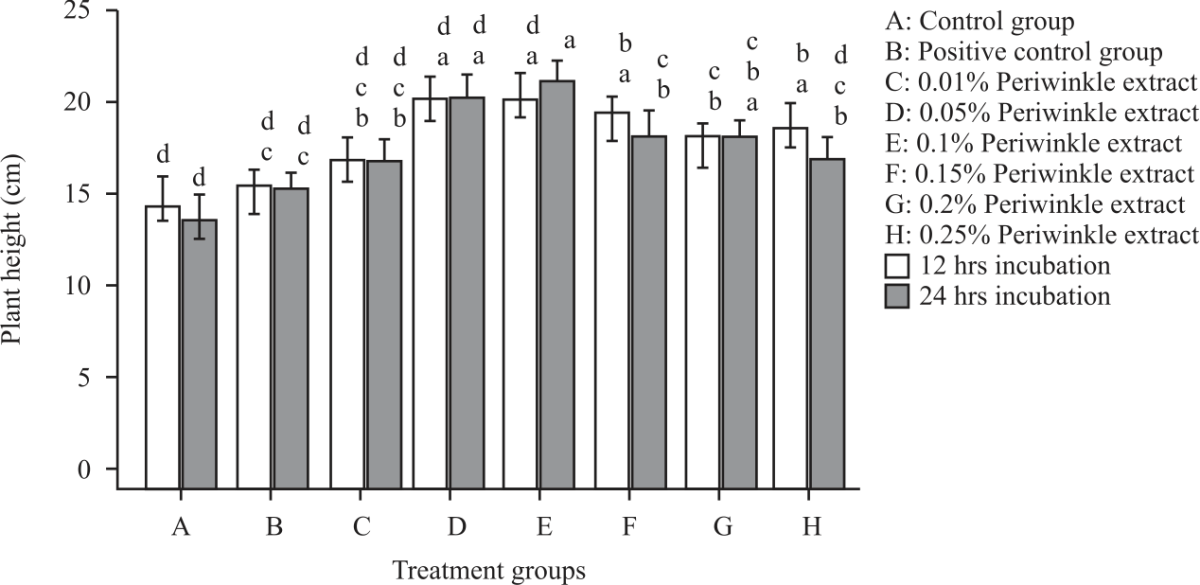
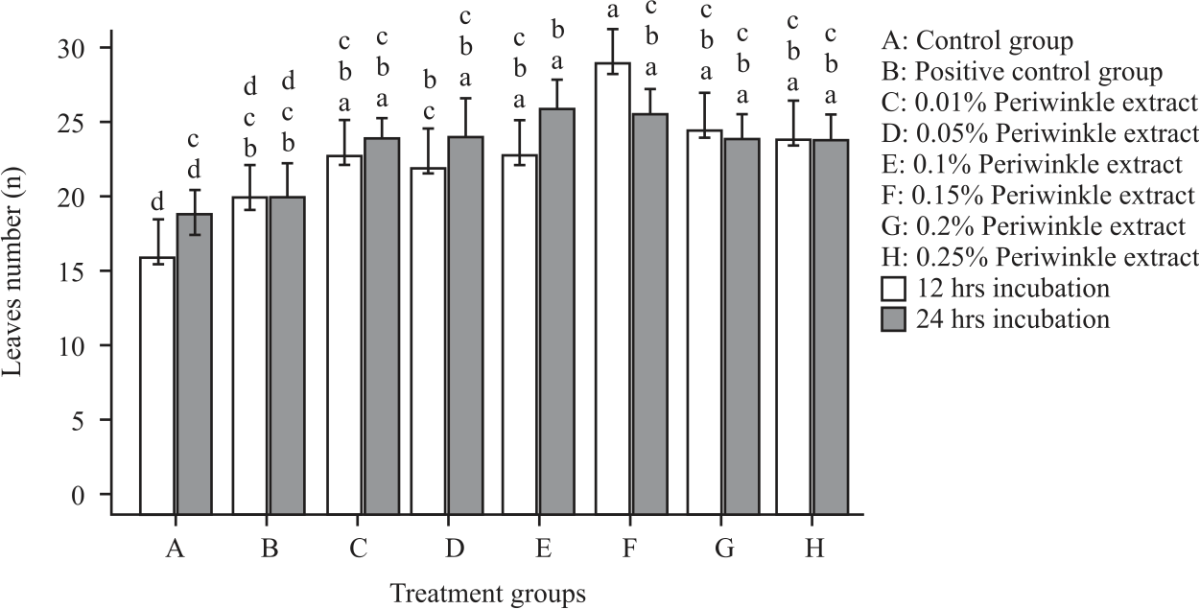
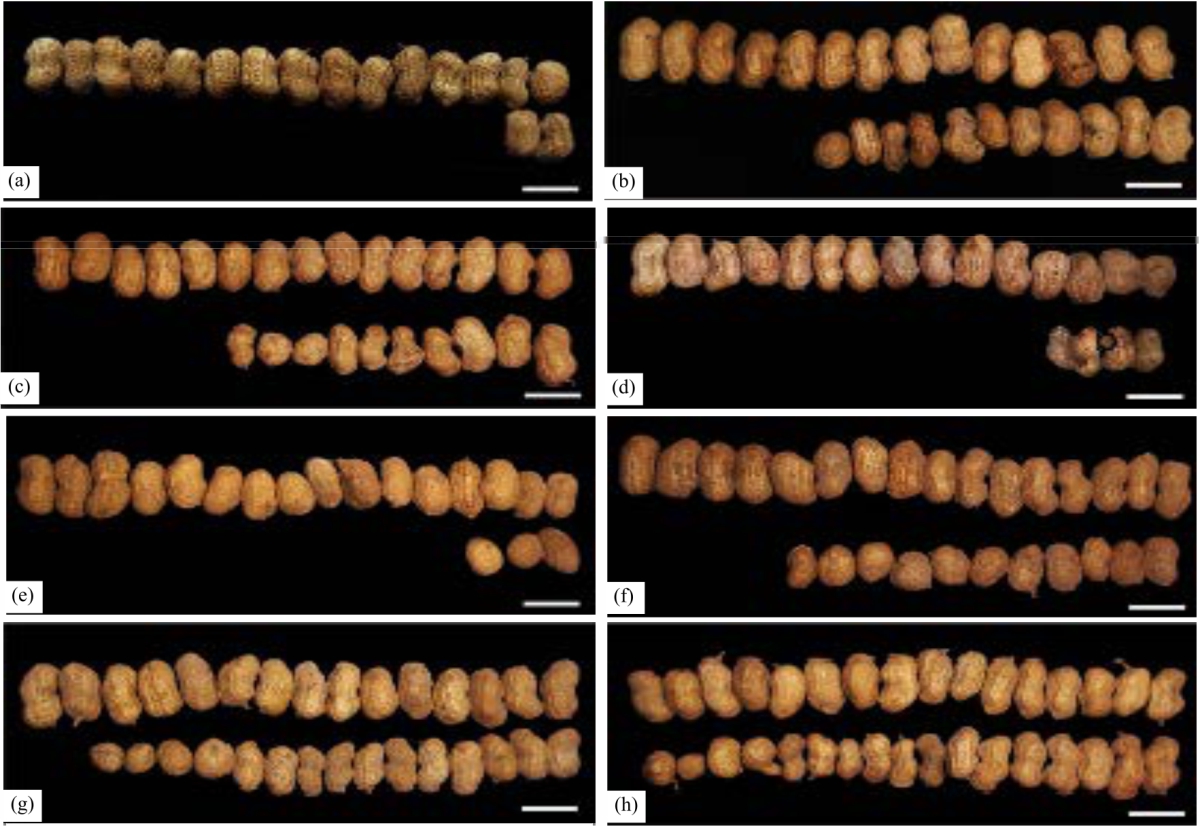
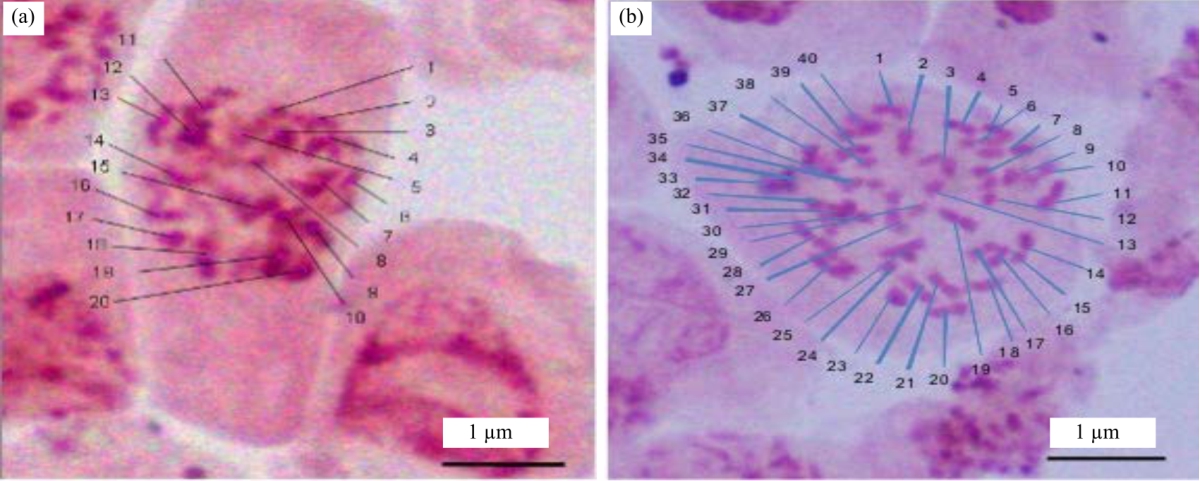
Research Article
Induced Polyploidy in Arachis hypogaea L. var. Talam using Catharanthus roseus Phenolic Extract
Laboratory of Genetics and Breeding, Faculty of Biology, Universitas Gadjah Mada, Jl. Teknika Selatan, Sekip Utara, 55281, Yogyakarta, Indonesia
Ayudha Bahana Ilham Perdamaian
Laboratory of Genetics and Breeding, Faculty of Biology, Universitas Gadjah Mada, Jl. Teknika Selatan, Sekip Utara, 55281, Yogyakarta, Indonesia
LiveDNA: 62.19319
Dian Sartika
Laboratory of Genetics and Breeding, Faculty of Biology, Universitas Gadjah Mada, Jl. Teknika Selatan, Sekip Utara, 55281, Yogyakarta, Indonesia
Budi Setiadi Daryono
Laboratory of Genetics and Breeding, Faculty of Biology, Universitas Gadjah Mada, Jl. Teknika Selatan, Sekip Utara, 55281, Yogyakarta, Indonesia
LiveDNA: 62.25765