Research Article
Sterile Shoot Production and Direct Regeneration from the Nodal Explants of Potato Cultivars
Not Available
Muhammad Hanif Munawwar
Not Available
Mozammil Hussain
Not Available
Masud Mahmood
Not Available
Potato (Solanum tuberosum L.) is an important cash crop widely cultivated throughout the world. In Pakistan it is cultivated over an area of 112 thousand hectare with an annual production of 2025 thousand tones (MINFAL, 2004-2005). The current yield level (18 tones ha-1) in Pakistan is low compared to other potato growing countries of the world. Potato is prone to several fungal, viral and bacterial pathogens which cause heavy economic losses every year. Recent advances in plant biotechnology have made it possible to produce resistant varieties by introducing desired genes from many different organisms into plants. It is possible to modify agricultural and horticultural crops now, which was otherwise difficult by conventional breeding techniques. A successful and reproducible plant transformation system requires a responsive in vitro regeneration system. Regeneration response in vitro is generally species and often genotype specific (Ritchie and Hodges, 1993).
Potato produces more food per unit area than cereals. It contains 75% water, 19% starch and 2-3% proteins. Apart from this, minerals and Vitamin C, so essential for human health and vigor are also found. In fact, potato has a great potential to ease out the present situation by minimizing pressure on cereals. Valuable foreign exchange can be earned by exporting it to the other countries. There are several reasons for this low productivity, the major one being the non-availability of disease free and certified seed of high yielding potato varieties resistant to pests and diseases for different ecological zones. Yield losses are directly proportional to the intensity of virus infection. Per hectare yield could be easily doubled by the use of healthy and sound seed (Bawden and Kassanis, 1965). There are several ways of bringing about vegetative propagation in vitro: by I) Fragmentation of the shoot, each fragment possessing at least one bud, the growth of which can be triggered by the more or less rapid initiation of the neo-formation of shoots and by 2) Excising the meristem and making it grow on a nutritive medium. In the first case homogenization of the plants is ensured, while in the second case healthy clones are produced (Mumtaz and Quraishi, 1990).
The degeneration in potato crop is a major limiting factor for its successful cultivation in the Punjab province. In spite of the benefits from the plant protection measures and also the comparatively unfavorable climatic conditions in autumn seasons for the increase in aphid population, the deterioration in stocks occurred. The reduction in yield was 22.50% in the seventh season of the crop from the same stock. The survey for the incidence of viruses in crops indicated that all the crops of the growers were contaminated with leaf roll and other viruses to a high degree. Contamination was as high as 50% in these crops (Mirza, 1977).
Anjum and Hakoomat (2004a, b) had made attempts to regenerate shoots from calli (initiated from tuber and leaf explants) of potato cultivars Maris Piper and Desiree on different media.
Hussain et al. (2005) observed that the variety Cardinal has produced maximum number of shoots (17.6) from nodal explant on MS medium containing 2.0 mg L-1 BAP and 0.5 mg L-1 IAA. Variety Diamont has produced 14.3 and variety Altamash has produced 9.0 shoots from the nodal explant on the same media combination. On the other hand shoot apices of Cardinal produced maximum number of shoots 6.3 on MS medium containing 2.0 mg L-1 BAP and 0.5 mg L-1 IAA. Variety Diamont has produced 4.3 and variety Altamash has produced 4.3 shoots on the same media combination. Leaf disc and internodal tissues were least responsive explant for direct regeneration in this study. The number of shoots produced from Cardinal, Diamont and Altamash were 2.0, 0.0, 0.0 both from leaf discs and internodal explants. From these results it was concluded that variety Cardinal has high regeneration potential than Diamont and Altamash and among the explants, nodal tissue is the most responsive tissue for direct regeneration as compared to the shoot apices, leaf discs and internodal explants. BAP at the rate of 2.0 mg L-1 along with 0.5 mg L-1 IAA was found to be the most appropriate media for maximum regeneration. Thus, cultivar, explant and media specific effects have been widely reported in the literature on regeneration of potato.
In this study, tissue culture techniques were used for vegetative propagation of the locally adapted potato cultivars like Cardinal, Desiree, Diamont, Dura, Adora and Burna. The main objective was to have basic virus free shoots and regenerants from potato sprouts for further multiplication and to establish direct in vitro regeneration protocol from nodal explants. The exploitation of the protocol on commercial scale can lead to the production and distribution of virus free seeds of potato in Pakistan.
