ABSTRACT
In order to identify the genotypic differences in photosynthesis and energy balance in internal energy balance in internal photosystem in maize (Zea mays L.), the photosynthetic rate (Pn) and some main parameters of chlorophyll fluorescence (Fo/Fv, qP, NPQ and Øp) were measured and the dynamic characteristics of Non-Photochemical Quenching (NPQ) were observed and analyzed during photosynthetic induction. The results showed that there was marked difference in Pn, but no difference existed between different genotypes in maize in some parameters such as Fv/Fm, qP and Øp, while apparent difference existed in the NPQ and its change characteristics, such as TNPQ, TNPQmax, RNPQ and RNPQmax, the maximum value among all tested genotypes was about 2 times the minimum one. Above all TNPQ and NPQmax were found to be significantly correlated to Pn, which meant that not only NPQ but also its dynamic characteristics such as TNPQ and NPQmax, especially TNPQ might be good index for selecting the high photosynthesis genotype in maize.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2006.709.712
URL: https://scialert.net/abstract/?doi=ajps.2006.709.712
INTRODUCTION
As a C4 crop, maize is a most efficient assimilator of CO2 under warm, intense light conditions. There was difference in leaf photosynthetic rates among genotypes (Dwyer and Tollenaar, 1989; Dwyer et al., 1989, 1991, 1992; Zhao and Meiyun, 1994). In C4 crop, CO2 fixation is closely linked to PSII activity due to minimal photorespiration was found from simultaneous CO2 exchange and fluorescence measurement (Krall and Edwards, 1992). It has been proposed that it might be possible to estimate maize leaf photosynthetic rates from chlorophyll fluorescence measurement (Krall and Edwads, 1992). The relationship between thylakoid electron transport and photosynthetic CO2 uptake in leaves of maize hybrids was studied (Hugh and Tollenaar, 1998) and a linear relationship between ETR (Electron Transport Efficiency) and AG (gross photosynthesis) was observed. Chlorophyll fluorescence kinetic and quenching analysis was also used for analysis of different inbred lines of maize (Krebs et al., 1996) and found that large differences in chlorophyll fluorescence kinetic parameter (Fv/Fm, Fv’/Fm’, QP, NPQ and Rfd) were mainly resulted from plant age and growing season, not from genotypes. However, Zhao et al. (2005) reported that genotypic difference existed in NPQ and its dynamic characteristics (TNPQ and RNPQ) in both wheat and maize, this research was mainly focused on the genotypic differences in photosynthesis, NPQ and its dynamic characteristics like TNPQ, TNPQmax, RNPQ, and RNPQmax during photosynthetic induction and exploring the relationship between dynamic characteristics of NPQ and photosynthesis at flowering stage in maize.
MATERIALS AND METHODS
Plant materials and culture condition: This experiment was conducted in a greenhouse from April to August in 1999 in Tokyo University. Four hybrids and eight inbreds of maize were studied. The four hybrids were H1 (Nongda108), H2 (Dk789), H3 (Nongda1236) and H4 (Tangkan5)and the eight inbreeds were I1 (P178), I2 (Huang C), I3 (3189), I4 (Dou18-15), I5 (CA186), I6 (Mo17), I7 (Chuang105) and I8 (502) were sown in 14.8 L pots. Each pot had only one plant. Five replications were used. Fertilizer was applied as (N: P2O5: K2O = 3: 1.5: 2). All plants were irrigated with tap water three times each week. The average day/night temperature was 28/22°C. The maximum photosynthetically active radiation was about 1600 μmol m-2 sec-1. Measurement started at flowering and the youngest fully expanded leaf was measured.
Measurement of changes in NPQ: Non-photochemical quenching was measured with the PAM-101 fluorometer (Walz, Effeltrich, Germany) using the pulse-method. The experimental protocol of Lu and Zhang (2000) was followed for the dark adaptation treatment. After the dark adaptation treatment, the leaves of the ear position in maize were measured in the laboratory.
