Research Article
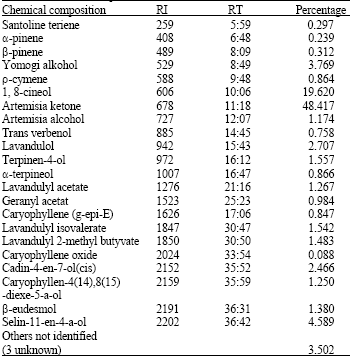
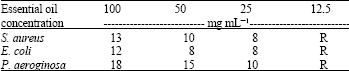
Chemical Composition and Antibacterial Activity from Essential Oil of Artemisia sieberi Besser subsp. Sieberi in North of Iran
Faculty of Rang Management Golestan University, Iran
G.A. Heshmati
Faculty of Rang Management Golestan University, Iran
M. Mazandarani
Islamic Azad University, Gorgan Branch, Iran
M.B. Rezaei
Forests and Rangeland Institute, Tehran, Iran
A.R. Ahmadi
Golestan University of Medical Sciences, Iran
E.O. Ghaemi
Golestan University of Medical Sciences, Iran
S. Bakhshandeh Nosrat
Golestan University of Medical Sciences, Iran