ABSTRACT
Longitudinal sections revealed that radicles of laboratory-grown slash (Pinus elliottii Engelm.) and loblolly (Pinus taeda L.) pine and lateral roots of naturally-grown and container-grown loblolly pine developed a rootcap that was 3-8 cell layers wide around the root proper at the level of the quiescent center and 8-32 cell layers from the quiescent center to the distal tip. Rootcap cells contained amyloplasts which were located mainly in the rootcap center and central columellar cells of the rootcap. Large amyloplasts were located in and around the rootcap center while small amyloplasts were found in other columellar cells. Amyloplast number per rootcap cell varied with pine species and with how seedlings were grown. There were fewer amyloplasts in radicles than in lateral roots. The spatial distribution of amyloplasts was similar between radicles and lateral roots and they were not found exclusively on the bottom of rootcap cells.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2007.605.610
URL: https://scialert.net/abstract/?doi=ajps.2007.605.610
INTRODUCTION
While the rootcap was widely regarded as the site of gravity perception in higher plants (Pilet, 1972; Barlow, 1974; Hensel, 1986; Moore and McClelen, 1989), others indicated that gravity perception is not limited just to the rootcap (Wolverton et al., 2002). Within the rootcap, the columellar cells affected gravitropism (Moore and Miller, 1993) and within these cells amyloplasts were reported to act as statoliths. However, studies failed to furnish a direct link between amyloplast behavior and root gravitropism (Perbal, 1974a and b). Therefore, the roles of the rootcap and amyloplasts in root gravitropism are uncertain (He, 2003).
Only a few papers on pine root gravitropism have been published. Westing (1964) pointed out that gravity was the orienting force for gravitropism in eastern white pine (Pinus strobus L.). South et al. (2001) reported that some taproots of container-grown longleaf pine (Pinus palustris Mill.) seedlings lost gravitropism but afterwards some lateral roots exhibited positive gravitropism. Data on the presence of amyloplasts in rootcaps of Pinus have not been published (He, 2003).
Publications on pine rootcap development have also been limited. The embryo of pinyon pine (Pinus edulis Engelm.) was reported to have rootcap initials and parts of the root promeristem in eastern white pine and maritime pine (Pinus pinaster Aït.) gave rise to the rootcap as well as to the cortex (Popham, 1966). Mycorrhizal pine roots did not have rootcaps (Zimmermann and Brown, 1971). Therefore, this research focuses on anatomical aspects of both radicle and lateral root tips. The objectives were: (1) to observe if a rootcap develops on the tip of a radicle and a lateral root of slash and loblolly pines and (2) to observe if rootcap cells of both pine radicles and lateral roots, if any, contain amyloplasts.
MATERIALS AND METHODS
Investigations were conducted from February to June 2001 (2001 test) and from June to September 2002 (2002 test) at the Southern Forest Nursery Management Cooperative laboratory. Microscopic work was carried out in Dr. Roland Dute=s lab and at the Advanced Microscopy and Imaging Lab (Dr. Michael Miller=s lab) of Department of Biological Sciences, Auburn University. The International Forest Seed Company (Odenville, AL) provided slash and loblolly pine seeds.
2001 test: Forty seeds for each species were germinated in moistened tissue paper in a plastic box, with the radicle end of the seed facing downward. Water was changed daily from the 3rd day. When radicles reached an average of 3.6 and 2.9 cm for slash and loblolly pines, respectively, 20 seedlings with straight radicles for each species were selected and transferred into erect CYG seed growth pouches (Mega International, Minneapolis, MN 55416). These pouches were rotated counterclockwise so that radicle growing tips were oriented horizontally. A loaded pouch holder was placed in a plastic tub with a layer of water 4 cm deep. The tub was placed under a fluorescent lamp in the lab. After the slowest growing loblolly pine radicles developed conspicuous gravity-induced curvatures [with an average seedling length of 8.4 cm], tips less than 1 cm (![]() = 0.7 cm) were cut from radicles of both slash and loblolly pine.
