed with the control diet ad libitum and the remaining three groups served as experimental (B1, B2 and B3) groups and were fed the feed supplemented with different strains (Staphylococcus sp. Streptococcus sp. Pseudomonas sp., respectively) of marine bacteria which were grown in Mineral Salt Medium (MSM) containing 5% sodium acetate. To avoid any pathogenic effect, the harvested bacterial cells were killed by heat treatment at 15 psi in an autoclave before blending with the commercial feed as an additive in such way to have 10% lipid in the feed. The control feed was made iso- proteinous and iso-lipidous (by adding exogenous casein and sunflower oil to the commercial feed) as compared to the experimental feeds. It was proved in the laboratory that 10% lipid in the feed was needed to have better growth of bird (Pujari and Roy, 2007). The proximate composition of the control as well as experimental feeds is represented in Table 1 and the fatty acid profiles of the feeds in Table 2. The birds were sacrificed after 30 days of feeding.
Growth of the birds, in terms of daily instantaneous growth rate (Gw) which was calculated from natural logarithm of weight gain per day and Feed Conversion Ratio (FCR), a ratio of the dry weight of feed intake and the daily weight gain were recorded (Hardy, 1989).
Tissue biochemical composition: Tissue protein and tissue lipid profiles viz., total triglycerides, total cholesterol and total phospholipid (Pujari and Roy, 2007) were recorded for liver, pectoral muscle and large intestine. The fatty acid profiles of liver and muscle, were analyzed with the help of Gas Chromatogram (Chemito make model 8610) equipped with flame ionizing detector and 10% DEG (Di Ethylene Glycol) packed column. The oven temperature was programmed (Roy et al., 1992). The flow of the carrier gas was fixed at 15 mL min-1. The identification of the obtained peeks was done with the help of prepared standard chromatogram of the known fatty acids under the same program.
Health profiles of the birds: Serum lipid profiles including total cholesterol, total triglycerides, HDL cholesterol, LDL-cholesterol, VLDL-cholesterol were also recorded using the diagnostic kits (M/s., Crest Biosystems, Goa, India). The activity of serum and hepatic alkaline phosphatase [EC 3.1.3.1], lactate dehydrogenase [EC 1.1.1.27], glutamate oxaloacetate transaminase [EC2.6.1.1], glutamate pyruvate transaminase [EC 2.6.1.2] (Godkar, 1994) were measured to find out the health status of liver and heart of the birds.
Cholesterol metabolism: The activity of two important regulatory enzymes viz. 3-hydroxy-3- methylglutaryl-CoA reductase [EC1.1.1.34] and 3-hydroxy-3-methylglutaryl-CoA synthase [E.C 2.3.3.10] of cholesterol metabolism were also monitored following the methods of Siedel (1983) and Miziorko (1985), respectively. The enzyme activities were measured with UV-visible spectrophotometer (Simadzu, model TCC-240A) capable of exact measurement at 339 nm with a thermostat cuvette holder. The reaction mixture for 3-hydroxy-3-methylglutaryl-CoA reductase contained phosphate buffer (0.1 mol L-1; pH 6.5), EDTA solution (0.1 mol L-1), Cysteamine solution (0.1 mol L-1), 1% serum albumin, NADPH solution (5 mmol L-1), 3-hydroxy-3 methyl CoA and serum or 1% liver homogenate. The same for 3-hydroxy-3-methylglutaryl-CoA synthase contained 0.2 M Tris-HCL (pH 8.2), 0.2 mM EDTA, 10 mM acetyl CoA (pH 4.5), 1 mM acetoacetyl CoA (pH 4.5) and serum or 1% liver homogenate.
All the recorded observations were expressed in the form of arithmetic mean of twelve birds and the standard error. Comparison test of the obtained data for each experimental group with control group was analyzed with student‘t’ test using Microsoft Office Excel sheet.
