Research Article
Genetic Variation and Segregation of DNA Polymorphisms in Gibberella zeae Detected with AFLP and RAPD Markers
Not Available
Heiko P. Parzies
Not Available
Thomas Miedaner
Not Available
Fusarium head blight, caused by Gibberella zeae (Schwein.) Petch (anamorph: Fusarium graminearum Schwabe) is a destructive disease of wheat, causing reduction in yield and impairing quality of grains by contamination of toxicogenic mycotoxins such as deoxynivalenol (DON), its derivative 3-acetyl deoxynivalenol (3-ADON) and nivalenol (NIV), which are harmful to humans and animals (Marasas et al., 1984; McMullen et al., 1997). The role of these mycotoxins have been implicated as aggressiveness factors in plant pathoge nesis (Proctor et al., 1995; Bai et al., 2002). Genetic variation of aggressiveness among isolates of G. zeae collected within a single field or from different geographical areas is commonly reported by Miedaner and Schilling (1996), Miedaner et al. (2001) and Muthomi et al. (2002) but the mechanisms of such variation is not well understood. The application of PCR-based technologies such as Amplified Fragment Length Polymorphisms (AFLP) and Random Amplified Polymorphic DNA (RAPD) and other DNA based markers have facilitated greatly the genetic analyses of phytopathogenic fungi (Brown, 1996). Based on the DNA sequences of six genes, O’Donnell et al. (2000) proposed that G. zeae consists of seven phylogeographical lineages coming from different geographical origins which may eventually be considered as species. A high-density genetic linkage map of G. zeae has been published (Jurgenson et al., 2002). The map was constructed from an interlineage cross between Kansas parent Z-3639 and Japanese parent R-5470 generating 99 progeny. Analysis of Quantitative Trait Loci (QTL) associated with pathogenicity and aggressiveness of this population has been done (Cumagun et al., 2004). For linkage mapping and QTL analysis, we attempted to use another population from an intralineage cross between two DON-producing parents FG24 (Hungary) and FG3211 (Germany) with 153 progeny. Both isolates were characterized by pink white colony, red pigmentation on Potato Dextrose Agar (PDA) and aerial growth habit. Aggressiveness and DON production of the two parents did not differ greatly, but we expected that these characters will segregate quantitatively in the progeny. Based on classification proposed by O’Donnell et al. (2000), these isolates belong to lineage 7. Map construction, however, has been laborious and unsuccessful due to the monomorphic character of the parents and the high frequency of segregation distortion, i.e., markers deviating from the Mendelian ratio. We therefore resorted simply to assessing the polymorphism of progeny population by AFLP and RAPD markers and comparing the genetic similarity and distance of the two parents with Z-3639 and R-5470.
Crossing population: Two pairs of parents, FG24 (Hungary)xFG3211 (Germany) and Z-3639 (Kansas)x R-5470 (Japan), were crossed in the lab of B. Bowden and J. Leslie at Kansas State University, USA (Bowden and Leslie, 1999). FG24, FG 3211 and Z-3639 belong to lineage 7 and R-5470 to lineage 6 (O’ Donnell et al., 2000). Only the first parent cross consisting of 153 progeny was analysed for segregation.
Culture media, DNA extraction and quantification: One hundred fifty seven isolates of G. zeae (including the four parents) were routinely cultivated in SNA (synthetic nutrient-poor mineral agar) according to Nirenberg (1981). Mycelia of the parent isolates and their progeny were produced in 100 mL flasks containing 20 mL of liquid SNA (without agar) and supplemented with 0.1% yeast extract and 10-fold increase of sugars. A 100 mL flask was inoculated with 1-3 mycelial plugs of a vigorously growing culture of each isolate and incubated at room temperature with natural light for 4 to 6 days on a shaker at 100 rpm. After incubation, pure mycelia were filtered off from the liquid culture on filter paper disks using a Buechner type funnel and a filter flask connected to a water jet pump. Mycelia were washed once with sterile-distilled water on the filter paper and scraped off after excess liquid had been removed. Mycelia were immediately frozen at -20°C for storage and then freeze-dried for 48 h prior to DNA extraction. Dried mycelia were crushed into a fine powder in a mixer-mill MM2 (Retsch, Haan, Germany) at 80 rpm for 30 sec.
