Research Article
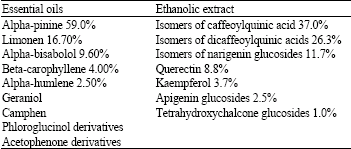
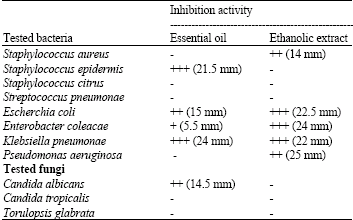
Chemical Constituents and Antimicrobial Activity of Helichrysum stoechas
Department of Biology, Faculty of Science, University of Omar El-Mukhtar, Libya
S.S. El-Feky
Department of Botany, Faculty of Science, University of Tanta, Tanta, Egypt