RESULTS
With the supplementation of different strains of bacteria for 30 days the average weight of the chicks was increased by about 10-45% (p<0.05-p<0.005) followed by an increase in the daily instantaneous growth rate (Gw) and decrease in the FCR value (Table 3). The maximum increase in the net weight gain was recorded for birds supplemented with Streptococcus strain with 5% increase in daily instantaneous growth rate and 25% decrease in FCR value.
An increase (about 28-68%, p<0.01) in the total protein concentration of the liver was noticed after 30 days of supplementing all the different strains of bacteria compared to the control group (Fig. 1). With the supplementation of Staphylococcus and Pseudomonas strains of bacteria no significant change in the total protein level of muscle and intestine was noticed. However, with the supplementation of Streptococcus strain of bacteria muscle protein and intestinal protein were increased by 18 and 9%, respectively (p<0.005).
A statistically significant increase (48%-3 fold, p<0.005) was recorded in the level of total triglycerides in liver, muscle and intestine after 30 days of feeding all three strains of bacteria compared to the control group.
| Table 3: | Growth chart of chick (Gallus domesticcus) supplemented with different bacteria along with the commercial feed for 30 days during post hatching development |
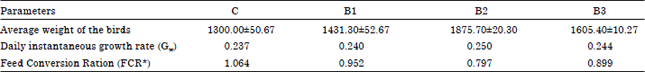 | |
| *FCR value was calculated cage wise and the average of the six cages were taken, C: Controls birds, B1: Staphylococcus sp., B2: Streptococcus sp. and B3: Pseudomonas sp. Data is Mean±SEM (n = 12) | |
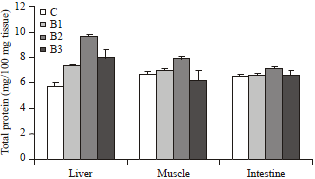 | |
| Fig. 1: | Changes in the total protein concentration in various tissues of broiler chicks, Gallus domesticus supplemented with different strains of bacteria along with commercial diet for 30 days during post hatching development. Values are Mean±SEM (n = 12) |
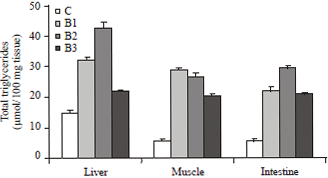 | |
| Fig. 2: | Changes in the total triglycerides concentration in various tissues of broiler chicks, Gallus domesticus supplemented with different strains of bacteria along with commercial diet for 30 days during post hatching development.Values are Mean±SEM (n = 12) |
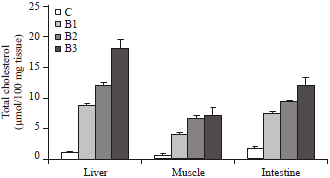 | |
| Fig. 3: | Changes in the total cholesterol concentration in various tissues of broiler chicks, Gallus domesticus supplemented with different strains of bacteria along with commercial diet for 30 days during post hatching development. Values are Mean±SEM (n = 12) |
The maximum elevation in the triglycerides content of liver and intestine was recorded in the chicks supplemented with Streptococcus bacterial strain but in muscle, maximum increase in triglycerides concentration was recorded with Staphylococcus strain (Fig. 2).
The 4 to 15-fold augmentation of total cholesterol concentration was noticed in the different tissues of Gallus domesticus due to supplementation of all three strains of bacteria for 30 days (p<0.005). The highest amount of cholesterol accumulation in the liver, muscle and intestine was noticed with the supplementation of Pseudomonas strain of bacteria (Fig. 3).