Total genomic DNA was isolated from 50 mg mycelium by a microextraction protocol according to Möller et al. (1992) including treatment with RNase A. DNA was run in an electrophoresis chamber for 3 h at 50 V. Bands were stained with ethidium bromide and photographed under UV. Intensity of bands was quantified using standard digested Lambda DNA.
AFLP analysis: AFLP analysis was based on Vos et al. (1995) with some modifications using non-radioactive staining (Zhong and Steffenson, 2001; Cumagun et al., 2006). AFLP core reagent kit (Life Technologies, Inc., Grand Island, NY) was used to digest and ligate DNA as template for PCR. AFLP primers (Life Technologies Inc., Bethesda, MD) with one selective base were used in preamplification. The sequences of the preamplification primers were: 5’-CTC GTA GAC TGC GTA CCA ATT C-3’ (E + C) and 5’-GAC GAT GAG TCC TGA GTA A-3’ (M +A). Primers with two selective bases were used for selective amplification (Table 1).
After the amplification reactions, samples were loaded onto 6% polyacrylamide gels (Sequa Gel, Biozym, Oldendorf) including Low Mass Ladder™ (Life Technologies Inc., Bethesda, MD) as a standard size marker. Gels were visualized by silver staining (DNA Silver Staining System, Promega, Madison, WI). AFLP bands from 100 to 2000 bp in size were scored using TotalLab v1.10 software package (Nonlinear Dynamics. Ltd., Newcastle upon Tyne).
RAPD analysis: RAPD analysis was according to Schilling et al. (1994) using RAPD decamer primer kit # UBC 100/1 (University of British Columbia, Vancouver) and OPT (Operon Technologies, Alameda, CA) with their sequences as shown in Table 2. PCR products were loaded and DNA fragments were resolved by electrophoresis (2 V cm-1) in 1.5% agarose and 1xTAE (Tris-acetate-EDTA) for 5 h aside with a 200 bp O’RangeRuler DNA ladder (MBI Fermentas, Germany). Gels were stained in ethidium bromide and photographed under UV light with a Polaroid camera.
TRI5 analysis: Ten ng of genomic DNA from each parent and progeny were digested with 4 units of MseI (New England BioLabs, Beverly, MA) for 2 h. Digested DNA were amplified with two primers: 5’-GGCATGGTTGTATACAGC-3’ and 5’-CAGAGTGATCTCATGGCAGG-3’ and run on 1% agarose gel at 75 V for 2 h. Gels were stained and visualized as described previously.
Data analyses: AFLP and RAPD bands were scored manually and analysed as binary data with 1 (band present) and 2 (band absent) at a particular location in each lane. Using the program Tools for Population Genetic Analyses (TFPGA) version 1.3 (Miller, 1997), genetic similarity between isolates were calculated (Nei, 1972). A dendrogram was constructed using the unweighted pair-group method (UPGMA) following Wright’s (1978) modification of Roger (1972) distance.
| Table 1: | AFLP primer combinations used and number of polymorphic bands for the parent isolates of Gibberella zeae |
 | |
 | |
| aNP: No amplification products, bND: Not Determined, *: Selected for further analysis (N = 6) | |
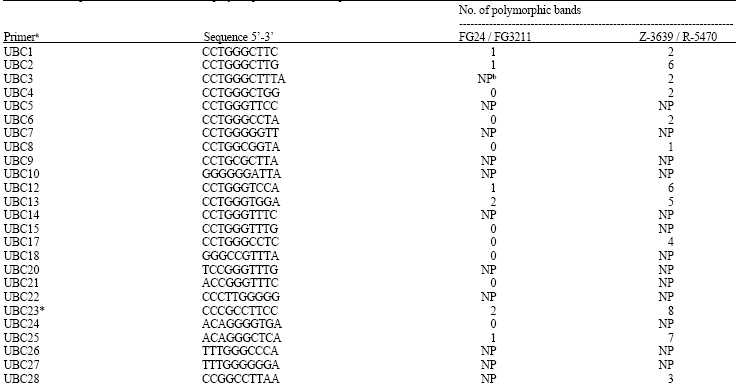
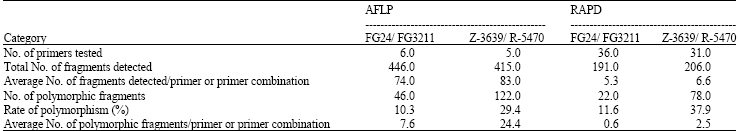
AFLP and RAPD analyses: The number of AFLP and RAPD polymorphic markers in the G. zeae parent isolates Z-3639 and R-5470 was about 4 times as much as in FG24 and FG3211 (Table 1 and 2), which is consistent with the rate of polymorphism (Table 3). Seven AFLP primers did not show amplification products for both parent isolates. The number of AFLP polymorphic bands ranged from 1 to 12 for FG24 and FG3211 and 4 to 35 for Z-3639 and R-5470 (Table 1). Six AFLP primer combinations namely E-AT+M-CA, E-GA+M-TA, E-GA+M-AT, E-GC+M-AG,E-CC+M-CG and E-TG+M-TT generating the highest number of polymorphic bands were selected for further analysis (Table 1).
