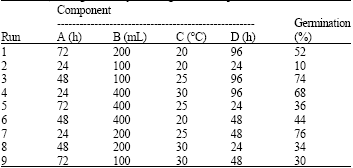
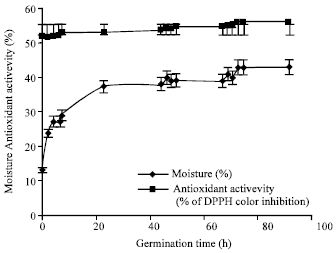
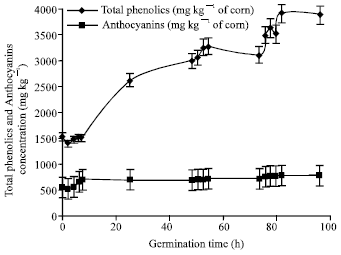
Research Article
Optimization of Germination Conditions of Blue Corn (Zea mays L.) by Taguchi Orthogonal Array Methodology
Departamento de Graduados e Investigacion en Alimentos, Escuela Nacional de Ciencias Biologicas, Instituto Politecnico Nacional, Carpio y Plan de Ayala, CP 11340 Mexico, DF, Mexico
A. Jimenez-Aparicio
Departamento de Biotecnologia, Centro de Desarrollo de Productos Bioticos, Instituto Politecnico Nacional, Yautepec, Morelos, Mexico
H. Hernandez-Sanchez
Departamento de Graduados e Investigacion en Alimentos, Escuela Nacional de Ciencias Biologicas, Instituto Politecnico Nacional, Carpio y Plan de Ayala, CP 11340 Mexico, DF, Mexico