Research Article
Histomorphological Studies on the Stomach of the Japanese Quail
Department of Histology, Faculty of Veterinary Medicine, South Valley University, Qena, Egypt
Gamal Kamel
Department of Anatomy and Histology, Faculty of Veterinary Medicine, Assiut University, Assiut, Egypt
Ahmed Abo El-Magd Ahmad
Department of Anatomy and Histology, Faculty of Veterinary Medicine, Assiut University, Assiut, Egypt









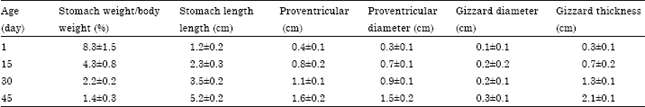
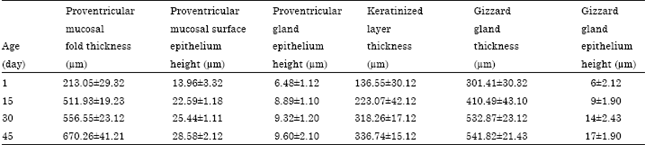
midhat abd elfatah elkasheif Reply
thanks u very much please i want link them to our best and over scentific services