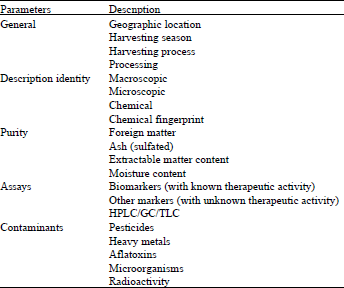
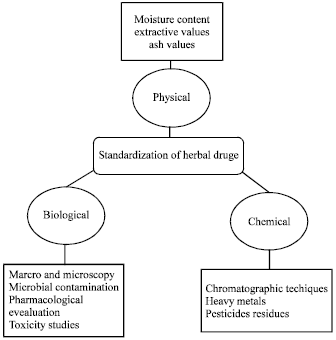
Review Article
Relevance of chemical Standardization and Innocuousness in the Process of Development of Herbal Medicines: A Review
Laboratorio de Fisiologia, Bioquimica y Biologia Molecular de Plantas,Programa de Doctorado en Biologia Experimental, UAM, Iztapalapa
Fernando Rivera-Cabrera
Laboratorio de Fisiolog�a, Biolog�a Molecular de Plantas,Depto. Ciencias de la Salud DCBS, UAM, Iztapalapa
Laura J. Perez-Flores
Laboratorio de Fisiolog�a, Biolog�a Molecular de Plantas,Depto. Ciencias de la Salud DCBS, UAM, Iztapalapa
Fernando Diaz de Leon Sanchez
Laboratorio de Fisiolog�a, Biolog�a Molecular de Plantas,Depto. Ciencias de la Salud DCBS, UAM, Iztapalapa
Jose Alberto Mendoza-Espinoza
Catedra Divisional Amelia Samano Bishop, Universidad Aut�noma Metropolitana Iztapalapa,Colegio de Ciencias y Humanidades, Plantel Casa Libertad,0000Universidad Aut�noma de la Ciudad de Mexico, Mexico