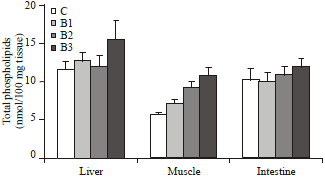 | |
| Fig. 4: | Changes in the total phospholipid concentration in various tissues of broiler chicks, Gallus domesticus supplemented with different strains of bacteria along with commercial diet for 30 days during post hatching development. Values are Mean±SEM (n = 12) |
A statistically significant increase (30%, p< 0.01) in the level of total phospholipid concentration of liver was observed with the supplementation of Pseudomonas strain of bacteria. Whereas, in muscles the total phospholipid concentration was increased (25% to 2-fold, p<0.005) with the supplementation of all three strains of bacteria for 30 days (Fig. 4). In the intestine, the phospholipid concentration remained unaltered due to the supplementation of bacteria for 30 days.
When the chicks were supplemented with different bacterial strains along with the commercial diet for 30 days, in general, the alpha linolenic acid concentration along with the eciosapentaenoic and docosahexaenoic acid was increased with the decrease in the concentrations of arachidonic and oleic acid and palmitic acid (Table 4). All these changes in the relative fatty acid composition of total lipid brought about a significant increase in ω3/ω6 ratios. The maximum decrease in the relative percentage composition of fatty acid profiles viz., oleic acid (in liver only), linoleic acid, arachidonic acid of total lipid in tissues of Gallus domesticus was observed with the supplementation of Streptococcus strain for 30 days as compared to the Staphylococcus and Pseudomonas strains. These changes are associated with the maximum increase of eciosapentaenoic acid, docosahexaenoic acid (in liver only) in the total lipid of birds.
The serum total cholesterol level was decreased marginally (p<0.005) with about 65% decrease in total triglycerides concentration (p<0.001), with about 10% increase in HDL cholesterol concentration (p<0.02) and about 70% decrease in VLDL concentration (p<0.001) with the supplementation of Streptococcus strain of bacteria along with feed for 30 days (Table 5). However, with the supplementation of Pseudomonas strain of bacteria, the serum TG was decreased by about 50% (p<0.005) with about 40% increase in LDL, 50% decrease in VLDL concentration (p<0.005).
Although three different strains of bacteria were selected for the present study, the activities of various enzymes were measured in the birds fed with Streptococcus strain only along with the control group of birds. With the supplementation of Streptococcus strain of bacteria, the alkaline phosphatase activity of liver was increased by 21% (p<0.005), however the activity of the same enzyme was decreased by 17% (p<0.005) in serum (Table 6). The activity of GPT and LDH in liver and serum remained unaltered. Whereas the GOT activity was decreased by about 23% (p<0.005) but remained unchanged in the serum.
About 2.5-fold augmentation (p<0.05) in serum HMG CoA reductase activity was recorded in the chicks supplemented with Streptococcus strain of bacteria for 30 days compared to the same of control birds. However, no change in liver HMG CoA reductase activity was detected due to dietary supplementation of bacteria.
| Table 4: | Fatty acid profiles (relative percent composition) of liver and muscle total lipid of chick (Gallus domesticus) supplemented with different bacteria along with the commercial diet for 30 days |
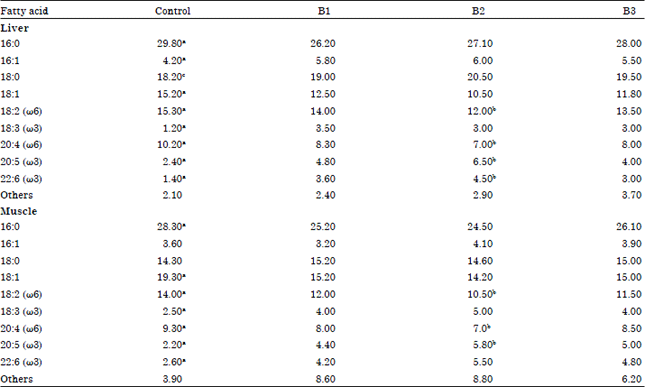 | |
| aSignificant change (p<0.05) over B1, B2 and B3, bSignificant change (p<0.05) over of B1 and B3, cSignificant change (p<0.05) over of B2, C: Control birds, B1: Staphylococcus sp., B2: Streptococcus sp. and B3: Pseudomonas sp. Mean values of six set of samples were taken | |