| Table 2: | RAPD primers used and number of polymorphic bands for the parent isolates of Gibberella zeae |
 | |
 | |
| a: Primer code (University of British Columbia, Vancouver, Canada; Operon Technologies, Alameda, CA), b: NP: No amplification products; *: Selected for further analysis (N = 4) | |
| Table 3: | Rate of polymorphism detected by AFLP (using selected primer combinations) and RAPD analysis for the parent isolates of Gibberella zeae |
 | |
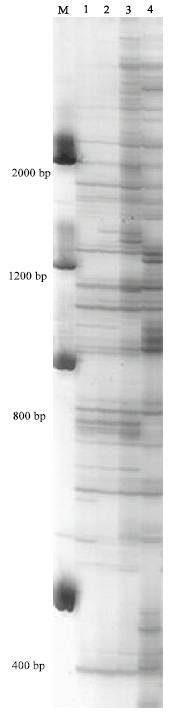 | |
| Fig. 1: | AFLP gel showing low polymorphism between FG24 (Lane 1) and FG3211 (Lane 2) and high polymorphism between Z-3639 (Lane 3) and R-5470 (Lane 4) of Gibberella zeae amplified with primer combination E-GA+M-AT. Molecular marker (M) is a 2000 bp ladder |
| Table 4: | Genetic similarity of the parent isolates of Gibberella zeae using AFLP and RAPD markers according to Nei (1972) |
 | |
| a: Based on 6 AFLP primer combinations, b: Based on 26 RAPD primers | |
Most RAPD primers did not generate amplification products with 17 primers for FG24 and FG3211 and 25 primers for Z-3639 and R-5470. The maximum number of polymorphic bands was four for FG24 and FG3211 and eight for Z-3639 and R-5470. RAPD primers UBC23, UBC29, UBC30 and UBC43 were selected for further analysis (Table 2).
It was obvious that AFLP markers generated higher number of polymorphic markers compared to RAPD. One to 12 polymorphic markers per AFLP primer combination within 100 bp to 2000 bp and one to four per RAPD primer were detected between FG24 and FG3211. Genetic similarity between these two parents was quite high despite their geographical separation as compared to Z-3639 and R-5470 (Fig. 1 and Table 4). Dendrograms generated from both AFLP and RAPD markers showed that the two European parents closely resembled the Kansas parent confirming their lineage grouping while the Japanese parent was obviously far distant from the three parent isolates, thus belonging to a separate lineage (Fig. 2A and B). The genetic distances of the Japansese parent is close to 0.70 and 0.80 based on AFLP and RAPD analyses, respectively (Fig. 2A and B). Molecular analysis was also associated with the cultural characters of the parents in which the European and Kansas parents are pink white and aerial whereas the Japanese parent is pink yellow and flat. The Japanese parent, being one of the parents used in the first genetic map of the fungus, belonged to lineage 6 (Jurgenson et al., 2002). About 50% difference of the observed AFLP bands between the Kansas and Japanese parent is consistent with that of Jurgenson et al. (2002) although we used silver staining method.