= 0.7 cm) were cut from radicles of both slash and loblolly pine.
Dissected radicle tips were immediately fixed in formalin-acetic acid-alcohol (FAA), dehydrated, infiltrated and then embedded in paraffin at the Tissue Embedding Center. Longitudinal sections were cut 8-10 μm thick using a microtome. Sections were stained with Schiff=s [periodic acid-Schiff stain (PAS)]/Fast Green staining mixture. Observations of the root tip structure were made with a light microscope.
2002 test: Laboratory-grown seedlings-Eighty seeds for each of the two species were germinated in moistened tissue paper in a plastic box, with the radicle end of the seed pointing to the ground. Water was changed daily (starting on the 5th and 7th days for slash and loblolly pine, respectively). To get a similar seedling growth status of these two species for microscopic observations on rootcap structure, slash pine was sown 4 day after loblolly pine. When radicles were 2-3 cm long, 10 seedlings were selected and transferred into an erectly placed growth pouch. Five to six pouches were used for each species. Growth pouches were loaded onto a pouch holder and the holder was then placed in a plastic tub containing 3-4 cm of water. Bottoms of these pouches were cut off so that water in the tub could be absorbed by the tissue paper inside the pouch. The tub was put on a lab desk under a Model M-209 Dazor Floating Fixture (lamp, 118 v, 0.5 A) (Dazor MFG Corp., St. Louis, MO). When radicles reached an average of 4.9 cm for slash pine and 4.7 cm for loblolly pine, 11 radicle tips were dissected (each segment about 0.4 cm long).
Naturally-grown seedlings-Lateral roots of 10 naturally-grown loblolly pine seedlings were obtained from the nursery of the Southern Forest Nursery Management Cooperative. Seedling height averaged 12.8 cm with 11.6 first-order lateral roots per plant. First-order lateral root tips about 0.4 cm long were dissected for microscopy.
Container-grown loblolly pine seedlings--Ten 11-week-old container-grown loblolly pine seedlings, sown in late April 2002, were also obtained from the nursery of the Southern Forest Nursery Management Cooperative. Seedling height averaged 19.3 cm and the number of first-order lateral roots per plant averaged 13.3. First-order lateral root tips about 0.4 cm long were dissected and fixed for microscopic observations.
Excised radicle and lateral root tips were fixed for 24 h in the buffered glutaraldehyde fixative (0.2 M phosphate buffer, pH 6.8) in a refrigerator. Specimens were washed with 10% ethanol and then dehydrated in a graded ethanol series of 10, 30, 50, 75, 95 and 95% (18 min per step). A JB-4 medium was used. The specimens were infiltrated in a catalyzed solution A (of the JB-4 medium) for 4 h in the refrigerator, evacuated for 30 min and then embedded in complete JB-4 plastic embedding medium. After sufficient polymerization, the cutting surface of the specimen block was trimmed. A Sorvall JB-4 microtome (Stocker and Yale, Inc., Beverty, MA) was used to cut examples into 4 μm in thickness. Slides were stained with periodic acid-Schiff=s-aniline blue black (PAS-ABB). After drying, coverslips were applied and the slides were left overnight on a slide warmer.
Observations on the root tip structure were made using a light microscope on 30 representative sections. Digital images were taken using the Zeiss Axiovert 200 LM with a Photometrics CoolSnap camera. Rootcap cell layers were counted from 3 representative sections and amyloplast numbers were counted by randomly selecting 30 amyloplast-containing rootcap cells for each treatment from different parts of the field of view. Data were analyzed by Excel 2000 and SPSS 10.0.
RESULTS
Pine radicles and lateral roots developed a rootcap: Longitudinal sections indicated the presence of a rootcap in laboratory-grown slash and loblolly pine radicles and naturally-grown and container-grown loblolly pine lateral roots (Fig. 1-4). Compared with maize (Zea mays L.) and wheat (Triticum aestivum L.), the rootcap of loblolly pine was not as easily recognized from other parts of the root tip. The rootcap of these two pine species took on a cone-shaped appearance and covered the tip of the radicle or lateral root proper.
