Research Article
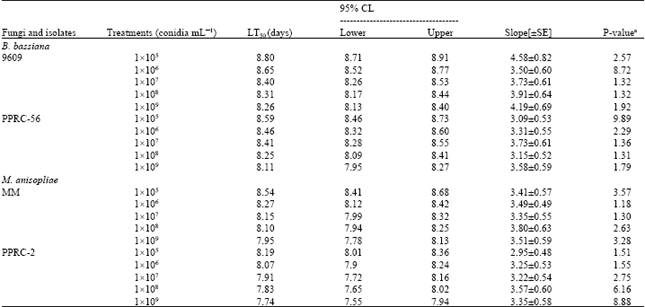
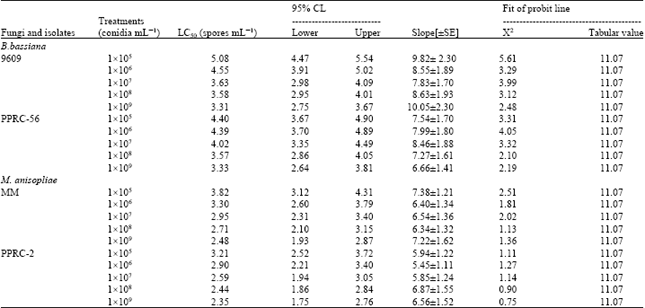
Laboratory Evaluation of Entomopathogenic Fungi Metarhizium anisophilae and Beauveria bassiana Against Termite, Macrotermes (Isoptera: Termitidae)
Jimma University College of Agriculture and Veterinary Medicine, P.O. Box 307, Jimma, Ethiopia
Waktole Sori
Jimma University College of Agriculture and Veterinary Medicine, P.O. Box 307, Jimma, Ethiopia
Mohamed Dawd
Ethiopian Institute of Agricultural Research, Ambo Plant Protection Research Center, P.O.Box 2003, Addis Ababa, Ethiopia