ABSTRACT
This study focuses on the comparative effects of copper and cadmium on the organic acid content and exudation in maize (Zea mays L.). We have also discussed the role of organic acids in copper and cadmium tolerance. The maize seedlings were grown in hydroponics for 8 days and then treated with 100 μM copper or cadmium for 4 days. The copper reduced more than cadmium root fresh wt and shoot dry wt, while Cd had more decreasing effect on root dry wt. Analysis of organic acids in root exudates showed that citrate exudation was not affected by both metals, suggesting that root exudation was not involved in copper or cadmium tolerance in maize. This prompted us to investigate the role of organic acids in internal tolerance mechanism. Total organic acids were decreased in roots but remained unchanged in shoots of Cu and Cd-treated plants. However, malate content decreased substantially in roots by 68 and 53%, respectively with Cu and Cd. The marked increase in the content of citrate with both metals in roots, as well as in those of malate with Cu and citrate with Cd in shoots, underlined the main role of these organic acids in Cu and Cd tolerance in maize.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2006.598.606
URL: https://scialert.net/abstract/?doi=ajps.2006.598.606
INTRODUCTION
In plants, a number of heavy metals such as zinc and copper are essential for normal growth as cofactors and as structural and catalytic components of proteins and enzymes. However, these micronutrients, as well as nonessential heavy metals such as cadmium, become stressful at high levels in the plant’s environment (Tomsett and Thurman, 1988). Heavy metals contamination of the environment can occur in some natural soils or as the result of environmental pollution from mining, smelting, manufacturing, agricultural or waste disposal technologies (Foy et al., 1978). These toxic pollutants affect plant growth at least by inducing the oxidation of molecules catalyzed by metals. Copper has an irreplaceable role as nutritional element in plants and as redox component required for a wide variety of processes including the detoxification of free radicals, lignification of plant cell walls, photosynthesis and electron transfer reactions of respiration (Fox and Guerinot, 1998). It is a cofactor of numerous metallo-enzymes such as cytochrome c oxidase, Cu, Zn-superoxide dismutase (SOD) and several ATPases implied in catalyzing redox reactions (Gupta, 1979). Excess of copper can cause oxidative damage by reacting with oxygen and producing toxic-free radicals (Fenton reaction). Moreover, copper inhibited numerous enzymes implied in the dark reaction of photosynthesis, particularly Rubisco (Stiborova et al., 1986a). Cadmium a non-essential element is strongly phytotoxic (Clijsters and van Assche, 1985; Sanità di Toppi and Gabbrielli, 1999). In the environment, cadmium is spilled extensively, present by the powerhouses, the heating systems, the metallurgic industries, the incinerators of waste, the urban traffic, the cement industrial unit and present as impurity in the phosphate fertilizers (Sanità di Toppi and Gabbrielli, 1999). Cadmium can cause an increase in lipid peroxidation and lipoxygenase activity (Gallego et al., 1996). Excess cadmium has been also shown to enhance leaf senescence (Gallego et al., 1996). Plants respond to heavy metal stress in a variety of ways, by exclusion mechanism avoiding the toxic effects of the metal, or by internal tolerance mechanisms enabling the plant to survive (Tomsett and Thurman, 1988). Generally, tolerance mechanisms in plants can be divided into (1) metal binding to cell wall, (2) reduced transport across the cell membrane, (3) active efflux, (4) compartmentalization and, (5) chelation of the metal by proteins, organic acids or inorganic compounds (Tomsett and Thurman, 1988). The principle classes of intracellular metal-binding ligands are organic acids, amino acids, phytochelatins (PCs) and metallothioneins (MTs). Organic acids, such as malate and citrate, can form complexes with metals in the vacuole (Godbold et al., 1984). Nevertheless, few recent data are available about the role of organic acids in copper and cadmium tolerance. This research was a comparative study on the effects of copper and cadmium on organic acid content and exudation in maize (Zea mays L). The role of organic acids in copper and cadmium tolerance was discussed. Metal effects on the growth and metal accumulation will be presented.