Sections from the 2001 test showed the rootcap of slash and loblolly pine radicles was 3-5 cells (![]() = 4.0,
= 4.0, ![]() = 4.3) wide around the radicle proper at the level of the quiescent center and 8-31 cells (
= 4.3) wide around the radicle proper at the level of the quiescent center and 8-31 cells (![]() = 20.0,
= 20.0, ![]() = 22.0) deep from the quiescent center to the most distal cell layer of the tip. Statistical analyses suggested that there was no significant difference (α = 0.05) between the two species in cell layers both around the radicle proper (t1 = 0.946, t0.025,17 = 2.110) and from the quiescent center to the distal tip (t2 = 0.56, t0.025,17 = 2.110).
= 22.0) deep from the quiescent center to the most distal cell layer of the tip. Statistical analyses suggested that there was no significant difference (α = 0.05) between the two species in cell layers both around the radicle proper (t1 = 0.946, t0.025,17 = 2.110) and from the quiescent center to the distal tip (t2 = 0.56, t0.025,17 = 2.110).
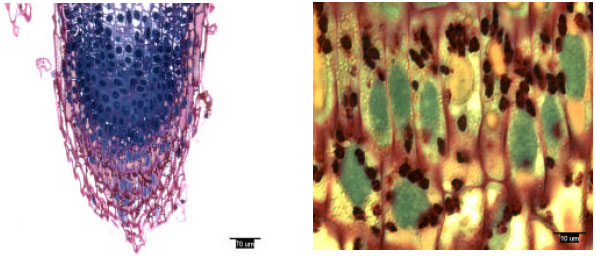 | |
| Fig. 1: | Radicle tip structure of laboratory-grown slash pine, showing amyloplasts |
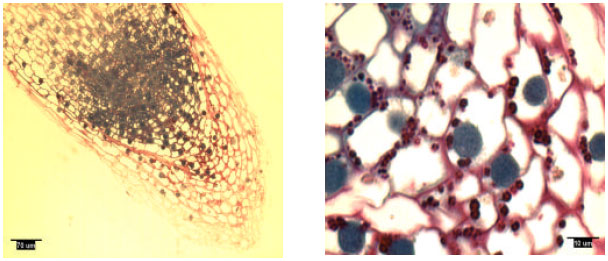 | |
| Fig. 2: | Radicle tip structure of laboratory-grown loblolly pine, showing amyloplasts |
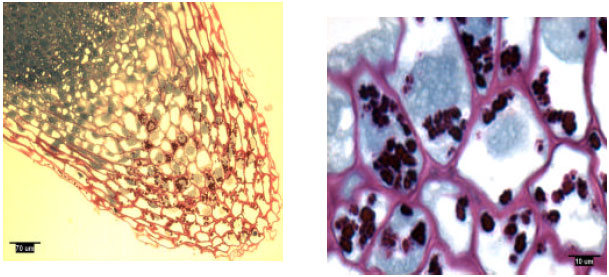 | |
| Fig. 3: | Lateral root tip structure of naturally-grown loblolly pine, showing amyloplasts |
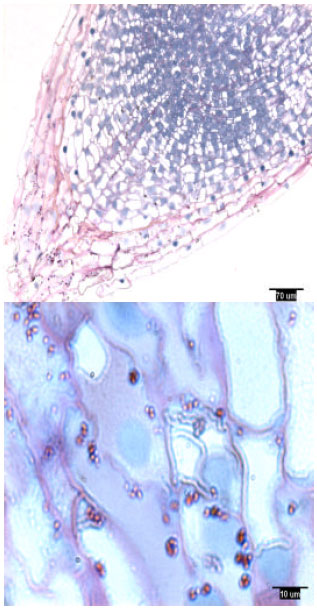 | |
| Fig. 4: | Lateral root tip structure of container-grown loblolly pine, showing amyloplasts |
In the 2002 test, the rootcap of laboratory-grown slash and loblolly pine radicles and that of naturally-grown and container-grown loblolly pine lateral roots were 3-8 cells (![]() = 4.0,
= 4.0, ![]() = 4.8,
= 4.8, ![]() = 5.3,
= 5.3, ![]() = 3.7) wide around the root proper at the level of the quiescent center. Cell layers distal to the quiescent center for laboratory-grown slash and loblolly pine radicles and naturally-grown and container-grown loblolly pine lateral roots were 11-32 (
= 3.7) wide around the root proper at the level of the quiescent center. Cell layers distal to the quiescent center for laboratory-grown slash and loblolly pine radicles and naturally-grown and container-grown loblolly pine lateral roots were 11-32 (![]() = 16.0,
= 16.0, ![]() = 19.0,
= 19.0, ![]() = 25.3,
= 25.3, ![]() = 29.3).
