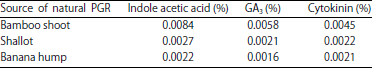
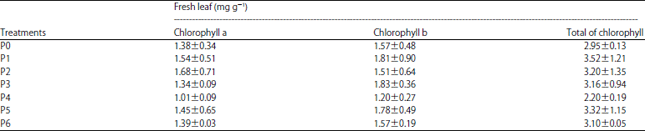
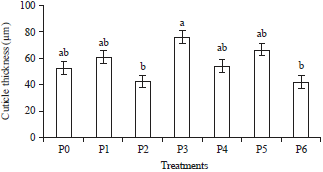
Research Article
Physiological Characteristics of Binahong (Anredera cordifolia (Ten.) Steenis) on Application of Natural Plant Growth Regulator
Faculty of Agriculture, Universitas Sumatera Utara, Jl. Prof. A. Sofyan No. 3, Kampus USU, Padang Bulan, Medan, 20155, Indonesia
LiveDNA: 62.20813
Lisa Mawarni
Faculty of Agriculture, Universitas Sumatera Utara, Jl. Prof. A. Sofyan No. 3, Kampus USU, Padang Bulan, Medan, 20155, Indonesia
Herla Rusmarilin
Faculty of Agriculture, Universitas Sumatera Utara, Jl. Prof. A. Sofyan No. 3, Kampus USU, Padang Bulan, Medan, 20155, Indonesia