Research Article
Shelf-Life Extension of Ripe Non-Astringent Persimmon Fruit Using 1-MCP
Department of Horticulture, College of Agriculture, Isfahan University of Technology, Isfahan 8415683111, Iran
Oriental persimmons (Diospyros kaki L.) are climacteric fruit whose ripening is regulated by ethylene (Wills et al., 1998). Unlike other climacteric fruit, persimmon harvested at commercial maturity produce only a low amount of ethylene (less than 1.0 nL g-1 h-1 even at peak production), although fruit harvested at a less mature stage produce higher amount (Harima et al., 2003). Persimmon fruit are at their best quality at the end of the pre-climacteric stage, since sugar content are at a maximum and the desirable orange color of the fruit develops just before the onset of the respiratory climacteric and induction of ethylene. Once the climacteric phase begins, rapid softening occurs, resulting in unmarketable fruit with jelly-like flesh within a few days (Nakano et al., 2003).
Sweet persimmon is a major fruit crop widely grown in the central parts of the Iran. Cultivar Nathanzy is one of the best cultivar and is commercially important as it has excellent texture and test. The most limiting factor for distribution and storage of this cultivar is rapid softening during postharvest period. Takata (1981) reported that vacuum infiltration of silver ions, as inhibitors of ethylene action, can delay softening in Fuyu persimmon fruit. However, silver salts can only be applied to flowers and not to food commodities. 1-Methylcyclopropene (1-MCP) has been developed as a new potent inhibitor of ethylene action. 1-MCP is a gas and is structurally very similar to ethylene by binding irreversibly to the ethylene receptor site and has the potential to extend storage life and shelf life of various fruits, vegetables and flowers (Blankenship and Dole, 2003; Sisler and Serek, 1997). The effects of 1-MCP on postharvest behavior of many commodities are being studied widely. It has been demonstrated that the inhibition of the ethylene action delays ripening and senescence in several species of fruits (Sisler and Serek, 1997; Menniti et al., 2006). The success of 1-MCP treatment depends on the method of application, duration and concentration as well as commodity factors such as maturity stage, cultivar and atmosphere (Nakatsuka et al., 1998; Watkins et al., 2000; Valero et al., 2003). Nakano et al. (2003) reported that 1-MCP treatments can inhibit softening in Japanese persimmon fruit (astringent cultivar) without any de-astringency treatment, confirming that the softening process in persimmon fruit is ethylene-dependent. Although Nakano et al. (2002) and Kim and Lee (2005) have reported positive results on astringent persimmon, there is little information about 1-MCP effects on non-astringent and ripe persimmon fruits.
It is not clear if 1-MCP is beneficial for non-astringent and ripe persimmon cultivar Nathanzy that widely grown in the central Iran. Therefore, the aim of this study was to evaluate the effect of 1-MCP concentrations on Nathanzy persimmon to extend postharvest shelf life.
Plant material: Nathanzy persimmon (Diospyros kaki L.) was harvested from a commercial orchard at Nathanz province (central Iran) in 11 October 2006 at commercial maturity (two thirds of the fruit surface orange colored). Fruits were transported immediately for experimentation to the Postharvest Laboratory at the Isfahan University of Technology. Fruits free from visual defects and of uniform weight and shape were sorted and then randomly divided into several lots for each treatment and stored overnight at 7 °C. Three sample lots were used for each treatment with each lot consisting of 25 fruits for assessment. A sample of 20 fruits was analyzed to determine initial firmness, color, Soluble Solid Content (SSC), respiration and titrable acid content.