| Table 5: | Changes in the serum lipid profiles of the chick (Gallus domesticus) supplemented with different bacteria along with the commercial feed for 30 days of post hatching development |
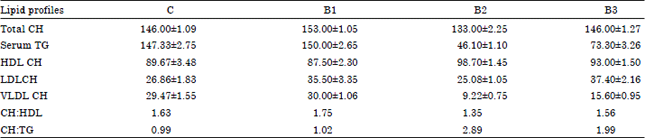 | |
| C: Controls, B1: Staphylococcus sp., B2: Streptococcus sp. and B3: Pseudomonas sp. Values are Mean±SEM (n = 12) | |
| Table 6: | The activity (μmol min-1 mg-1 protein) of liver function and cardiac function enzymes in Gallus domesticus supplemented with Streptococcus strain of bacteria for 30 days along with commercial diet |
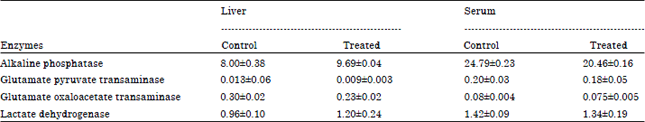 | |
| Values are Mean±SEM (n = 12) | |
| Table 7: | The activity (U min-1 mg-1 protein) of 3-HMG CoA reductase and 3-HMG CoA synthase in Gallus domesticus supplemented with Streptococcus strain of bacteria for 30 days along with the commercial diet |
 | |
| Values are Mean±SEM (n = 12) | |
2.3-folds increase in the liver HMG CoA synthase activity was detected with the supplementation of Streptococcus strain of bacteria after 30 days but no such change was detected in the serum due to the feeding experiment (Table 7).
DISCUSSION
It is well established that both linoleic acid and α-linolenic acid are essential fatty acids for entire animal kingdom and are to be consumed through diets. The natural distribution of these two essential fatty acids is not cosmopolitan. The availability of α-linolenic acid is very much restricted and more confined to the marine ecosystem rather than the terrestrial and freshwater ecosystem. This might be the reason for lower level of accumulation of omega-3 PUFAs in terrestrial and freshwater animals and higher level of ω3 PUFA in marine animals (Roy et al., 1999). It was observed in our laboratory that marine particulate materials contain about 10% α-linolenic acid in comparison to 5% in brackish water particulate materials and 0.5% in fresh water particulate materials (unpublished data). Three bacterial strains (Pseudomonas, Streptococcus and Staphylococcus) collected from coastal sediment samples contained about 15-20% α-linolenic acid when grown in sodium acetate medium (Pujari et al., 2004).
The use of live microbial supplementation in the diet as an additional ingredient for enhancing the growth of an animal has been a thrust area for nutritionists in the recent past (Doreau and Chilliard, 1997). Marine bacteria are known to produce wide range of compounds which have potential application as bioactive compounds, probiotics and nutritional supplements. These micro organisms are now been screened for the production of PUFA as well as specific fatty acids (Yazawa, 1996; Pujari et al., 2004). The bacterial strains identified and cultured in the laboratory might be the pathogenic strains and hence, the diet was supplemented with inactivated bacterial cells (rich in α-linolenic acid) instead of live microbial cells. Out of these 3 strains viz., Pseudomonas, Streptococcus and Staphylococcus, the Streptococcus strain seemed to be more effective strain that could be used as a source of alpha linolenic acid in a diet. The observed 45% increase in the net weight with supplementation of Streptococcus strain of bacteria in diet for a period of 30 days (Table 3) indicates that this strain of bacteria is a growth-promoting microorganism. The increased growth might be due to the increased proportion of alpha linolenic acid content in the experimental diet supplemented with bacteria (Table 2). These observations once again confirm the involvement of dietary fat in preventing dietary protein to undergo energy-yielding process and thus are in agreement with the findings of Mieczkowska et al. (2001).