= 29.3).
Rootcap cells of pine radicles and lateral roots contained amyloplasts and amyloplasts were not found exclusively on the bottom of rootcap cells: Amyloplasts were present within the rootcap cells of laboratory-grown slash and loblolly pine radicles and naturally-grown and container-grown loblolly pine lateral roots (Fig. 1-4). These amyloplasts were distributed mainly in: (1) the rootcap center, a central area just below the quiescent center (Fig. 5); (2) other upper central columellar cells of the rootcap and (3) the middle cell layers of the rootcap around the root proper.
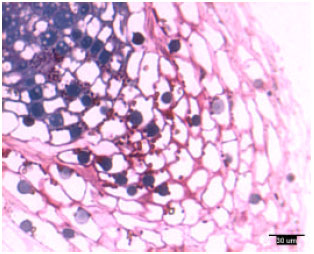 | |
| Fig. 5: | Radicle tip structure of laboratory-grown loblolly pine, showing the amyloplasts-containing rootcap center and central columellar cells |
Within a rootcap cell, there was no evidence that amyloplasts tended to settle on the bottom of the cell walls of radicles and lateral roots. Amyloplast distribution in a rootcap cell of slash and loblolly pine radicles was similar to that of loblolly pine lateral roots (Fig. 1-4).
Amyloplast number per rootcap cell and its size differed by seedling types and varied from rootcap cell to cell: Amyloplast number per rootcap cell differed among samples. Laboratory-grown loblolly pine radicles contained the fewest amyloplasts per rootcap cell (![]() = 3.7, s = 2.5) while slash pine radicles contained an average of 6.8 (s = 3.1). Statistical analyses indicated that differences in amyloplast number between laboratory-grown loblolly and slash pine radicles and between laboratory-grown slash pine radicles and container-grown loblolly pine lateral roots were significant (α = 0.05). However, there was no significant difference in amyloplast number between container-grown and naturally-grown loblolly lateral roots.
= 3.7, s = 2.5) while slash pine radicles contained an average of 6.8 (s = 3.1). Statistical analyses indicated that differences in amyloplast number between laboratory-grown loblolly and slash pine radicles and between laboratory-grown slash pine radicles and container-grown loblolly pine lateral roots were significant (α = 0.05). However, there was no significant difference in amyloplast number between container-grown and naturally-grown loblolly lateral roots.
Amyloplast size differed by seedling types and from cell to cell within one rootcap. Among the four populations in the 2002 test, container-grown loblolly pine lateral roots had the smallest amyloplasts. The other three populations were similar in amyloplast size. Large amyloplasts were usually located in and around the rootcap center and small amyloplasts were found in the upper central columellar cells of the rootcap and/or in cells above the rootcap center.
DISCUSSION
Cell division and growth zones of pine root tip: Generally, a root tip typically includes a rootcap, the meristematic zone, the elongation zone and the maturation zone (Li, 1984). The rootcap protects the meristematic zone (Esau, 1977) and the quiescent center (QC), located in the distal center of the meristematic zone and does not include the initials for the rootcap. Above the distal center are located the precursors of dermal, fundamental (ground) and vascular tissue systems. According to Hanstein=s histogen theory, these initials included the dermatogen, periblem and plerome, respectively (Esau, 1977). Above the meristematic zone was the elongation zone that mostly affected root growth (Esau, 1977; Li, 1984). Lateral roots and ectomycorrizal infections of short roots occurred in and above the maturation zone.