1-MCP treatment: Persimmon fruits were treated triplicate, using 25 fruits per replicate, with 1-MCP at different concentrations in sealed polyethylene box. 1-MCP gas concentrations were 0.5, 1 and 1.5 μl L-1, based on free container volume. Untreated control and 1-MCP-treated fruit were held in hermitic 28 L polyethylene containers. Ethylblock powder has 0.14% active ingredient and is distributed by Floralife. The compound was weighed (according to the manufacture procedure) and placed in a 2 mL Eppendorf tube. To release the 1-MCP gas, appropriate distilled waster at 20 °C was added to the Eppendorf tubes. After shaking for 45 sec the Eppendorf tubes were placed inside the containers and the tube cap opened. The container lid was immediately sealed and the container was stored at 20 °C for 24 h. Concentrations were referred to the free volume of the container. After treatment with 1-MCP, the containers were ventilated for 1 h in air and the fruit were randomly divided into treatment and inspection day used for quality analysis were obtained by sampling five fruit from three different bags. Fruit were sealed in 30 μm thick, polyethylene bags (O2 transmission 6800 mL m-2/24 h/atm and CO2 transmission 3500 mL/24 h/atm at 20 °C). Bags were stored at 20 ± 1 °C in gas-tight container with a continuous 95% RH. Quality parameters and senescence were evaluated at 20 °C at harvest and on the 5th, 10th, 15, 20, 25 and 30 days after treatment. Senescence was evaluated visually and by measuring fruit softening. Color was measured using a color meter (Minolta CR-200 colorimeter, Japan) calibrated with a white reference plate (Y = 94.3, x = 0.3142 and y = 0.3211). Fruit firmness was evaluated on three points of the fruit equator with a fruit pressure tester with a 9 mm probe tip. Juice was squeezed and pH was measured using a pH meter. Soluble Solid Content (SSC) were measured with a hand refractometer. Titrable acidity was measured by titration of 5 mL samples with 0.1 M NaOH to an endpoint of pH = 8.1. Respiration was measured by placing each treatment and replication of 12 fruits in 4 L glass jar hermitically sealed with a rubber stopper for 1 h. CO2 evaluation in samples taken from the exit flow from each jar, using infrared CO2 detection, following storage the fruits at 20 °C after 15 and 30 days of 1-MCP treatment.
Data were subjected to Analysis of Variance (ANOVA) and Least Significant Difference (LSD) at the 5% level was used for comparing of means using the MSTAT-C. The values presented in the figures were means of three replications.
Decrease in flesh firmness was observed as the fruits ripened and softened with longer storage duration (Fig. 1). Fruit firmness is regarded as a decisive factor determining the degree of fruit ripening and shelf life. In this study, fruit firmness of 15 N (approximately half the initial firmness of 30 N) was considered a minimum threshold as marketing. The shelf life of the fruits treated with 0.5-1.5 μl L-1 1-MCP was 30 and for control (untreated fruits) it was ca. 15 days under room temperature conditions. The shelf life was 2 times longer for fruits treated with 1-MCP than untreated fruits (Fig. 1). On the other hand, the proportion of softened Nathanzy fruit did not significantly (p<0.05) increase until 15 days with 0.5 μl L-1 1-MCP-treated fruit and until 25 days for fruit treated with more than 0.5 μl L-1 (Fig. 1). These observations indicate that 1-MCP treatment at 0.5 μl L-1 or higher extend the shelf life of non-astringent type Nathanzy persimmon fruit by at least 2 times over that of non-treated control fruit.
The amount of Soluble Solid Content (SSC) of fruits showed decrease over storage either in control or treated fruits (Fig. 2). However, the magnitude of the decrease
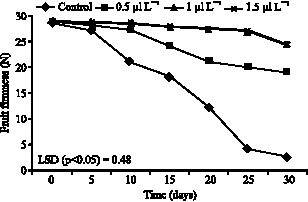 | |
| Fig. 1: | Effects of 1-MCP on fruit firmness in persimmon during storing at ambient air temperatures (20 °C) |
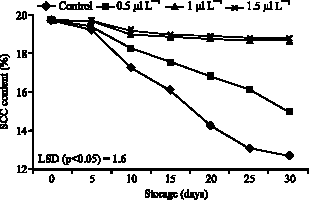 | |
| Fig. 2: | Effects of 1-MCP on SSC content of persimmon during storing at ambient air temperatures (20 °C) |
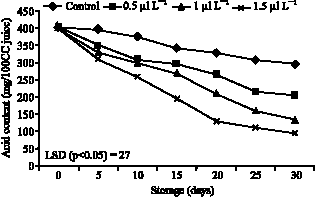 | |
| Fig. 3: | Effects of 1-MCP on acid content of persimmon during storing at ambient air temperatures (20 °C) |
was significantly (p<0.05) affected by 1-MCP treatment. Fruits treated with 1-1.5 μl L-1 1-MCP did not significant change during storage period at 20 °C. In control fruit, SSC decreased from 19.6% (day 0) and reached a minimum of 12.8% (day 30). In fruits treated with 1-1.5 μl L-1 1-MCP, SSC remained high (ca. 18%) after 30 days fruits stored at 20 °C (Fig. 2).
1-MCP treated fruit effectively reduced loss of titratable acidity after 30 days storing at 20 °C (Fig. 3). Fruits treated with 1-MCP (1 μl L-1) were showed that has high fruit titratable acidity after 30 days storing at 20 °C, compared to the control and the differences was highly significant (p<0.05).
