Research Article
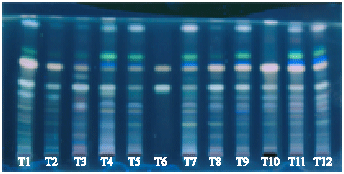
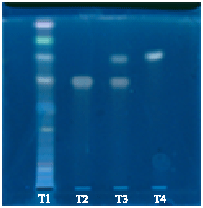
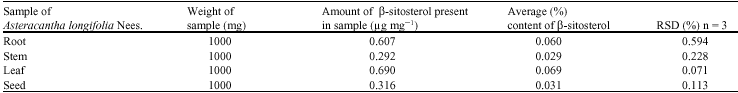
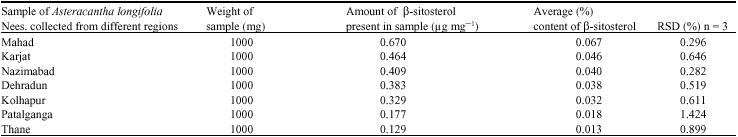
A Comparative Evaluation of Phytochemical Fingerprints of Asteracantha longifolia Nees. Using HPTLC
Laboratory of Herbal Research, Ramnarain Ruia College, Matunga (East), Mumbai-400019, India
S. Abhishek
Laboratory of Herbal Research, Ramnarain Ruia College, Matunga (East), Mumbai-400019, India