According to Li (1984), the length of the meristematic and elongation zones was about 1-2 and 2-5 mm, respectively. However, depending on species, the length of the maturation zone could range from several millimeters to several centimeters. Esau (1977) reported that, in maize roots, the point of the greatest rate of cell formation was located about 1.25 mm basipetally from the rootcap tip. Cell division stopped about 2.5 mm from the rootcap tip. Also, the length of various cell division and growth zones differed with root types or orders within a genotype and even differed among individual seedlings with the same root types or orders.
Anatomical observations indicated slash and loblolly pine roots had cell division and growth zones that were smaller than those mentioned above. The measured length of the rootcap distal to QC was about 0.3-0.7 mm. The length of meristematic zone was about 0.2-0.6 mm. In one naturally-grown loblolly pine lateral root tip, the zone of active cell division was located 0.7 mm from the rootcap tip. The elongation zone began about 0.5-1.5 mm from the rootcap tip.
Amyloplasts in rootcap cells and pine root gravitropism: Geotropic growth was observed in radicles and it typically does not occur in lateral roots. Although lateral roots were not geotropic, they contained amyloplasts. Contrary to common belief, amyloplasts in vertically grown radicles were fewer in number than those in horizontally grown lateral roots. Moreover, the spatial distribution of amyloplasts was similar for both radicles and lateral roots. Amyloplasts were found throughout the rootcap cell and did not appear to settle on the bottom of cells. Based upon such evidence, the absence of geotropic growth in lateral pine roots cannot be attributed to either an absence of amyloplasts or to a difference in settlement pattern of amyloplasts. If amyloplasts are required for geotropic response in pine taproots, then perhaps an additional factor is needed in order for amyloplasts to function as statoliths.
CONCLUSIONS
This study demonstrated that normal rootcaps developed, just as in other species, in radicles and lateral roots of slash and loblolly pines. It also demonstrated amyloplasts developed within radicles and lateral roots in the two pine species. When the spatial distribution of amyloplasts was concerned, they were not found exclusively on the bottom of rootcap cells. Therefore, it is interesting to reveal the relationship between amyloplasts showing up in rootcaps and gravitropism development in pine roots.
ACKNOWLEDGMENT
We express our appreciation to Drs. Roland Dute and Michael E. Miller at Auburn University, Department of Biological Sciences, for providing labs and to the International Forest Seed Company (Odenville, AL) for providing seeds of slash and loblolly pine seeds.
REFERENCES
- Barlow, P.W., 1974. Recovery of geotropism after removal of the root cap. J. Exp. Bot., 25: 1137-1146.
Direct Link - Hensel, W., 1986. Cytodifferentiation of polar plant cells. Use of anti-microtubular agents during the differentiation of statocytes from cress roots (Lepidium sativum L.). Planta, 169: 293-303.
Direct Link - Moore, R. and C.E. McClelen, 1989. Characterizing pathways by which geotropic effectors could move from the root cap to the root of primary roots of Zea mays. Ann. Bot., 64: 415-423.
Direct Link - Moore, R. and I. Miller, 1993. Cellular differentiation in root caps of Zea mays that do not secrete mucilage. Plant Cell Environ., 16: 1003-1009.
Direct Link - South, D.B., J. Shelton and S.A. Enebak, 2001. Geotropic lateral roots of container-grown longleaf pine seedlings. Native Plants J., 2: 126-130.
Direct Link - Wolverton, C., J.L. Mullen, H. Ishikawa and M.L. Evans, 2002. Root gravitropism in response to a signal originating outside of the cap. Planta, 215: 153-157.
Direct Link