MATERIALS AND METHODS
Growth conditions and metal analysis: Maize (Zea mays L.; var. LG 23/01) seeds were surface sterilized with 10% (v/v) H2O2 for 20 min, rinsed many times with tap water and germinated on filter paper moistened with distilled water in the dark, at 25°C for 3 day. The germinated seedlings were transferred to 12l basal nutrient solutions for 4 days. Selected plants of uniform size were selected and then transferred to identical solutions in 6l plastic pots (12 plants each) for 4 days. For treatment purposes, eight-day-old seedlings were transferred to solutions that contain 0 (control), 100 μM CuSO4 or Cd(NO3)2 for 4 days. Plants were grown hydroponically according to Chaffai et al. (2005). At harvest, the roots were rinsed three times with distilled water; the fresh wt was determined and frozen in liquid N2. The metal content was determined in roots and shoots and in two fractions: root cell wall and soluble fractions. The root cell wall was isolated by 1% (v/v) Triton X-100 as previously described (Chaffai et al., 2005). The metal content was determined by an atomic absorption spectrophotometer (Perkin Elmer-model 2380).
Organic acid extraction and HPLC analysis: Preparation of organic acid extracts of plant tissue and their subsequent separation by high performance liquid chromatography (HPLC) on a Dupont Zorbax SAX-5 μm column (250x4.6 mm, HP) was according to the procedure described previously by Chaffai and El Ferjani (2005).
Determination of MDH activity: Frozen material was homogenized in 20 mM Tris-HCl buffer (pH 8.0) containing 5 mM ascorbate. The homogenate was centrifuged at 25000 g for 20 min (4°C). The malate dehydrogenase (MDH, EC 1.1.1.37) activity was determined in protein extracts as previously described (Chaffai and El Ferjani, 2005). The protein was determined by the Bradford (1976) method using BSA as a standard.
RESULTS
Effects of Cu and Cd on growth: After exposure to 100 μM Cu or Cd for 4 days, maize plants showed a significant decrease in root and shoot growth. The root fresh wt was decreased by 54.5 and 32.5% in the presence respectively of excess Cu and Cd in solution; however, shoot fresh wt was similarly decreased by 31.2% (Fig. 1A and B). The Cd decreased by 42.7% and Cu by 23.6% the root dry wt. However, Cu and Cd decreased the shoot dry wt, respectively by 49 and 24.5% (Fig. 1C and D).
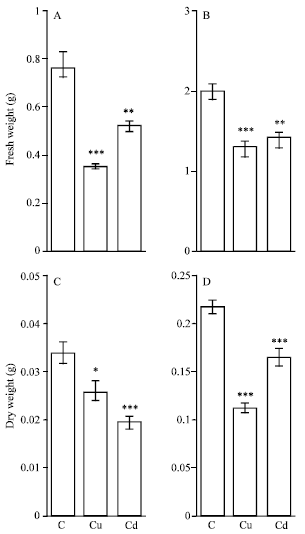 | |
| Fig. 1: | Effects of copper and cadmium on root (A, C) and shoot (B, D) fresh and dry weight of maize (Zea mays L.). The seedlings were grown in nutrient solutions containing 100 μM Cu or Cd for 4 days. Histograms represent means±SE (n = 6) of six separate experiments. One Way ANOVA is performed for comparison between control and treated plants. Asterisks show statically different means between control and treated plants: *, p<0.05; **, p<0.01;***, p<0.001 |
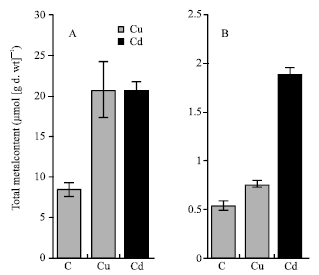 | |
| Fig. 2: | Total content of copper and cadmium in roots (A) and shoots (B) of maize (Zea mays L.). The seedlings were grown in nutrient solutions at 100 μM Cu or Cd for 4 days. Histograms represent means±SE (n = 4) of four separate experiments |
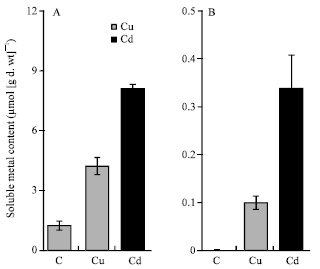 | |