The minimal fluorescence level (F0) with all PSII reaction centers open was measured by measuring modulated light that was sufficient (<0.1 μmol m-2 sec-1) not to induce any significant variable fluorescence. The maximum fluorescence level (Fm) with all PSII reaction centers closed was determined by a 0.8 sec saturating pulse at 8000 μmol m-2 sec-1 in dark-adapted leaves. Then, the leaves were continuously illuminated with a white Actinic Radiation (AR) at 250 μmol m-2 sec-1. Under AR, the steady-state value of fluorescence (Fs) was recorded and saturating pulse at 8000 μmol m-2 sec-1 was imposed at 30 sec intervals to determine the maximum fluorescence level in the light-adapted state (Fm’). The AR was removed and the minimum fluorescence level in the light-adapted state (Fo’) was determined by illuminating the leaf with a 3-s pulse of Far-Red light (FR). All measurements of Fo and Fo’ were performed with the measuring beam set to a frequency of 1.6 kHz, whereas all measurements of Fm and Fm’ were performed with the measuring beam automatically switching to 100 kHz during the saturating flash. Under each saturation pulse, NPQ was recovered until arriving at the steady state. The whole process of NPQ was analyzed. TNPQ, TNPQmax means the time needed to reach the stable stage and the maximum value respectively. RNPQ, RNPQmax was the ratio of NPQ to TNPQ, NPQmax to TNPQmax, respectively (Fig. 1).
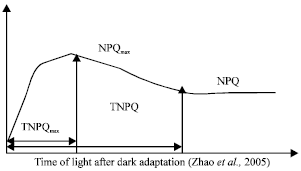 | |
| Fig. 1: | Parameters of change characteristics in NPQ: maximum NPQ (NPQmax), the time needed to reach NPQmax(TNPQmax) and the time to reach steady-state of NPQ (TNPQ) were expressed |
Measurement of photosynthetic rate: Photosynthetic rate (Pn) of the leaf ear was measured at 9:00 am with the CIRAS photosynthesis system (CIRAS-1 PP-system UK). The photosynthetically active radiation, temperature and CO2 were controlled at about 1600 μmol m-2 sec-1, 25°C and 350 ppm, respectively.
RESULT AND ANALYSIS
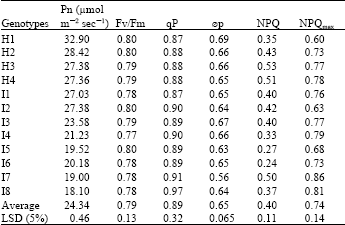
Genetic difference in photosynthetic rate (Pn ) and chlorophyll fluorescence parameters: There was large variance in photosynthetic rate (Pn) among different genotypes (Table 1). H1(ND108) showed the highest Pn (32.90 μmol m-2sec-1) and its parents I1, I2 were also higher than other inbreeds in Pn. The lowest Pn value was only 18.10 μmol m-2sec-1. The comparative analysis in some main chlorophyll fluorescence parameters showed that there was marked difference in NPQ and NPQmax, while no significant genotypic difference existed in Fv/Fm, qP and øp. The maximum value of NPQ and NPQmax was about 1.5-2 fold higher than the minimum one among twelve tested genotypes.
| Table 1: | Genotypic difference in photosynthetic rate (Pn) and some main chlorophyll fluorescence parameters |
 | |
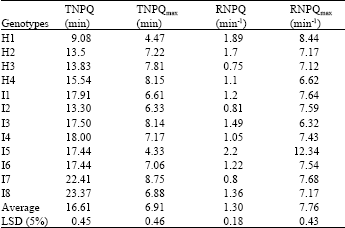
| Table 2: | Genotypic difference in some NPQ change characteristics during photosynthetic induction |
 | |
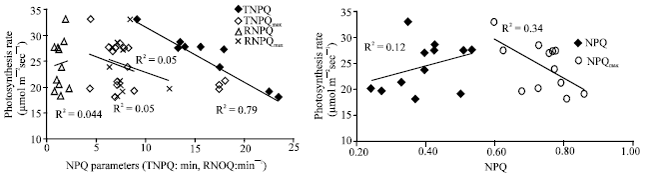 | |
| Fig. 2: | Relationship between photosynthesis rate and NPQ, NPQmax, TNPQ, TNPQmax, RNPQ, RNPQmax, based on data from twelve tested genotypes |
So the maximal efficiency of PSII to photochemistry (Fv/Fm) was relatively stable between genotypes in maize, compared with photosynthesis and non-photochemical quenching coefficient.
