Research Article
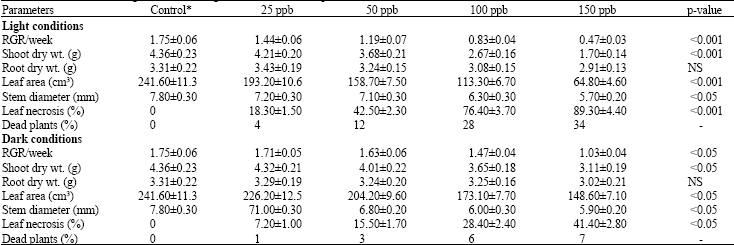
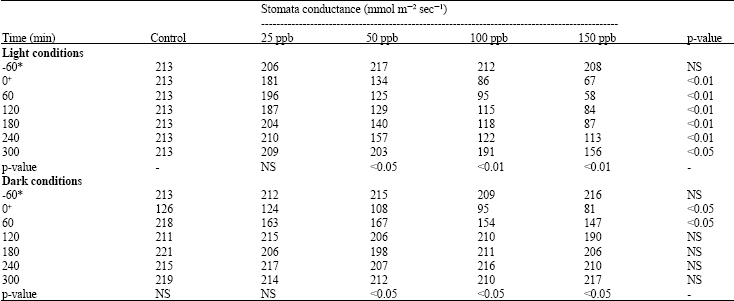
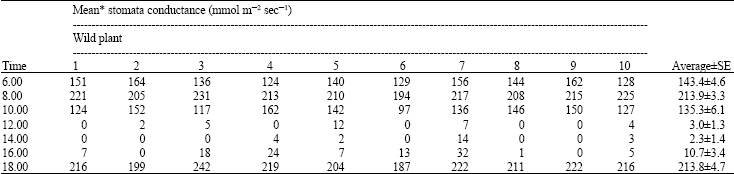
Growth and Stomatal Conductance of Prosopis cineraria (Ghaff Tree) Exposed to Sulphur Dioxide
Department of Biology, College of Science, Sultan Qaboos University, P.O. Box 36, Alkhod 123, Sultanate of Oman
Haitham Al-Amri
Department of Biology, College of Science, Sultan Qaboos University, P.O. Box 36, Alkhod 123, Sultanate of Oman
Abdulrahman Al-Hinai
Department of Biology, College of Science, Sultan Qaboos University, P.O. Box 36, Alkhod 123, Sultanate of Oman
Neelam Sherwani
Department of Biology, College of Science, Sultan Qaboos University, P.O. Box 36, Alkhod 123, Sultanate of Oman