| Fig. 3: | Soluble fractions of copper and cadmium in roots (A) and shoots (B) of maize (Zea mays L.). The seedlings were grown in nutrient solutions at 100 μM Cu or Cd for 4 days. Histograms represent means± E (n = 4) of four separate experiments |
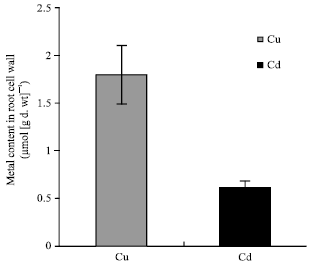 | |
| Fig. 4: | Cell wall fractions of copper and cadmium in roots of Cu and Cd-treated plants. The seedlings were grown in nutrient solutions at 100 μM Cu or Cd for 4 days. Histograms represent means±SE (n = 6) of six separate experiments. |
Cu and Cd content: The results showed that Cu concentrations in roots of maize plants grown at 100 μM Cu were 27-fold higher than those in shoots; however, Cd concentrations were 11-fold higher (Fig. 2). The total concentrations of Cd in roots of Cd-treated plants were similar to those of Cu in roots of Cu-treated ones (Fig. 2A). In contrast, the Cd concentrations in shoots were 2.5-fold higher than Cu in shoots of Cu-treated plants (Fig. 2B). The soluble fraction of both metals was greater in roots than in shoots (Fig. 3). The soluble fraction of Cu was 41.5-fold greater in roots than in shoots; however that of Cd was 23.7-fold greater (Fig. 3). The soluble fraction of Cu represented 20.1% of total Cu content and that of Cd 37.2% of total Cd content of maize treated plants. With regard to cell wall fraction, the Cu in root cell walls in the presence of 100 μM Cu, was 3-fold greater than Cd in root cell walls in the presence of 100 μM Cd. This fraction represented 8.3 and 2.7% of the total metal content of roots treated with Cu and Cd respectively (Fig. 4).
| Table 1: | Effects of Cu and Cd on organic acid concentrations in roots of maize (Zea mays L.). The seedlings were grown in nutrient solutions containing 100 μM CuSO4 or Cd(NO3)2 for 4 days |
 | |
| Values represent means±SE (n = 5) of five separate experiments. One way ANOVA is performed for comparison between control and treated plants. Different letters within the same column indicate significant differences. Asterisks show statically different means between control and treated plants: *, p<0.05; **, p<0.01; ***, p<0.001. C, control | |
| Table 2: | Effects of Cu and Cd on organic acid concentrations in maize (Zea mays L.) shoots. The seedlings were grown in nutrient solutions containing 100 μM CuSO4 or Cd(NO3)2 for 4 days |
 | |
| Values represent means±SE (n = 5) of five separate experiments. One way ANOVA is performed for comparison between control and treated plants. Different letters within the same column indicate significant differences. Asterisks show statically different means between control and treated plants: *, p<0.05; **, p<0.01; ***, p<0.001. C, control; nd, not detected | |
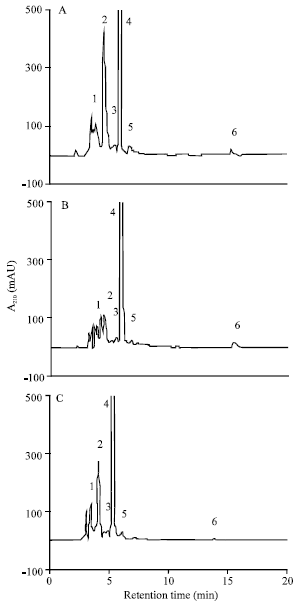 | |
| Fig. 5: | HPLC chromatograms of organic acids in root extracts of maize (Zea mays L.). The seedlings were grown in nutrient solutions containing 0 (A); 100 μM Cu (B) or Cd (C) for 4 days. The organic acids were separated on a 250x4.6 mm i.d. HP Zorbax SAX, a strong anion exchange column. The mobile phase consisted of 0.2 M KH2PO4 (pH 3.0) at a flow rate of 1 mL min-1 and at 25°C. The elution peaks: 1, succinate; 2, malate; 3, citrate; 4, tartarate; 5, lactate; 6, unknown |
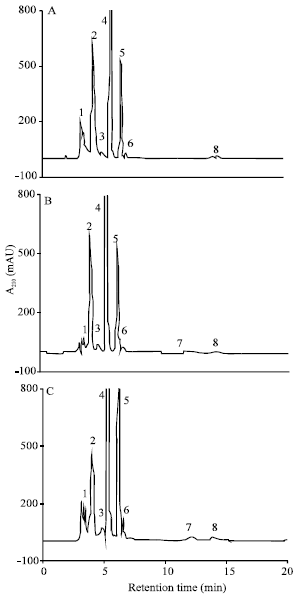 | |