Differences in dynamic characteristics of NPQ during photosynthetic induction between genotypes in maize: In order to identify the difference of dynamic characteristics of NPQ during photosynthetic induction between genotypes in maize, some parameters of NPQ defined in our former research were referred here (Zhao et al., 2005),TNPQ, TNPQmax and RNPQ, RNPQmax, which meant the time and rate to reach the stable, maximum value of NPQ, respectively. The result showed there was much difference between genotypes in dynamic characteristics of NPQ and the difference between the maximum and minimum value of TNPQ, TNPQmax, RNPQ and RNPQmax among genotypes was with about 2 factors (Table 2). Among those tested genotypes, H1(Nongda108) with highest photosynthesis took shortest time (TNPQ and TNPQmax)to reach the stable or maximum NPQ stage, on the contrary, some inbreeds like I7(Chuang105) and I8 (502) showed longest both TNPQ and TNPQmax, so there was difference between genotype in the process of the balance in NPQ.
The correlation analysis of dynamic characteristic of NPQ with photosynthesis: The correlation analysis showed that Pn was positively correlated with the quantum yield of PSII electron transport (øp) and TNPQ and NPQmax were both negatively and closely related to photosynthetic rate (Pn) (R2 = 0.79**, 0.34*, respectively) (Fig. 2), so the time of NPQ to reach the stability stage and peak value of NPQ might be related to photosynthesis in maize and the genotype with higher photosynthesis usually performed shorter TNPQ and lower NPQmax after dark adaptation when illumination pulse on. However, there was no significant correlation between Pn and NPQ, TNPQmax, RNPQ, RNPQmax, which meant that different genotypes in maize might have different feature in energy transfer and balance between photosystem. So TNPQ and NPQmax, especially TNPQ might be good index for selecting the high photosynthesis genotype in maize.
DISCUSSION
Chlorophyll fluorescence has also been commonly used for comparing among genotypes in maize (Larroque and Planchon, 1990; McCullough et al., 1994; Krebs et al., 1996; Janda, 1998; Hugh and Tollenaar, 1998). In this study, the results also showed that there was marked difference in NPQ and NPQmax between genotypes in maize. And the maximum value of NPQ and NPQmax were about 1.5-2 fold higher than the minimum one among those twelve tested genotypes. Comparison of dynamic characteristics of NPQ including TNPQ, TNPQmax and RNPQ, RNPQmax showed there was much difference between genotypes in TNPQ and RNPQ, which implied that the characheristics of NPQ change was more important to analyze energy transfer and balance between photosystems among different genotypes in maize.
Chlorophyll fluorescence could be used to study components of the photosynthetic apparatus and their reactions to changes in the environment as well as photosynthesis as a whole (Fracheboud et al., 1998, 1999; Edwards and Baker, 1993). Horton et al. (1996) also reported chlorophyll fluorescence could be used as the basis of a fair comparison of leaf photosynthetic rates among different maize cultivars. In this study, in maize, TNPQ and NPQmax were found to be closely and negatively correlated with Pn, the genotype with higher photosynthesis usually performed shorter TNPQ and lower NPQmax after dark adaptation when illumination pulse on. In our former research, the NPQmax and the time to reach the steady-state primarily depended on the formation of qE when illumination occurred after dark adaptation. In other words, the genotypes with higher NPQmax and longer time to reach the steady-state need more energy to maintain the proton gradient of transthylakoid to adapt to strong radiation after dark adaptation, so that photosynthesis rate was lower. So TNPQ and NPQmax might be used to select the high photosynthesis genotype in maize. However this is inconsistent with what existed between different crops, in our former research (Zhao et al., 2005), compared with wheat, maize as a C4 crop, although showed a higher NPQmax and took a longer time to reach the peak (TNPQmax), photosynthesis rate was still higher, so the mechanisms behind this is still to be further explored and explained.