| Fig. 2: | Dendrograms of the parent isolates of Gibberella zeae based on AFLP (A) and RAPD markers (B) Genetic distances were assessed by UPGMA cluster analysis using Roger’s distance (1972) and Wright’s (1978) modification. Genetic distances for AFLP and RAPD were calculated from the combined data of six primer combinations and 26 primers, respectively |
A total of 71-122 bands per lane were generated using AFLP compared to 1-11 using RAPD. AFLP examines the whole genome and is highly reproducible (Vos et al., 1995). These features make AFLP a more reliable technique than RAPD. Reproducibility is due to the specificity of primer annealing and is homologous to the adapter sequence and the restriction site sequence. RAPD has been criticized for lack of reproducibility (Williams et al., 1990). We found, however, a good association between the two markers although the two methods of analysis could reveal genetic variation in different regions of the genome. Despite the advantages of AFLP, use of additional primer combinations could not detect enough polymorphism between FG24 and FG3211. This is consistent to the finding that some isolates of G. zeae coming from Southern and Eastern Europe are closely associated based on their principle coordinate analysis (Schilling, 1996). A total of 31 AFLP primer combinations were screened for polymorphisms between the parents FG24 and FG3211 (Table 1 and 3), from which only six primer combinations that generated the high estpolymorphisms were used for the progeny population. For RAPD analysis, only four (UBC 23, 29, 30 and 43) out of 56 primers, which generated two to four polymorphic markers, were selected for the progeny population. Both marker systems showed a very low polymorphism between the two parents in this cross and high segregation distortion among the progeny which made it difficult to construct the map. Bowden et al. (2002) encountered the same problem using a narrow cross within lineage 7. The Kansas parent Z-3639 was paired with PH-1 from Michigan. To solve the problem of segregation distortion associated with the nit marker technique, one parent that had a deletion in the MAT2 (mating type 2) gene was used which made it heterothallic. This technique avoided segregation distortion associated with nit markers. Segregation distortion was also reported in the Z-3639xR-5470 cross (Jurgenson et al., 2002). This problem was confirmed in the FG24xFG3211 cross where out of 45 AFLP polymorphic markers detected, only 28 loci (62%) segregated in a 1:1 Mendelian ratio. It is even worse in the case of RAPD, where one out of nine loci has a 1:1 segregation distortion. (Cumagun, 2004). Segregation distortion was also observed in Phytophthora infestans and Leptosphaeria maculans (Van der Lee et al., 1997; Pongam et al., 1998). This is a common phenomenon in linkage analysis when linkage between markers and genes favors or acts against the survival of the individual progeny. Haploid organisms are more affected because the selection that causes the distortion can act virtually during the whole haploid life cycle (Pedersen et al., 2002). Segregation distortion could also be due to differential viability of ascospores isolated from different progeny genotypes. Extending the length of the PCR primers may reduce segregation distortion for AFLP markers (Nikaido et al., 1999). Frequent linkages among AFLP markers were also observed in present study.
Electrophoretic analysis of the TRI5 fragment digested with MseI revealed monomorphic bands between the parents and the progeny, suggesting genetic homology between the European isolates for this locus. There was no association between aggressiveness from three field environments (Cumagun and Miedaner, 2004) and molecular markers (Cumagun, 2004). Similarly, variation in aggressiveness and mycotoxin production was not related to variation in RAPD patterns (Miedaner et al., 2001; Muthomi et al., 2002). The degree of correlation between virulence and molecular markers is often low in populations that reproduce sexually (Burdon and Roelfs, 1985), suggesting that DNA analysis provides a weak predictive information about the variation of aggressiveness and the potential for new pathotypes to evolve.
Overall, the high genetic similarity we found between the two European parents limits construction of a genetic map. Test of polymorphisms of a collection of G. zeae isolates within lineage 7 was done and a high level of genetic diversity among isolates was found with a mean genetic similarity of 0.21 and genetic diversity of 0.41 (Cumagun et al., 2006). Highly polymorphic lineage 7 population (Cumagun et al. 2006) does not support the high genetic similarity of the two European parents. It is possible that we selected the monomorphic FG24 and FG 3211 parents by chance. Therefore, polymorphism of the lineage 7 parent isolates to be used should first be considered in genetic mapping of G. zeae.
We thank Andrea Merz for doing the RAPD analysis. This research was funded by the German Academic Exchange Service (DAAD).