| Fig. 6: | HPLC chromatograms of organic acids in shoot extracts of maize (Zea mays L.). The seedlings were grown in nutrient solutions containing 0 (A); 100 μM Cu (B) or 100 μM Cd (C) for 4 days. The organic acids were separated on a 250x4.6 mm i.d. HP Zorbax SAX, a strong anion exchange column. The mobile phase consisted of 0.2 M KH2PO4 (pH 3.0) at a flow rate of 1 mL min-1 and at 25°C. The elution peaks: 1, succinate; 2, malate; 3, citrate; 4, tartarate; 5, lactate; 6, unknown; 7, salicylate; 8, unknown |
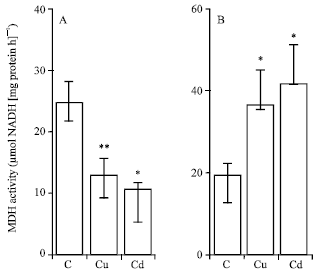 | |
| Fig. 7: | Effects of copper and cadmium treatment on MDH activity in roots (A) and shoots (B) of maize (Zea mays L.). The seedlings were treated with 100 μM Cu or Cd for 4 days. Histograms are means±SE (n = 6) of six independent experiments. Asterisks show statically different means between control and treated plants: *, p<0.05; **, p<0.01 |
Organic acid content in roots: Five organic acids in root extract from control and Cu and Cd-treated plants were identified by comparing their retention time (Rt) with those of known standards. Organic acids found were succinate (peak 1, Rt 3.80), malate (peak 2, Rt 4.08), citrate (peak 3, Rt 4.57), tartarate (peak 4, Rt 5.31) and lactate (peak 5, Rt 6.11). A non identified peak (peak 6) was detected with Rt of 13.92 min (Fig. 5). The organic acid analysis showed that total organic acids in roots decreased significantly with both metals (Table 1). The decrease was to 31.4 and 47.8%, respectively with Cu and Cd. Both metals decreased markedly the malate content by 67.7 and 52.7%, respectively with Cu and Cd. In the same way, the tartarate content decreased significantly by 22.6 and 49.8% with Cu and Cd (Table 1). However, citrate content increased substantially by 52.9 and 55.5% respectively with Cu and Cd. The succinate content showed a lesser increase only by Cd. Moreover, lactate content of the roots did not change under Cu or Cd treatment (Table 1).
Organic acid content in shoots: In the chromatograms of organic acids in shoot extracts, 6 peaks were identified corresponding to succinate (peak 1, Rt 3.52), malate (peak 2, Rt 3.98), citrate (peak 3, Rt 4.63), tartarate (peak 4, Rt 5.36), lactate (peak 5, Rt 6.19) and salicylate (peak 7, Rt 12.19). In addition to these peaks, the chromatograms revealed two other unidentified peaks with Rt 6.64 (peak 6) and Rt 14.08 (peak 8), respectively (Fig. 6). Exposure to 100 μM Cu or Cd had no significant effect on total organic acid content in shoots (Table 2).
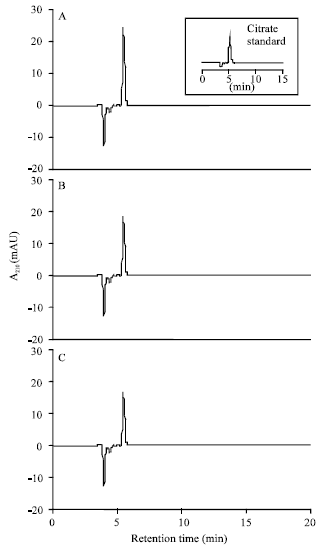 | |
| Fig. 8: | HPLC chromatograms of organic acids in root exudates of maize (Zea mays L.). The seedlings were grown in nutrient solutions containing 0 (A); 100 μM Cu (B) or Cd (C) for 4 days. The organic acids were separated on a 250x4.6 mm i.d. HP Zorbax SAX, a strong anion exchange column, using 0.2 M KH2PO4 (pH 3.0) as mobile phase at a flow rate of 1 mL min-1 and at 25°C |
Similarly, the succinate and the lactate content were not affected with Cu and Cd treatments. However, the tartarate content was decreased with both metals (Table 2). By contrast, the malate and citrate content increased 1.9-fold, respectively with Cu and Cd (Table 2). In addition, salicylic acid was detected in shoots of treated plants but not in control plants (Table 2).
MDH activity: A marked decrease in MDH activity was observed in roots of Cu and Cd-treated plants (Fig. 7A). The activity of this enzyme was deceased by 47.8 and 56.9%, respectively with Cu and Cd.
