Research Article
A High Internal Phosphorus Use Efficiency in Tea (Camellia sinensis L.) Plants
Department of Plant Science, University of Tabriz, 51666 Tabriz, Iran
R. Hajiboland
Department of Plant Science, University of Tabriz, 51666 Tabriz, Iran
The productivity of crop plants is often reduced by a variety of environmental stresses particularly by nutrient deficiency. Phosphorous (P) is an essential macronutrient for plants and P deficiency is the most or even the prime limiting factor for plant growth and development. It was estimated that crops productivity is limited by P deficiency on 30-40% of the world arable lands (Vance et al., 2003; Kochian et al., 2004; Lambers et al., 2006). This problem is of particular concern for acid soils (Kochian et al., 2004). Phosphorous availability in acid soils is low due to binding to soil mineral surfaces and fixation into organic forms (Kochian et al., 2004), that results in a low rate of P diffusion towards the root surface. Application of P-containing fertilizers is usually the recommended treatment for enhancing soil P availability (Vance et al., 2003). However, only 20% or less of added P can be used by plants due to fixation of 80-90% of applied P by soil particles (Lambers et al., 2006). The cost, limited availability of P fertilizers and their limited resources as well as the effects of fertilizers on quality of environment can be problematic in extensive agriculture (Vance et al., 2003; Smith, 2002; Lambers et al., 2006). Therefore attention must be turned to use soil phosphate more efficiently by plants and application of management systems. Accordingly, we need enough information on the P uptake and utilization by plants.
There is genetically based inter and intra-specific variation in the ability of plants to tolerate P deficiency stress (Shenoy and Kalagudi, 2005). Tolerant species/genotypes developed strategies for a high P acquisition and utilization such as alteration of root morphology and architecture, induction of changes in the rhizosphere by root exudates, lower cellular P requirement, efficient remobilization of P, increased production and secretion of phosphatases and association with vesicular-arbuscular mycorrhiza (Alves et al., 2001; Vance et al., 2003; Kochian et al., 2004).
Tea (Camellia sinensis L.) is a popular beverage in most countries and its consumption is increasing because of its positive effects on human health (Zoysa et al., 1999; Luczaj and Skrzydlewska, 2005). Tea is cultivated in humid and sub-humid tropical regions of the world mainly on acid soils having pH of 4.5-5.5 (Zoysa et al., 1999). In spite of a large amount of works on tea plants particularly its phytochemistry (Luczaj and Skrzydlewska, 2005; Dalluge and Nelson, 2000), research works on mineral nutrition of this plants are rare.
In Iran, tea is cultivated on acid soils (pH 3-5) in the north of the country. Annually, 300 kg ha-1 P fertilizers are being used in tea plantations (Malakoti and Sadagat, 1999) that enhance production coast and environmental risks. This work therefore was undertaken to investigate the P deficiency tolerance in tea plants and some involving physiological mechanisms in its response to low P supply. In addition of growth and P uptake, gas exchange and chlorophyll fluorescence parameters were studied using five seed stocks of tea plants in hydroponic medium.
Plants culture and treatment: Five different seed stocks from distinct tea bushes were used in this study. Seeds were collected in October 2005 from the garden of Tea Research Station in Fuman, (Guilan, Iran) and after surface sterilization with 1% active hypochlorite were germinated on vermiculite in dark and moistened by distilled water every day. Twenty-days old seedlings were transferred on washed sand and grown under fluorescent light at about 400 μM m-2 sec-1, with 18/6 h light/dark photoperiod, 25/18C day/night temperature and relative humidity of 75/85%. Fifty-days old seedling with 3 fully expanded leaves were transferred to 1.2 L plastic pots containing aerated nutrient solution with the following composition (Ghanati et al., 2005; slightly modified): (NH4)2SO4 0.713, NH4NO3 0.73, K2SO4 0.46, CaCl2 0.5, MgSO4 0.41 (mM) and Fe-EDTA 32, H3BO3 46, CuSO4 2, MnSO4 90, Na2MoO4 2.6, ZnSO4 9.1 (μM).
Phosphorus was added as KH2PO4 at three concentrations including 0, 50 and 100 μM and double distilled water was used for preparation of nutrient solutions without P. pH was adjusted to 4.2, nutrient solutions were changed every week and plants were allowed to grow for 90 days.
