Research Article
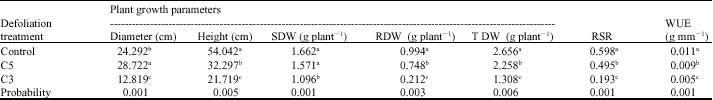
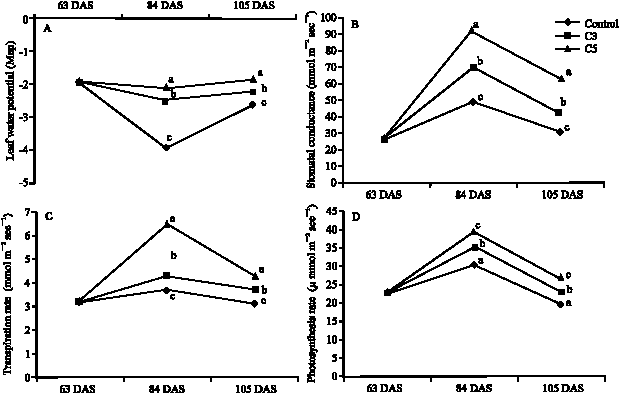
Clipping Effects on the Growth Variation, Water Use Efficiency and Photosynthetic Activity in Buffel Grass (Cenchrus ciliaris L.) Poaceae
Institut des Regions Arides, 4119 Medenine, Tunisia
M. Zaman Allah
SUPAGRO-INRA, Centre International d`Etudes Superieures en Sciences Agronomiques, F 34060 Montpellier Cedex 1, France
A. Ferchichi
Institut des Regions Arides, 4119 Medenine, Tunisia
E. Ferjani
Physiologie et Genetique des Plantes a Intret Agronomique, Faculte des Sciences de Bizerte, 7021 Jarzouna, Tunisia