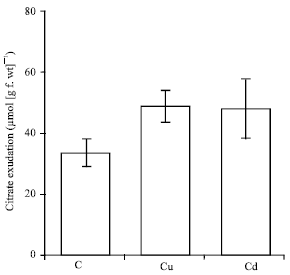 | |
| Fig. 9: | Effects of copper and cadmium treatments on citrate exudation. The maize (Zea mays L.) seedlings were treated with 100 μM Cu or Cd for 4 days. The root exudates were collected and then analyzed by HPLC. Histograms are means±SE (n = 3) of three independent experiments |
By contrast, this activity increased 1.9 and 2.1-fold, respectively in Cu and Cd-treated shoots (Fig. 7B).
Organic acid exudation: Organic acid profiles of root exudates showed only one peak with Rt corresponding to citrate (Fig. 8). Both Cu and Cd did not affect the pattern of citrate exudation (Fig. 9).
DISCUSSION
Effects of Cu and Cd on growth and on Cu and Cd content: The result of the current study indicated that root fresh and shoot dry wt was more affected by Cu than by Cd (Figs. 1A and D). There have been a number of studies that reported that Cu inhibited the growth even at low concentrations. Cu produced 50% growth inhibition of Lolium perenne root at copper concentrations not exceeded 0.02 ppm. In Zea mays, growth was inhibited by 0.06 ppm (Stiborova et al., 1986b). In the same way, 0.6 ppm inhibited root and shoot growth in Oryza sativa (Gupta and Mukherji, 1977). Enhanced susceptibility to Cu observed in roots of maize plants after Cu treatment could be related to a high accumulation of Cu in the roots and/or limited translocation of the metal to the aerial part of plants (Stiborova et al., 1986b). Cathala and Salsac (1975) have reported that the roots have a tendency to accumulate higher amounts of Cu than the aboveground of plants as found in our study (Fig. 2A). Preferential accumulation of metals in roots could be a strategy for the plants to limit metal translocation, thereby avoiding metal toxicity as well for Cu (Ouzounidou et al., 1992) as for Cd (Jarvis et al., 1976). Many species accumulated Cu in major part in roots: Lolium perenne (Jarvis and Whitehead, 1981), Mimulus guttatus (Macnair, 1981), Spinacia oleracea (Baszy½ski et al., 1982) and Avena sativa (Tukendorf and Baszy½ski, 1985). Restricted translocation of Cu from roots to shoots is earlier reported in Agrostis stolonifera (Wu et al., 1975). The pronounced effect of Cu on shoot growth (Fig. 1D) as compared to Cd could be attributed to more susceptibility of photosynthetic apparatus to Cu. There is evidence that Cu should be excluded from the leaves because of its inhibitory effect on photosynthesis. It has been argued that Cu is a strong inhibitor of photosynthesis in algae and higher plants. Cu-induced inhibition of photosynthetic electron chain transport by acting on the oxidant sites of PSII and the reductant sites of PSI, concomitant with inhibition of photophosphorylation and dark reactions (Shioi et al., 1978a,b). Furthermore, Sandmann and Böger (1980a,b) have reported that Cu+ and Cu2+ ions blocked photosynthetic electron transport in Scenedesmus and in isolated chloroplasts of spinach plants. Similarly, exposure of maize plants to Cd causes many toxic effects. In wheat, 20 μM Cd decreased to the half the fresh wt of the roots. A similar reduction of fresh wt of the shoots was observed when 50 to 70 μM Cd were applied (Trivedi and Erdei, 1992). In this study, the relative larger amount of Cd in shoots than that of Cu (Fig. 2B) may indicate higher mobility of Cd. Salt et al. (1995) have suggested that the translocation of Cd from roots to shoots was likely to occur via xylem and to be driven by transpiration from the leaves. This is founded from the reduced Cd accumulation in shoots after ABA-induced stomatal closure of Indian mustard. The low levels of both metals in the cell wall fraction of roots compared to the soluble fractions of these metals in treated plants (Fig. 3A and 4), suggests that root cell wall couldn’t be the best site for metal accumulation.