Measurement of gas exchange and chlorophyll fluorescence parameters: Hundred forty days old tea plants treated for 3 months at different P levels, were used for measurements. Gas exchange parameters were measured by a portable photosynthesis system (LCA4, ADC, UK), in the third attached fully expanded young leaf under a Photosynthetic Photon Flux Density (PPFD) of 450 μmol m-2 sec-1. During gas exchange measurements, the temperature in the leaf chamber was 25±1C and CO2 concentration varied between 350 and 400 μmol mol-1. All measurements were conducted in the same day on four plants per each treatment. Values for net CO2 assimilation rate (A), stomatal conductance (gs) and transpiration rate (E) were calculated by the instrument and instantaneous Water Use Efficiency (WUE) was calculated as the ratio between net photosynthesis (A) and transpiration (E) (Jiang et al., 2006).
Chlorophyll fluorescence parameters were recorded in parallel for gas exchange measurements in the same leaf using a portable fluorometer (Fim 1500, ADC, UK). Prior to measurements, the attached leaves were dark adapted for 10 min using leaf-clips. The processing software generates values for initial (F0), maximum (Fm) and variable (Fv = Fm–F0) fluorescence. The maximum quantum efficiency of PSII was calculated as Fv/Fm.
Relative chlorophyll content was measured using a chlorophyll meter (Minolta, SPAD 502) on third leaves.
Plant harvest: Hundred forty days after sowing, tea plants were harvested. Before separating shoot and root, root axes were gently separated from each other in a water-filled container, then root tips number was determined by direct counting using a haemocytometer and the tap root length of plants was measured using a ruler. After washing with distilled water and blotting dry, separated shoot and root samples were oven-dried at 70C for 48 h and used for determination of P after measurement of dry weight. Root length was measured using another group of plants according to Tennant (1975).
For determination of P, 5 mL of 0.5 N Mg (NO3)2 and 10 mL double distilled water was added on oven-dried samples. Samples were air dried at room temperature after evaporating the mixture on a steam bath. Finally, samples were ashed in a muffle furnace at 500C for 30 min (Jaiswal, 2004). Samples were treated by 1:3 HNO3 and further digested on a heating plate. After resuspending in 2 mL 10% HCl, samples were made up to volume by double distilled water. Phosphorus concentration was determined by ammonium-vanadate or blue-paramolybdate methods depending on P concentration in samples (Page, 1982).
The internal P utilization efficiency was calculated as the amount of dry matter produced per unit of P absorbed by the plants (shoot DW/shoot P content).
For a comparison between tea and an extremely susceptible species for P deficiency, bean (Phaseulus vulgaris) plants (Santos et al., 2006) were cultivated under similar conditions. Seeds were germinated on moistened sand after surface sterilization, then 7 days old seedlings were transferred to 2.3 L plastic pot containing aerated nutrient solution (Neumann and Römheld, 1999) and treated with two levels of P including 0 and 250 μM. pH was adjusted to 6.2 and nutrient solutions were changed every 5 day. Plants were allowed to grow for 21 days and then used for P determination after measurement of dry weight.
All experiments were conducted using 4 independent replications. Statistical analysis were carried out using Sigma stat (3.02) and Tukey test at p≤0.05.
Plant growth: Plants growth was affected significantly by P deficiency (p≤0.05), however, no visual leaf symptoms were developed in plants until harvest. Considering both shoot and root growth of all 5 seed stocks, P concentration of 50 μM could be introduced as optimum P supply level for tea plants (Table 1). All shoot yield components including shoot dry weight, shoot height, number of leaves and leaf area were greater at 50 μM and further decreased at 100 μM P (Table 1). Root dry weight and root deep growth were diminished continuously with increasing P supply, but root length and number of root tips, similar with shoot growth, responded differently and were decreased at 100 μM P (Table 1). The ratio of root/shoot increased under low P supply (data not shown), that was the result of reduction in shoot dry weight (mainly leaves) concomitant with increase in root biomass.
Shoot and root concentration of P: Shoot and root concentration of P increased with increasing P supply level, however, difference in shoot P concentration between pants supplied with 50 and 100 μM P was mainly non-significant (p≤0.05) (Table 2). A considerable P taken up by plants was transported into shoots. Relative amounts of P transported into shoots were as high as 80% and did not change by different P supply. P uptake and transport were decreased at the highest P supply level in comparison with 50 μM P (Fig. 1), which was the result of the reduction of P content in shoot (data of P content were not shown).
