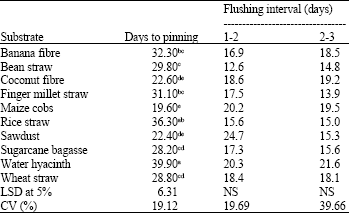
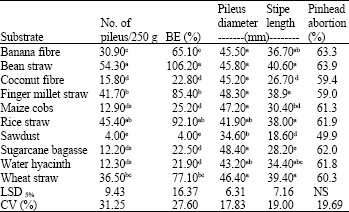
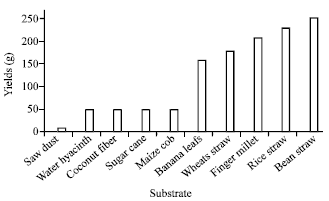
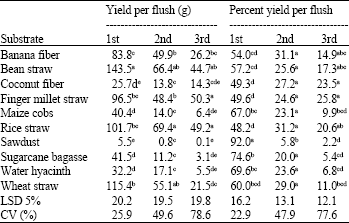
Research Article
Suitability of Locally Available Substrates for Oyster Mushroom (Pleurotus ostreatus) Cultivation in Kenya
University of Nairobi, P.O. Box 30197 00100, Nairobi, Kenya
G.O.M. Odero
University of Nairobi, P.O. Box 30197 00100, Nairobi, Kenya
E.W. Mutitu
University of Nairobi, P.O. Box 30197 00100, Nairobi, Kenya
P.M. Wachira
University of Nairobi, P.O. Box 30197 00100, Nairobi, Kenya
R.D. Narla
University of Nairobi, P.O. Box 30197 00100, Nairobi, Kenya
W.M. Muiru
University of Nairobi, P.O. Box 30197 00100, Nairobi, Kenya