Role of organic acids in Cu and Cd tolerance: We have shown that citrate exuded in response of Cu and Cd did not differ from that from roots of control plants (Fig. 8 and 9). This result suggests that external metal detoxification was not involved in Cu and Cd tolerance. Indeed, the root secretion of organic acid was reported to be metal-specific response. Previous studies have suggested that root organic acid exudation was specifically induced by Al than by other heavy metals such as Cu and Cd, or by other environmental stresses. Indeed, the exclusion of Al by chelation with organic acid anions excreted from root apices has been suggested to be the basis of Al tolerance mechanism in many species. Thus, under Al stress, malate was released from the roots of Al-tolerant cultivars of wheat (Triticum aestivum) (Delhaize et al., 1993); citrate from Al-tolerant cultivars of snapbean (Phaseolus vulgaris) (Miyasaka et al., 1991); maize (Zea mays) (Chaffai and El Ferjani, 2005) and Cassia tora (Ma et al., 1997c); and oxalate from buckwheat (Fagopyrum esculentum) (Ma et al., 1997b) and taro (Colocasia esculenta) (Ma and Miyasaka, 1998). In contrast, phosphorus (P) deficiency, 50 μM lanthanum (La3+), 200 μM manganese (Mn2+), 10 μM cadmium (Cd2+), or 50 μM lead (Pb2+) were not able to induce the exudation of organic acids in rye (Li et al., 2002). Similarly, neither P deficiency nor application of La or ytterbium (Yb) induced secretion of organic acids in Casia tora or buckwheat (Ma et al., 1997b,c; Zheng et al., 1998). Delhaize et al. (1993) have also reported that La was not able to induce secretion of malate in Al-resistant cultivars of wheat. The observed difference in organic acid exudation between Cu and Cd-treated and Al-treated plants suggested a specific role for the Al-induced activation of protein channel mediating citrate efflux. Indeed, patch-clamp studies have demonstrated that Al activated an Al-dependant anion channels in intact maize root apices, particularly sensible to Al (Piñeros and Kochian, 2001). Therefore, there is evidence that a highly effective resistance mechanisms based on internal than external tolerance mechanisms appeared to operate in the case of Cu and Cd. The internal tolerance mechanism involved metal chelation or compartmentalization of the ligand-metal complex. Evidence for a potential role of metal chelating substances such as organic acids in heavy metal tolerance was particularly demonstrated in metal accumulating species. Organic acids were reported to form strong complexes with Al ions and were implicated in internal Al detoxification in Al-accumulating species. For example, in Hydrangea, an Al accumulator plant, the formation of an Al-citrate 1:1 complex in leaves has been proposed as an Al tolerance mechanism (Ma et al., 1997a). Also in the Al hyperaccumulator Melastoma malabathricum Al seems to be transported in the form of citrate, while oxalate would be the storage form in leaf vacuoles (Watanabe et al., 2000). Moreover, organic acids have been found to accumulate to higher concentrations in roots and leaves of metal tolerant plants (Kishinami and Widholm, 1987). We examined the organic content and found that roots accumulated high levels of citrate in roots of Cu and Cd treated plants (Table 1) and shoots accumulated malate in Cu-treated plants and citrate in Cd-treated ones (Table 1 and 2) suggesting that these organic acids may play a role in metal detoxification. Increases of organic acid content (particularly malate and citrate) in Cu2+ and Cd2+ stressed plants, observed in the present study have been correlated with increased tolerance to heavy metals, such as Al, Cu, Zn, Ni... Organic acid accumulation has been correlated with metal resistance in selected Al resistant carrot cells, where increased citrate concentrations were observed (Ojima et al., 1984). Mathys (1977) has reported that Zn tolerance was associated with malate accumulation. Following the Zn tolerance model proposed by Mathys (1977), Zn was chelated by malate in the cytoplasm, transported to the vacuoles and accumulated there. The accumulation of citrate and malate has been reported in Zn-tolerant grasses of Deschampsia caespitosa grown in the presence of Zn (Godbold et al., 1984; Thurman and Rankin, 1982). In these species, the Zn tolerance was correlated with citrate levels. Similarly, in Ni accumulating plants of 17 species, Ni accumulation was correlated with high levels of citrate in their leaves (Lee et al., 1978). In cultured cells of Nicotiana plumbaginifolia, both the Cu and Zn resistant cells accumulated higher levels of citrate and malate than the wild type cells in the presence of the metals (Kishinami and Widholm, 1987). The tolerance to Cu or Zn of Nicotiana plumbaginifolia cells was in part due to the ability to accumulate higher than normal levels of citrate and malate to chelate the toxic metal ions. Thus, citrate and malate content were increased 3 and 12-fold, respectively in the metal resistant cells grown in the presence of Cu or Zn compared to wild cells gown in the absence of Cu or Zn. It has been also argued that organic acids have proposed roles in the heavy metal storage and transport in plants. Strong argument is the observation that heavy metal ions would be largely present in various forms as organic acid complexes. In buckwheat, Al in both roots and leaves is found to be bound into an Al-oxalate 1:3 complex (Ma, 2000), while in leaves with high Al concentrations, an Al-citrate complex was also identified in this specie (Shen et al., 2004). The marked decrease in malate content in roots with metals was correlated to the decrease in MDH activity (Fig. 7A). We have also showed that the MDH activity was significantly stimulated by both metals in shoots (Fig. 7B). This is consistent with Lee et al. (1976) study, where an increase in MDH activity at 0.90 and 1.35 μM Cd2+ in leaf blades of soybean seedlings, 10 days after Cd treatment was observed. The salicylic acid which was only detected in shoots of Cu and Cd treated plants (Fig. 6 and Table 2) could be a part of a general stress response. The salicylic acid is essential in regulating the growth, the development and the microorganism-plant interactions and in Systemic Acquired Resistance (SAR) in plants (Hahlbrock and Scheel, 1989). It would be interesting to extend the study to other maize cultivars in order to correlate the tolerance with organic acid changes.