Comparison of P deficiency tolerance in tea and bean plants: In contrast to tea, clear visual leaf deficiency symptoms were observed in bean plants, including stunted growth, overall leaf chlorosis and leaf necrosis of old leaves. Reduction of weight due to P deficiency during 90 and 21 days treatment was 57 and 66% for tea
| Table 1: | Effect of different levels of P supply (0, 50 and 100 μM) on growth of five tea seed stocks |
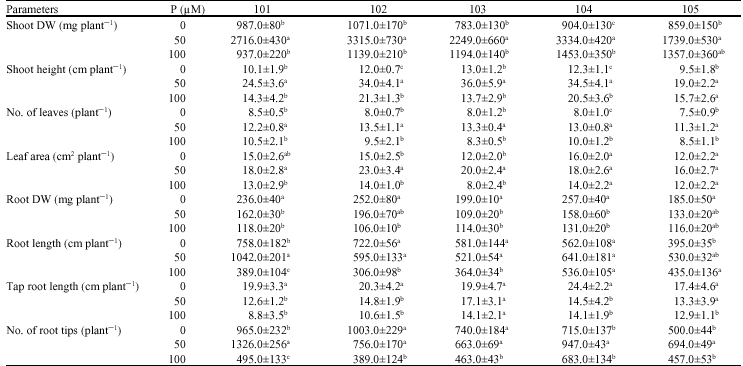 | |
Values are means±SD, Difference among values of each measured parameter from each seed stock followed by different letter(s) are significant (p≤0.05) | |
| Table 2: | P concentration (mg g-1 DW) of shoot and root and internal P use efficiency (shoot DW/shoot P content) of five different tea seed stocks grown in nutrient solution with three P levels (0, 50 and 100 μM) |
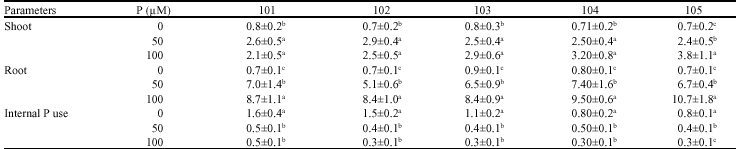 | |
| Values are means±SD, Difference among values of each measured parameter from each seed stock followed by different letter(s) are significant (p≤0.05) | |
and bean plants respectively (Fig. 2). For comparison of uptake and transport potential of P between tea and bean plants, the uptake rate (per week) was calculated (Fig. 2). Reduction of P uptake rate in tea due to low P supply was higher (94%) than bean (72%) plants. The same trend was
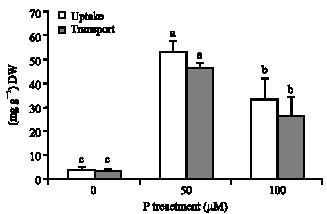 | |
| Fig. 1: | Effect of different concentrations of P (0, 50 and 100 μM) on P uptake and transport in tea plants. Difference among values of each measured parameter followed by different letter(s) are significant (p≤0.05) |
observed for P transport. The absolute amount of P uptake rate in deficient bean plants was significantly higher than tea (p≤0.05). In contrast to uptake and transport capacity, the internal P use efficiency of deficient plants was much greater in tea than bean plants (Fig. 2). P deficiency caused an increase up to 287 and in P use efficiency in tea (Table 2, Fig. 2), such an increase in bean plants was only 35% (Fig. 2).