MATERIALS AND METHODS
Materials: This study was conducted at Agricultural Biotechnology Programme, National Agricultural Research Center (NARC), Islamabad. The tubers of potato varieties Diamont, Cardinal, Desiree, Adora, Dura and Burna were obtained from Potato Programme, (NARC). These seed tubers were kept at room temperature for a period of 2-3 months under diffused lights to promote sprouting. Pieces of sprouts were used as the starting material to produce shoots in vitro.
Preparation of medium: Knops medium modified by Quraishi et al. (1986) was used. Nutrients (Macro elements and Micro elements) used in medium and their quantities were as mentioned below:
Knops medium modified by Quraishi et al. (1986)
Solution A
Distilled water. …………………………1000 mL
Macronutrients (mg)
Ca (No3)2, ……………………………..500
KH2 PO4 ………………………………….125
Mg SO4 ………………………………….125
KNO3 .. …………………………………..625
Micronutrients (g)
MnSO4 7 H2 O …………………………….0.2
KI . ………………………………0.25
NiCl2 6 H2 O ……………………………….0.05
CoCl2 6 H2 O ………………………………0.05
TiO2 SO3 5 H2 O ……………………………0.2
ZnSO4 7 H2 O ……………………………..0.1
CuSO4 5 H2 O ……………………………..0.05
BeSO4 4H2O ……………………………….0.1
H3BO3 ………………………………0.05
0.5 mL H2 SO4 at 66°C
Fifty milliliter of the solution (A) was taken and volume was made up to one liter with distilled water. Then 10 mL of this diluted solution (A) was taken for one liter of medium.
Solution B
Fe2 (SO4)3 6H2 O ……50 g was added to 1 L of water and 0.5 mL H2SO4 at 66°C
Fifty millilitre Solution B was taken and added to 1 L of distilled water. Then 10 mL of this diluted solution was taken for completing 1 L of the medium.
Sucrose………………………………….. 20 g
Agar …………………………………….. 8 g
pH was adjusted at 5.8-6.0
The medium was then poured in test tubes covered with hydrophilic cotton and sterilized in autoclave at a temperature of 121°C for 20 min.
Preparation of the sprouts: Sprouts were separated from the tubers and washed thoroughly with ordinary tap water to which a small quantity of detergent (Zip) was added. Sprouts were sterilized with 5% solution of Calcium hypo-chlorite as a sterilizing agent for 15 min; they were finally rinsed with distilled water three times successively at intervals of 15 min under aseptic conditions. Now the material was ready for culture. Either entire sprouts or portions of sprouts were put in test tubes under aseptic conditions. For sub culturing, the shoots were taken out from the test tubes under aseptic conditions and put in culture without any processing. The cultures were then incubated at a temperature of 25±2°C with a light regime of 16 h, under 4,000 lux light intensity.
Fully grown shoots were produced either from entire sprouts or from portions of the sprouts of tubers of potato varieties Diamont, Cardinal, Desiree, Adora, Dura and Burna at the end of month. Each shoot had simple leaves at the base and compound leaves at the top. The range of leaflets in compound leaves was from 3-9. Each shoot on the average possessed 8-10 nodes and was fragmented into 8-10 parts having at least one node. Each node, when planted on the same medium, produced a complete shoot having the same number of nodes, in about a month. The rate of vegetative multiplication is, therefore, very rapid, as from one shoot thousands of shoots can be produced in a period of 12-13 months. Sterile shoots production and regeneration capacities in potato varieties were studied under aseptic conditions in order to have virus free potato seed.