ACKNOWLEDGMENTS
We thank Dr. Sami Ben Jamaa for reading, reviewing and checking the English of the manuscript and Mrs. Najeh Galai for her great help and assistance.
REFERENCES
- Baszyski, T., M. Krol, Z. Krupa, M. Ruszkowska, U. Wojcieska and D. Wolinska, 1982. Photosynthetic apparatus of spinach exposed to excess copper. J. Plant Physiol., 108: 385-395.
Direct Link - Bradford, M.M., 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem., 72: 248-254.
CrossRefPubMedDirect Link - Cathala, N. and L, Salsac, 1975. Absorption of copper by roots of maize (Zea mays L.) and sunflower (Helianthus annuus L.). Plant Soil, 42: 65-83.
Direct Link - Chaffai, R. and E.El-Ferjani, 2005. Alteration of the profile of organic acid content and exudation under aluminum stress in maize (Zea mays L.). Int. J. Bot., 1: 64-73.
CrossRefDirect Link - Chaffai, R., T. Ali and E. El Ferjani, 2005. Comparative effects of copper and cadmium on growth and lipid content in maize seedlings (Zea mays L.). Pak. J. Biol. Sci., 8: 649-655.
CrossRefDirect Link - Clijsters, H. and F. Van-Assche, 1985. Inhibition of photosynthetic by heavy metals. Photosynthesis Res., 7: 31-40.
Direct Link - Delhaize, E., P.R. Ryan and P.J. Randall, 1993. Aluminum tolerance in wheat (Triticum aestivum L.) (II. Aluminum-stimulated excretion of malic acid from root apices). Plant Physiol., 103: 695-702.
Direct Link - Fox, T.C. and M.L. Guerinot, 1998. Molecular biology of cation transport in plants. Ann. Rev. Plant Physiol. Plant Mol. Biol., 49: 669-696.
Direct Link - Foy, C.D., R.L. Chaney and M.C. White, 1978. The physiology of metal toxicity in plants. Ann. Rev. Plant Physiol., 29: 511-566.
CrossRefDirect Link - Gallego, S.M., M.P. Benavides and M.L. Tomaro, 1996. Effect of heavy metal ion excess on sunflower leaves: Evidence for involvement of oxidative stress. Plant Sci., 121: 151-159.
CrossRefDirect Link - Godbold, D.L., W.J. Horst, J.C. Collins, D.A. Thurman and H. Marschner, 1984. Accumulation of zinc and organic acids in roots of zinc tolerant and non-tolerant ecotypes of Deschampsia caespitosa. J. Plant Physiol., 116: 59-69.
CrossRefDirect Link - Hahlbrock, K. and D. Scheel, 1989. Physiology and molecular biology of phenylpropanoid metabolism. Annu. Rev. Plant Physiol. Plant Mol. Biol., 40: 347-369.
CrossRefDirect Link - Jarvis, S.C., L.H.P. Jones and M.J. Hopper, 1976. Cadmium uptake from solution by plants and its transport from root to shoots. Plant Soil, 44: 179-191.
Direct Link - Kishinami, I. and J.M. Widholm, 1987. Characterization of Cu and Zn resistant Nicotiana plumbaginifolia suspension cultures. Plant Cell Physiol., 28: 203-210.