Chlorophyll fluorescence and gas exchange parameters: Relative chlorophyll content did not change significantly under low P conditions (p≤0.05) (Fig. 3). However, a reduction of relative chlorophyll content, significantly or in tendency was observed in plants supplied with 100 μM P (p≤0.05). In leaves of tea plants subjected to P deficiency, minimal fluorescence (F0) and Tm values increased significantly in P deficient plants (p≤0.05). The values of maximal (Fm) and variable (Fv) fluorescence, in contrast, were decreased significantly in P deficient plants as well as plants supplied with supra-optimal P concentration (p≤0.05) (data not shown). The highest maximum quantum yield of PSII (Fv/Fm ratio) was recorded in plants supplied with 50 μM P. Although, data of Fv/F0
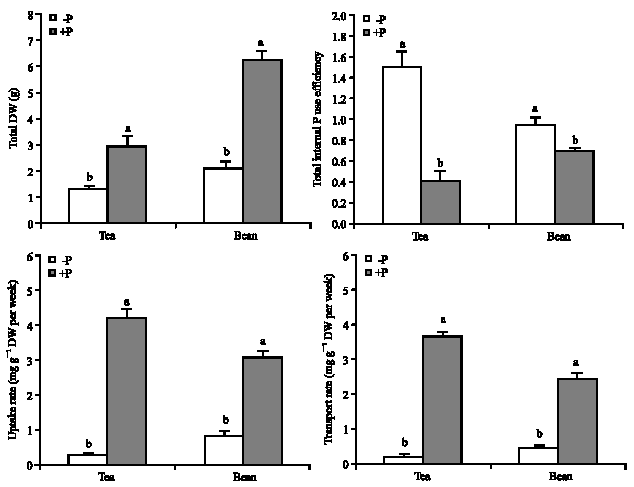 | |
| Fig. 2: | Effect of low and adequate P treatments on total dry weight, internal P use efficiency and P uptake and transport rate per week in bean and tea plants. Difference among values of each measured parameter from each plant followed by different letter(s) are significant (p≤0.05) |
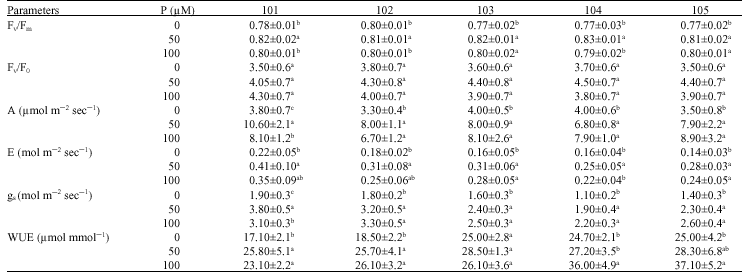
| Table 3: | Effect of different concentrations of P (0, 50 and 100 μM) on some fluorescence and gas exchange parameters (net assimilation rate (A), transpiration rate (E), stomatal conductance (gs) and instantaneous Water Use Efficiency (WUE)) in five different tea seed stocks |
 | |
| Difference among values of each measured parameter from each seed stocks followed by different letter(s) are significant (p≤0.05) | |
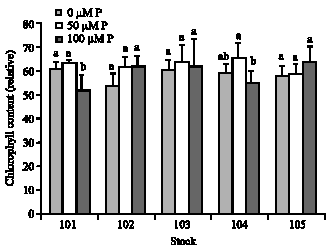 | |
| Fig. 3: | Effect of different concentrations of P (0, 50 and 100 μM) on relative chlorophyll content of five tea seed stocks. Difference among values of each measured parameter from each seed stocks followed by different letter(s) are significant (p≤0.05) |
ratio were changed only in tendency by P supply levels, the highest Fv/F0 values were recorded at optimum P supply (50 μM) (Table 3).
Phosphorous deficiency caused a significant reduction of net photosynthesis (A) and transpiration rate (E) in all tested seed stocks (p≤0.05). This reduction was also observed in some cases in plants supplied with 100 μM P (Table 3). Reduction of net photosynthesis rate was 41-64% and for transpiration rate was 50-64% (Table 3). Phosphorus deficiency affected negatively stomatal conductance (gs) of plants, the lowest stomatal conductance was observed in plants grown in nutrient solution without P (Table 3). The lowest intracellular CO2 concentration (Ci) was observed in P deficient plant and the highest values were recorded in plants supplied with 50 μM P (data not shown). Instantaneous Water Use Efficiency (WUE) was also decreased significantly in plants grown in nutrient solution without P addition and the highest values were observed in the presence of adequate P (p≤0.05) (Table 3).
In contrast to bean, tea plants did not show any visual leaf symptoms during relatively long period of growth (90 days) in nutrient solution with nil P concentration (double distilled water).
Growth and chlorophyll data suggested that, tea plant is an extremely tolerant species to P deficiency. Nevertheless, the role of seed P reserves should not be ruled out. Plants in this work were obtained from seeds, therefore, seed P reserves could have a significant contribution in providing P for young seedlings. This was well documented in some plants with big size seeds like maize (Alves et al., 2001). The quantitative contribution of seeds in meeting P requirement of young tea seedlings in comparison with rooting medium should be further studied.
