Research Article
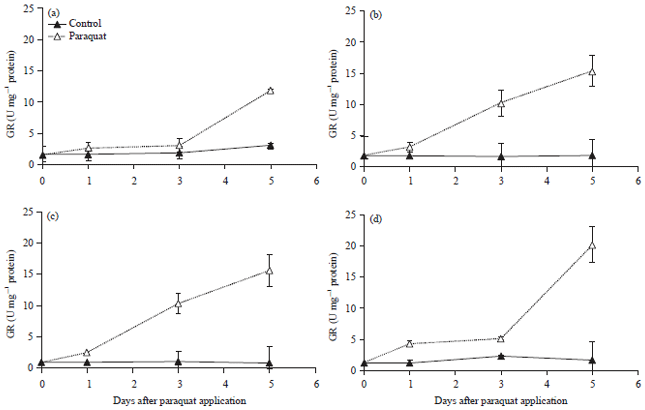
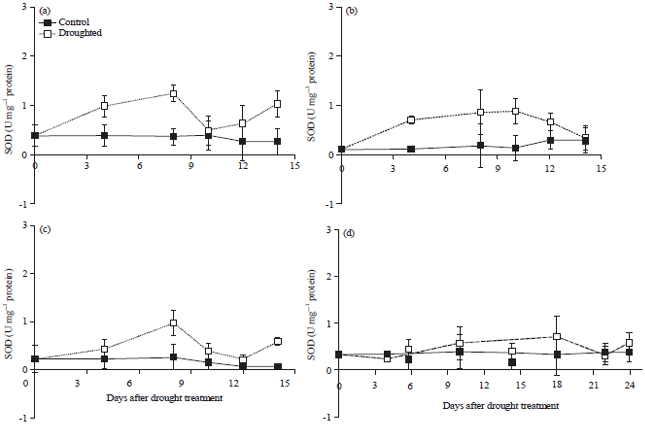
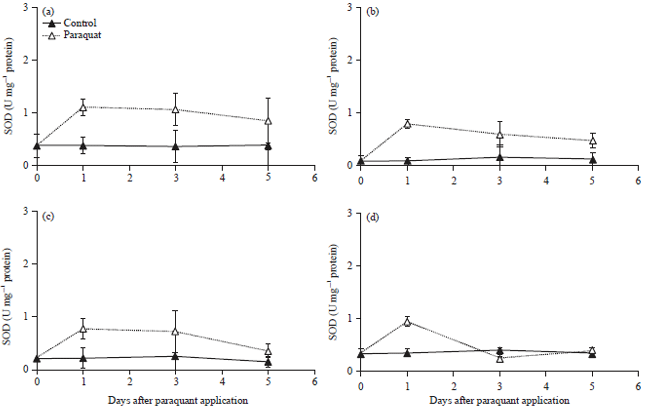
Oxidative Stress and Photosynthesis Reduction of Cultivated (Glycine max L.) and Wild Soybean (G. tomentella L.) Exposed to Drought and Paraquat
Department of Biology, Faculty of Mathematics and Natural Sciences, Bogor Agricultural University, Bogor, Indonesia
LiveDNA: 62.15393
Violita Violita
Department of Biology, Faculty of Mathematics and Natural Sciences, State University of Padang (UNP), Padang, Indonesia
Triadiati Triadiati
Department of Biology, Faculty of Mathematics and Natural Sciences, Bogor Agricultural University, Bogor, Indonesia
Miftahudin Miftahudin
Department of Biology, Faculty of Mathematics and Natural Sciences, Bogor Agricultural University, Bogor, Indonesia