RESULTS AND DISCUSSION
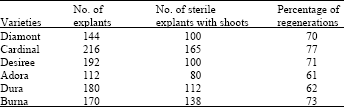
Number of sterile shoots produced and Percentage of Regenerants from the sprouts of Diamont, Cardinal, Desiree, Adora, Dura and Burna are presented in Table 1. At the end of one month, in Diamont out of 144 explants, number of sterile explants with shoots obtained was 100 and percentage of regeneration was 70. In Cardinal at the end of one month, out of 216 explants, numbers of sterile explants produced with shoots were 165, with percentage of regeneration at 77. In case of Desiree variety, out of 192 explants, numbers of sterile explants produced with shoots were 100 with percentage of regeneration at 71. Out of 112 Adora explants, numbers of sterile explants produced with shoots were 80 with percentage of regeneration at 61. In Dura at the end of one month, out of 180 explants, numbers of sterile explants produced with shoots were 112, with percentage of regeneration at 62. Out of 170 Burna explants, numbers of sterile explants produced with shoots were 138 with percentage of regeneration at 73. Number of sterile shoots production and percentage of regenerations was the highest in Cardinal (165: 77) followed by Burna (138: 73), Desiree (100: 73), Diamont (100: 70). Two cultivars, Dura (112: 62) and Adora (80: 61), exhibited the lowest regeneration capacity under the experimental conditions employed in our studies (Table 1). Cardinal exhibited the highest regeneration in our studies (Table 1). The high morphogenic and direct regeneration capacity of the cultivar Cardinal has been recently reported by Hussain et al. (2005). Diamont gave the second highest regeneration in their studies but was at fourth position in our studies (Table 1).
| Table 1: | Number of sterile shoots produced and percentage of regenerants from the nodal explants of various varieties of potato |
 | |
Our results are similar to results of Anjum and Hakoomat (2004a, b). They recorded that stem segments of 4 potato genotypes, in 2 cultivars of Solanum tuberosum (Desiree and Maris Piper) and 2 wild species (S. commersonii and S. acaule) and tuber explants of the 2 cultivars of S. tuberosum were cultured on 3 regeneration media. Among the genotypes, shoot regeneration was, in general, quickest in Maris Piper. Shoots from the stem explants of S. commersonii were only regenerated on the media of Iapichino and Ahloowalia, while the explants of S. acaule failed even to survive on either of the media. Percentage regeneration frequency and number of shoots regenerated in both stem and tuber explants were highest on the medium of Iapichino, in Maris Piper.
Sajid et al. (1986) studied meristem tips of 0.4 mm excised from Solanum tuberosum cultivars Cardinal and Desiree having undergone thermotherapy at 34 degree centigrade for 11-60 days regenerated into rooted plantlets. The effect of duration of thermotherapy on regeneration potential of meristem was insignificant (p>0.95). Thermotherapy for 60 days in cv. Cardinal resulted in 8, 35 and 46% elimination of potato viruses S, X and Y respectively and 9, 48 and 59% from cv. Desiree.
Shibli et al. (2002) subcultured potato variety Spunta on liquid MS media containing Benzyladenine (BA) and Kinetin. Significant reduction in stem and internodes length was observed by increasing BA and kinetin concentrations. Rafique (2002) studied the effect of various concentrations of BAP and sucrose on in vitro potato micro tuber induction. Explants from in vitro grown plants with 6% sucrose and 1 muM BAP showed maximum number of micro tubers. Sucrose and BAP had also significant effect on shoot and root length. MS medium supplemented with sucrose and BAP significantly induced the micro tubers in Santa, Cardinal, Diamont and Desiree.
Rabbani et al. (2001) studied the effects of different concentrations (1, 2, 3, 4 and 5 mg L-1) of GA3 [gibberellic acid] and benzylaminopurine (BAP) on the in vitro multiplication of potato cv. Desiree. Nodal fragments and stem segments were studied. The maximum shoot length (8.96 cm) was obtained when 4 mg GA3 L-1 was applied. The number of nodes was not significantly affected by any of the GA3 concentrations used in this study. The maximum number of shoots (14) was obtained when 2 mg BAP L-1 was applied.
Muhammad (1998) noted maximum number (88.75%) of buds sprouting of explants was significantly increased at the low level (0.01%) of HgCl2. Contamination in cultured tubes (79.75%) were controlled at the higher concentration (1%) of HgCl2. The chemical (HgCl2) at the same concentration (1%) also increased the number (70%) of green color plantlet. Early buds initiation (7.75 days) was achieved at 0.01% HgCl2. Chemical at 0.01% was also effective in producing higher number of leaves (13.50) and roots (8.25) per plantlet. HgCl2 at 0.01% had also given maximum length (8.72 cm) of plantlet and roots length (3.30 cm) and number (10) of nodes per plantlet.