REFERENCES
- Dwyer, L.M., D.W. Stewart, L. Balchin D. Hoing, C.J. Marur and R.I. Hamilton, 1989. Potoaynthetic rates of six maize cultivars during development. Agron. J., 81: 597-602.
Direct Link - Dwyer, L.M., D.W. Stewart and M. Tollenaar, 1991. Changes in plant density dependence of leaf photosynthesis of maize (Zea mays L.) hybrids, 1959 to 1988. Can. J. Plant Sci., 71: 1-11.
CrossRefDirect Link - Edwards, G.E. and N.R. Baker, 1993. Can CO2 assimilation in maize leaves be predicted accurately from chlorophyll fluorescence analysis? Photosyn. Res., 37: 89-102.
Direct Link - Fracheboud, Y., J. Leipner and P. Stamp, 1998. Selection for Cold Tolerance in Maize Using Chlorophyll Fluorescence. In: Crop Development for the Cool and Wet Regions of Europe, Sowinski, P., B. Zagdanska, A. Aniol and K. Pithan (Eds.). Official Publications of the European Communities, Brussels, pp: 116-121.
- Fracheboud, Y., P. Haldimann, J. Leipner and P. Stamp, 1999. Chlorophyll fluorescence as a selection tool for cold tolerance of photosynthesis in maize (Zea mays L.). J. Exp. Bot., 50: 1533-1540.
Direct Link - Hugh, J.E. and M. Tollenaar, 1998. Relatiomshio between thylakoid electron transport and photosynthetic CO2 uptake in leaves of three maize hybrids. Photosyn. Res., 58: 245-257.
Direct Link - Horton, P., A.V. Ruban and R.G. Walters, 1996. Regulation of light harvesting in green plants. Annu. Rev. Plant Physiol. Plant Mol. Biol., 47: 665-684.
Direct Link - Janda, T., G. Szalai, J.M. Ducruet and E. Paldi, 1998. Changes in photosynthesis in inbred with different degrees of chilling tolerance griwn a optimum ad subopmim temperatures. Photosynthetica, 35: 205-212.
Direct Link - Krall, J.P., G.E. Edwards, 1992. Relationship between photosynstemII activity and CO2 fixation in leaves. Physiol. Plant., 86: 180-187.
Direct Link - Krebs, D., H. Synkova, N. Avratovscukova, M. Kocova and N. Sestak, 1996. Chlorophyll fluoresence measurements for genetic analysis of maize cultivars. Photosynthetica, 32: 695-708.
Direct Link - Larroque, C.M. and C.L. Planchon, 1990. Lignification and physiological factors of yield in maize. Crop Sci., 30: 1105-1109.
Direct Link - Lu, C.M. and J.H. Zhang, 2000. Photosystem II photochemistry and its sensitivity to stress in maiz plants as effected by nitrogen deficiency. J. Plant Physiol., 157: 124-130.
Direct Link - McCullough, D.E., P.H. Cirardin, M. Mihajlovic, A. Aguilera and M. Tollenaar, 1994. Influence of N supply on development and dry matter accumulation of an old and a new maize hybrid. Can. J. Plant Sci., 74: 471-477.
Direct Link - Zhao, M., W.D. Jiang-Zai-Song, R. Ishhill and C. Li, 2005. Differences between maize and wheat for non-photochemical quenching (NPQ) during photosynthetic induction. Acta Agronomica Sinica, 31: 1544-1551.
Direct Link








