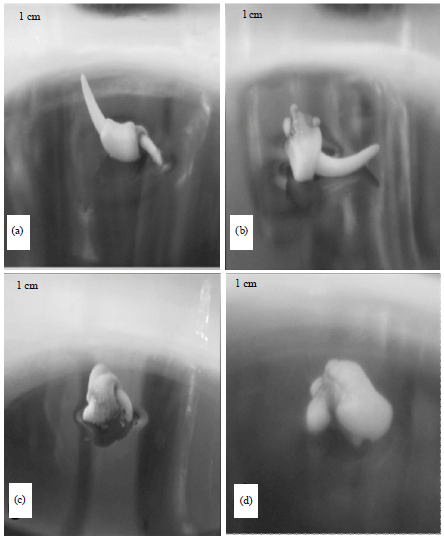
Research Article
Utilization of Sago Palm (Metroxylon sagu Rottb.) Zygotic Embryos for Cryopreservation
Department of Biological Sciences and Environmental Studies, College of Science and Mathematics, University of the Philippines Mindanao, Mintal, Davao City, Philippines
O.P. Damasco
Laboratory of National Plant Genetic Resources, Institute of Plant Breeding, University of the Philippines Los Ba�os, Laguna, Philippines
A.U. Novero
Department of Biological Sciences and Environmental Studies, College of Science and Mathematics, University of the Philippines Mindanao, Mintal, Davao City, Philippines