Nasrin (2003) observed that the callus developed from both nodal and internodal segments was induced to develop shoot when subcultured on to MS containing same growth regulator formulations. MS containing 3 mg L-1 KIN with 1.5 mg L-1 NAA was the most responsive medium for shoot regeneration. Plants regenerated through callus culture after transplanting in field displayed somaclonal variation for plant height, number of leaves/plant, number of tubers/plant and tuber weight/plant. Abbasi (2001) investigated that after 6 weeks of incubation, microtubers formed were 56% in cultivar Patrones, 44% in Diamont and 50% in Desiree. Subsequently tuberization depended on the type of genotype, culture media used and temperature with 1-3 tuberiods formed per culture bottle.
It was also observed by Hussain et al. (2005) that internodal and leaf disk tissue initially underwent callus inducing phase before regeneration took place. Dobigny et al. (1996) reported regeneration from single leafy nodes at the frequency ranging from 1 to 5 shoots per fragment, after 30 days of culture and also from leaf explant which was used as one step method regeneration. Trujillo et al. (2001) obtained regeneration of plants after passing through callus phase. Direct regeneration systems successfully demonstrated in these studies has an edge over regeneration after passing through callus phase to maintain the true-to-type nature of the regenerated plantlets and to avoid the variation. Sarker and Mustafa (2002) have used three explants viz., leaf, node segments and inter nodal segments of two potato varieties. They regenerated plantlets from leaf explant which was followed by internodes and reported high regeneration frequency from internode and leaf tissue explant of potato. BAP showed better response in terms of shoot per explant, shoot length, number of nodes and leaves in potato varieties Lal Pari and Jam Alu. Similar behavior was also observed in varieties Diamont, Altamash and Cardinal. Lal Pari showed better response as compared to Jam Alu.
Philip and Hampson (1995) also noted like us that nodal explants resulted in high regeneration potential followed by shoot explants in 12 different varieties.
In potato different approaches have been adopted to obtain efficient in vitro regeneration system either from petioles with intact leaflets (Shirely et al., 2001), leaves (Sarker and Mustafa, 2002), tuber discs (Vasquez and Clarence, 2002), stem (Chang et al., 2002) and leaf disc tissues (Osusky et al., 2005). Recently, Hussain et al. (2005) observed variation among various explants in regeneration potential on different media. The highest potential was observed in nodal tissues followed by the shoot apices while the lowest level of shoot regeneration was in leaf discs and internodes in direct regeneration protocol. Sarker and Mustafa (2002) obtained highest regeneration from leaf explant followed by internodal and nodal segments. Philip and Hampson (1995) have also reported high regeneration frequency from internode and leaf tissue explant of potato.
The genotypic differences in direct regeneration system observed in the present studies are similar to many other workers (Philip and Hampson, 1995; Sarker and Mustafa, 2002; Shirley et al., 2001; Hussain et al., 2005). The variable response of different varieties was due to genetic diversity which leads to in vitro regeneration response. Hussain et al. (2005) have concluded that media choice may depend on the variety to be used. There are many advantages of the nodal tissue as a source of explant used in this study. A large number of aseptic plants can be obtained quickly and easily and plants produced may remain true to type because of direct regeneration protocol.
These studies along with our results reported in this study clearly indicated that the rate of vegetative multiplication was very rapid. From one shoot thousands of shoots could be obtained in a period ranging from 12-13 months. We intend to use this technique on a large scale for the vegetative multiplication of indigenous cultivars. This study would help greatly in the cloning of disease-free locally adapted cultivars of potato. These cultivars could be multiplied throughout the year in the laboratory and green house. The requisite number of tubers could be supplied to the farmers at an appropriate time. The disease-free tubers imported from Netherlands could be likewise multiplied. This technique could, therefore, be exploited commercially for a large scale supply of virus free potato tubers to the farmers in Pakistan and other countries in the region.
The plant material for this study was provided by the Potato Programme of National Agriculture Research Centre Islamabad, Pakistan. The facilities provided by Dr. Hamid Rashid, P.I., Agricultural Bio-technology Labs., NARC for conducting this research are thankfully acknowledged. Thanks are also due to my colleague Dr. Sajjad-ur-Rehman Chughtai and his team for help in preparation of the manuscript.