When 1-MCP was applied on ripe persimmon fruit, changes in the peel color of the fruit were not significantly observed during storage at 20 °C (Fig. 4). In contrast, visible changes in persimmon skin color do not so much occur after treating fruits with 1-MCP. However, a decrease in hue angle (H °) values became apparent in persimmon during storage. 1-MCP slowed down these changes when utilized after treatment. H° value of 1-MCP treated fruit after 30 days of storing were similar to those of control none-treated fruit. Increasing the 1-MCP
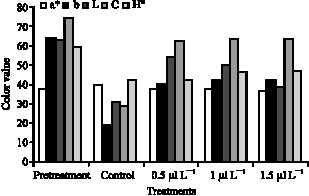 | |
| Fig. 4: | Effects of 1-MCP on color values of persimmon storing at ambient air temperature for 30 days (20 °) LSDa* = 1.1, LSDb = 8, LSDL = 11, LSDC = 9 and LSDH° = 9 |
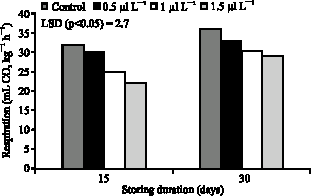 | |
| Fig. 5: | Effects of 1-MCP on respiration of persimmon storing at ambient air temperature (20 °C) |
concentration from 1 to 1.5 μl L-1 should fewer changes in hue angle between days 15 but no significant difference was measured at the end of the storage period.
Treatment of fruits with 1-MCP significantly inhibited respiration of sweet persimmon at room temperatures. Respiration was markedly reduced by 1-MCP until 30 days after treatment (Fig. 5). The initial level of respiration rate for persimmon at 20 °C was 34 mL CO2 kg-1 h-1. Fruit treated with 1.5 μl L-1 1-MCP has significantly lower respiration for day 30, compared to the control none-treated fruits. Respiration in persimmon fruits at the end of storage (30 days) slightly increased, but the final levels being significantly higher in control than in 1-MCP treated fruits.
Present results showed that 1-MCP delayed softening, reduced the loss of Soluble Solid Content (SSC) and titratable acidity in Nathanzy non-astringent persimmon fruits after 30 days storage at ambient air temperatures (20 °C). These results support the observations of Takata (1983), Harima et al. (2003) and Kim and Lee (2005) storing other cultivars in cold or at room conditions. Nathanzy persimmon treated fruits were still acceptable for consumption after 30 days at 20 °C.
While 1-MCP delayed softening of most fruits, other crop species were not affected. 1-MCP delayed softening in avocado by 4.4 days, mango by 5.1 days and papaya by 15.6 days (Hofman et al., 2001). Apple tissue mechanical properties were found to change less in 1-MCP-treated fruit than in untreated fruit (Baritelle et al., 2001). More detailed examinations of fruit softening showed that Polygalacturonase (PG) and cellulose activities were lowered by 1-MCP (Blankenship and Dole, 2003). Given that 1-MCP blocks ethylene perception, it has the potential to prevent or slow limiting factors and process contributing to the loss of shelf life that are dependent upon ethylene (Sisler and Serek, 1997). In pear fruit at the climacteric stage, treatments of 1-MCP above 10000 nl L-1 significantly suppressed ethylene production and fruit softening whereas treatments below 1000 nl L-1 had no effect (Kubo et al., 2003). Rupasinghe et al. (2000) found that apple fruit treated with 1-MCP at 1000 nl L-1 and greater exhibited complete inhibition of ethylene production and reduced fruit softening after 60 days storage, but not at 100 nl L-1 and less. This indicated that the threshold concentration of 1-MCP in apple is about 1000 nl L-1. These observations suggest that 1-MCP concentrations required to inhibit ethylene action will vary greatly with stage of maturity or ripening and/or crop species. A higher concentration of 1-MCP may be required to get maximal effects as fruit produce more ethylene. In this experiment, persimmon fruit responded to a concentration of as low 0.5 μl L-1 and the beneficial effects were almost saturated at 1.5 μl L-1 which resulted suppress respiration, reducing loss of SSC and titratable acid content of fruits.
In conclusion, present results indicated that 1-MCP treatment markedly extended the shelf life of non-astringent persimmon cultivar Nathanzy fruit and hence helps keeping quality for up to 30 days at 20 °C as compared to control that maintained the quality for 2 week. Therefore, we recommended a 1-MCP treatment at the concentration of only 1 μl L-1 following harvest at early ripening stage.