The increased net weight gain of the bird with bacterial supplementation is reflected in the liver and muscle protein concentration (Fig. 1), along with triglycerides concentration (Fig. 2), cholesterol concentration (Fig. 3) and phospholipid concentration (Fig. 4) in all tissues, which, once again confirm the dietary role of α-linolenic acid in the growth and lipid metabolism of birds. Increased level of Eciosapentaenoic Acid (EPA) and Docosahexaenoic Acid (DHA) at the cost of oleic acid, linoleic acid and arachidonic acid (Table 4) confirmed the competition of α-linolenic acid with linoleic acid to bind with Δ5 and Δ6 desaturases enzyme system for the production of long chain PUFA. The similar observations were made on dietary supplementation of n3 fatty acid rich fish oil in chickens (Al-Athari and Watkins, 1988; Mieczkowska et al., 2001).
Decrease in the concentration of total cholesterol and triglycerides in the serum along with little increase in HDL cholesterol concentration without altering LDL or VLDL cholesterol due to dietary supplementation of Streptococcus strain of bacteria (as a source of α-linolenic acid) resulted in the reduction of CH: HDL ratio and increase in CH: TG ratio in Gallus domesticus (Table 5). These changes in serum lipid profiles indicate no health hazards to the birds with supplementation of bacteria in the diet. High content of alpha linolenic acid in the diet converted into ω-3 long chain PUFA (EPA and DHA) by Gallus domesticus mobilize cholesterol for the production of lipoprotein by the action of the LCAT enzymes. Little increase in liver alkaline phosphatase and decrease in serum alkaline phosphatase activity (Table 6) with decrease in liver GOT activity and insignificant changes in GPT and LDH activity in liver and serum once again confirm the well being state of bird due to dietary supplementation of Streptococcus strain of bacteria for 30 days. Little change in alkaline phosphatase activity in liver and serum and GOT activity in liver might be due to shifting of some metabolic pathways (which need to be confirmed in future) in Gallus domesticus due to supplementation of Streptococcus bacterial strain over a period of 30 days. Olurede and Longe (2001) reported the change in the serum GPT activity in chicks due to dietary supplementation of palm oil. It is reported that dietary fatty acids alter the inositol phosphate metabolism and protein kinase C activity to regulate intracellular signaling system (Olurede and Longe, 2001) and this might alter the functioning of desaturation system in the endoplasmic reticulum to convert linoleic acid and/or linolenic acid to their respective PUFA.
Increase in HMG CoA reductase activity in serum and HMG CoA synthase activity in liver (Table 7) due to Streptococcus bacterial strain supplementation for 30 days along with increase in tissue cholesterol and triglycerides concentration (Fig. 3 and 4) confirm the involvement of dietary accumulation of alpha linolenic acid towards the production of cholesterol. This also clearly indicates that no de novo biosynthesis of cholesterol takes place in the liver. A feedback regulation of hepatic CoA activity by the dietary fat was not due to altered mRNA levels of cellular nucleic acid binding protein which is essential to bind sterol regulatory element protein in Chinese hamster (Ness et al., 1991). The change in cholesterol metabolism by exogenous dietary fatty acids as observed in the present study might be due to induction of gene transcription encoding protein for the lipid oxidation or by suppressing the gene expression for encoding protein for lipid synthesis (Jump and Clarke, 1999) which are yet to be confirmed. The present results indicate that the marine bacteria, particularly the Streptococcus sp. could be supplemented as a source of an important essential fatty acid to keep the birds in good health.
ACKNOWLEDGEMENT
Department of Ocean Development, Govt. of India supported the study, vide research grant No. MRDF/11/4/14/Uni-97 through Ocean Science Technology Cell, Microbiology. Besides, support from UGC SAP programme under DRS1 [No. F.3-3/2011 (SAP-11)] is also acknowledged.