According to present results, the optimum P supply in hydroponic grown tea plants is 50 μM, far below the recommended concentration in the literature (Ghanati et al., 2005). Plants grown at 100 μM P, showed significant growth inhibition 21-65% depending on seed stocks (p≤0.05). Therefore, 100 μM P, could be considered as toxic concentration for tea plants in hydroponic culture. Tissue P concentration of 1% and higher was considered as toxic for many plant species (Marschner, 1995). In this study, this critical concentration was observed only in roots of some tested seed stocks. The phosphorus concentration of leaves was 3-4 orders of magnitude lower than threshold concentration for P toxicity reported for other species.
Phosphorus concentration of leaves in severely deficient plants was lower in tea (0.66-0.80 mg g-1 DW) than bean (1.11 mg g-1 DW) plants, though a great difference in growth response of these two species. Growth reduction due to P deficiency was 44-67% in tea depending on seed stock and 66% in bean plants. Considering different growth period of these 2 species in this work, difference in growth response of tea and bean plants could be much greater than above mentioned data. Tea plants did not show any difference in growth compared with control plants when grown in nutrient solution without P for 45 days.
Lower growth reduction in combination with lower P concentration demonstrated high internal use efficiency in tea. Phosphorus use efficiency in deficient tea plants (1.45) was 1.6 times higher than bean (0.92). Interestingly, P use efficiency in tea plants increased up to 3.65 times in response to low P supply, the corresponding amount for bean was only 1.35.
Although root/shoot ratio increased significantly in response to low P (p≤0.05), morphological characteristics important for P acquisition in soil grown plants did not improve in P deficient tea plants. Root length and number of root tips as indicator of degree of root branching were rather diminished by low P treatment. It means that, though allocation of higher biomass to roots, P deficiency did not result in higher P absorption surface and higher soil exploration capacity in plants. The stimulation of root length and surface area is one of typical plant responses to P deficiency and is the results of allocation of more assimilates to roots (Vance et al., 2003). However, like tea in this work, there is also plant species showing no induction of root surface area under P deficiency.
The values of uptake rate in bean in both sufficient and deficient plants were higher than tea plants. On the other hand, reduction of uptake rate in P deficient tea plants was higher (94%) in comparison with bean (72%). It could be concluded that, in addition of lower external P efficiency in terms of P acquisition capacity due to root surface area, tea plants has lower uptake capacity than bean plants. For an exact description of difference between these two species, an experiment with radiolabelled P in a short term uptake test is necessary.
In general, effect of P deprivation on shoot growth (leaf number, leaf area and shoot weight) was much more prominent than on roots. Reduction of hydraulic conductance, cell division and cell expansion as well as decrease in leaf initiation and emergence of new leaves were proposed as factors negatively affect shoot growth of P deficient plants (Mollier and Pellerin, 1999; Nielsen et al., 2001; Chiera et al., 2002).
Reduction of leaf area could be an important cause of growth inhibition by low P supply. Lower leaf area results in lower photosynthesis and carbohydrate supply for plant. Moreover, as was demonstrated in this work, photosynthesis rate per unit leaf area was strongly inhibited in P deficient plants. Considering data of stomatal conductance and Ci values it could be concluded that the main cause of reduction of net photosynthesis rate was lower stomatal conductance, so that, reduction of net photosynthesis rate (A) was 54% and decrease in stomatal conductance was 42%. Another contributing factor in reduction of net photosynthesis rate, was inhibition of photochemistry of P deficient leaves, as judged by significant reduction of Fv/Fm in P deficient plants (p≤0.05). Decrease in Fv/Fm value along with Fv/F0 ratio was considered as photoinhibition and photodamage to photochemical apparatus (Innocenti et al., 2003; Mielke et al., 2003).
Results suggested that, tea plant is an extremely P deficiency tolerant species and tested seed stocks did not differ in response to low P. Because tea plants were being cultivated usually on P deficient acid soils, it seems likely that high tolerance to P deficiency had been developed in this species during past 2500-3000 years of its domestication. This higher tolerance was mainly due to a high internal P use efficiency, which strongly increased with decreasing P supply. The underlying mechanisms for internal use efficiency should be further investigated. Phosphorus deficient tea plants did not develop any strategy for higher P uptake from medium, neither in terms of changes in root architecture nor induction of high affinity P uptake systems. In contrast, an extremely high tolerance of tea plants to P deficiency is mainly due to high internal use efficiency.
The authors acknowledge the Tea Research Institute of Iran, Lahijan, for collection of tea seeds and technical guide for plants culture.