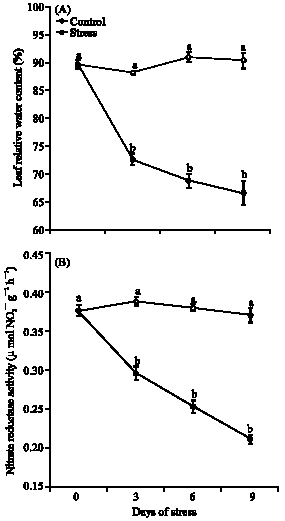
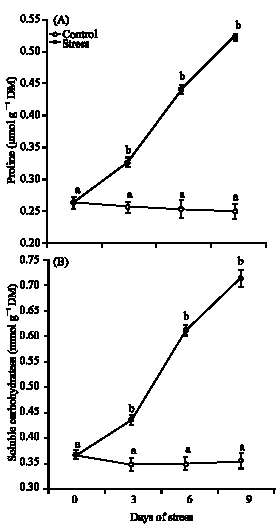
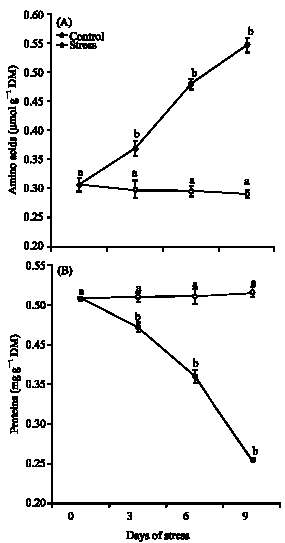
Research Article
Biochemical and Physiological Behavior of Vigna unguiculata (L.) Walp. Under Water Stress During the Vegetative Phase
Instituto de Ciências Agrárias, Universidade Federal Rural da Amazônia, Belém, Brazil
C.F. Oliveira Neto
Instituto de Ciências Agrárias, Universidade Federal Rural da Amazônia, Belém, Brazil
R.C.L. Costa
Instituto de Ciências Agrárias, Universidade Federal Rural da Amazônia, Belém, Brazil
B.G. Santos Filho
Instituto de Ciências Agrárias, Universidade Federal Rural da Amazônia, Belém, Brazil
F.J.R. Cruz
Instituto de Ciências Agrárias, Universidade Federal Rural da Amazônia, Belém, Brazil
H.D. Laughinghouse IV
Department of Biology, John Carroll University, University Heights, OH, USA