REFERENCES
- El Razak, A.A., A.C. Ward and J. Glassey, 2013. Screening of marine bacterial producers of polyunsaturated fatty acids and optimisation of production. Microb. Ecol., 67: 454-464.
CrossRef - Devi, N.K.A., R. Rajendran and S.K. Sundaram, 2011. Isolation and characterization of bioactive compounds from marine bacteria. Indian J. Nat. Prod. Resour., 2: 59-64.
Direct Link - Banerjee, S., S.A. Azad, S. Vikineswary, O.S. Selvaraj and T.K. Mukherjee, 2000. Phototrophic bacteria as fish feed supplement. Asian-Aust. J. Anim. Sci., 13: 991-994.
CrossRefDirect Link - Budowski, P. and M.A. Crawford, 1986. Effect of dietary linoleic and α-linolenic acids on the fatty acid composition of brain lipids in the young chick. Prog. Lipid Res., 25: 615-618.
CrossRef - Doreau, M. and Y. Chilliard, 1997. Digestion and metabolism of dietary fat in farm animals. Br. J. Nutr., 78: S15-S35.
CrossRefPubMedDirect Link - Jump, D.B. and S.D. Clarke, 1999. Regulation of gene expression by dietary fat. Ann. Rev. Nutr., 19: 63-90.
CrossRef - Mieczkowska, A., V.C. Nguyen and S. Smulikowska, 2001. Effect of dietary fat on fatty acid composition of lipids from breast muscle and abdominal fat of broiler chickens. J. Anim. Feed Sci., 10: 279-284.
Direct Link - Ness, G.C., R.K. Keller and L.C. Pendleton, 1991. Feedback regulation of hepatic 3-hydroxy-3-methylglutaryl-CoA reductase activity by dietary cholesterol is not due to altered mRNA levels. J. Biol. Chem., 266: 14854-14857.
Direct Link - Patterson, J.A. and K.M. Burkholder, 2003. Application of prebiotics and probiotics in poultry production. Poult. Sci., 82: 627-631.
CrossRefPubMedDirect Link - Perez-Bonilla, A., C. Jabbour, M. Frikha, S. Mirzaie, J. Garcia and G.G. Mateos, 2012. Effect of crude protein and fat content of diet on productive performance and egg quality traits of brown egg-laying hens with different initial body weight. Poult. Sci., 91: 1400-1405.
CrossRefDirect Link - Pujari, S., R. Roy and S. Bhosle, 2004. Screening of bacteria from sediments of coastal ecosystem, as potential sources of alpha linolenic acid. Indian J. Mar. Sci., 33: 243-247.
Direct Link - Roy, R., S. Singh and S. Pujari, 2008. Dietary role of omega-3 polyunsaturated fatty acid (PUFA): A study with growing chicks, Gallus domesticus. Int. J. Poult. Sci., 7: 360-367.
CrossRefDirect Link - Roy, R., E. Fodor, K. Kitajka and T. Farkas, 1999. Fatty acid composition of the ingested food only slightly affects physicochemical properties of liver total phospholipids and plasma membranes in cold-adapted freshwater fish. Fish Physiol. Biochem., 20: 1-11.
CrossRefDirect Link - Roy, R., D. Ghosh and A.B. Das, 1992. Homeoviscous adaptation of different membranes in the brain of an air-breathing indian teleost, Channa punctatus, during seasonal variation of environmental temperature. J. Therm. Biol., 17: 209-215.
CrossRefDirect Link - Sven, D., H. Ingird, J. Heinz, B. Wolfgang, A. Peggy, Z. Rainer and G. Sigmar, 2000. Effect of soya oil supplementation and protein leveling laying hen diets on praecaecal nutrient digestibility, performance, reproductive performance, fatty acid composition of yolk fat and on other egg quality parameters. Eur. J. Lipid Sci. Technol., 3: 218-232.
- Yazawa, K., 1996. Production of eicosapentaenoic acid from marine bacteria. Lipids, 31: S297-S300.
CrossRef








