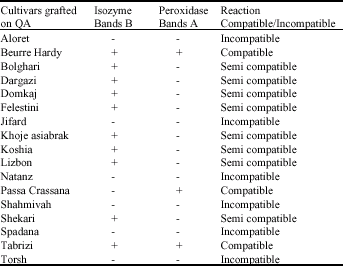
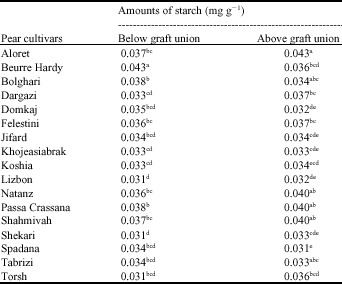
Research Article
Identification of Graft Incompatibility of Pear Cultivars on Quince Rootstock by Using Isozymes Banding Pattern and Starch
Department of Horticultural Science, College of Agriculture, Ferdowsi University, P.O. Box 91775-1163, Mashhad, Iran
Shahriari Farajollah
Department of Plant Biotechnology and Breeding, Ferdowsi University, P.O. Box 91775-1163, Mashhad, Iran
Hamid Hassanpour
Department of Horticultural Science, College of Agriculture, Ferdowsi University, P.O. Box 91775-1163, Mashhad, Iran