Direct Link - Lee, K.C., B.A. Cunningham, G.M. Paulsen, G.H. Liang and R.B. Moore, 1976. Effects of cadmium on respiration rate and activities of several enzymes in soybean seedlings. Physiol. Plant., 36: 4-6.
CrossRefDirect Link - Lee, J., R.D. Reeves, R.R. Brooks and T. Jeffre, 1987. The relation between Nickel and citric acid in some Nickel accumulating plants. Photochemistry, 17: 1033-1035.
Direct Link - Li, X.F., J.F. Ma and H. Matsumoto, 2002. Aluminum-induced secretion of both citrate and malate in rye. Plant Soil, 242: 235-243.
Direct Link - Ma, J.F., S. Hiradate, K. Nomoto, T. Iwashita and H. Matsumoto, 1997. Internal detoxification mechanism of Al in Hydrangea. Identification of Al form in the leaves. Plant Physiol., 113: 1033-1039.
Direct Link - Ma, J.F., S.J. Zheng, H. Matsumoto and S. Hiradate, 1997. Detoxifying aluminium with buckwheat. Nature, 390: 569-570.
CrossRefDirect Link - Ma, J.F., S.J. Zheng, H. Matsumoto and S. Hiradate, 1997. Detoxifying aluminium with buckwheat. Nature, 390: 569-570.
CrossRefDirect Link - Ma, Z. and S.C. Miyasaka, 1998. Oxalate exudation by taro in response to Al. Plant Physiol., 118: 861-865.
Direct Link - Ma, J.F., 2000. Role of organic acids in detoxification of aluminum in higher plants. Plant Cell Physiol., 41: 383-390.
PubMedDirect Link - Mathys, W., 1977. The role of malate, oxalate and mustard oil glucosides in the evolution of zinc resistance in herbage plants. Physiol. Plant., 40: 130-136.
Direct Link - Miyasaka, S.C., J.G. Buta, R.K. Howell and C.D. Foy, 1991. Mechanism of aluminum tolerance in snapbeans root exudation of citric acid. Plant Physiol., 96: 737-743.
Direct Link - Ojima, K., H. Abe and K. Ohira, 1984. Release of citric acid into the medium by Al-tolerant carrot cells. Plant Cell Physiol., 25: 855-858.
Direct Link - Ouzounidou, G., E.P. Eleftheriou and S. Karatagfis, 1992. Ecophysiological and ultrastructural effects of copper in Thlaspi ochroleucum (Cruciferae). Can. J. Bot., 70: 947-957.
Direct Link - Pineros, M.A. and L.V. Kochian, 2001. A patch clamp study on the physiology of aluminum toxicity and aluminum tolerance in maize. Identification and characterization of Al(3+)-induced anion channels. Plant Physiol., 125: 292-305.
Direct Link - Salt, D.E., C.R. Prince, I.J. Pickering and I. Raskin, 1995. Mechanisms of cadmium mobility and accumulation in Indian mustard. Plant Physiol., 109: 1427-1433.
Direct Link - Sandman, G. and P. Boger, 1980. Copper-mediated lipid peroxidation process in photosynthetic membranes. Plant Physiol., 66: 797-800.
Direct Link - Sanita di Toppi, L.S. and R. Gabbrielli, 1999. Review: Response to cadmium in higher plants. Environ. Exp. Bot., 41: 105-130.
Direct Link - Shen, R., T. Iwashita and J.F. Ma, 2004. Form of Al changes with Al concentration in leaves of buckwheat. J. Exp. Bot., 55: 131-136.
Direct Link - Shioi, Y., H. Tamai and T. Sasa, 1978. Effects of copper on photosynthetic electron transport systems in spinach chloroplasts. Plant Cell Physiol., 19: 203-209.
Direct Link - Shioi, Y., H. Tamai and T. Sasa, 1978. Inhibition of photosystem II in the green alga Ankistrodesmus falcatus by copper. Physiol Plant., 44: 434-438.
Direct Link - Thurman, D.A. and J.L. Rankin, 1982. The role of organic acids in zinc tolerance in Deschampsia caespitosa. New Phytol., 91: 629-635.
Direct Link - Tomsett, A.B. and D.A. Thurman, 1988. Molecular biology of metal tolerances of plants. Plant Cell Environ., 11: 383-394.
Direct Link - Trivedi, S. and L. Erdei, 1992. Effects of cadmium and lead on the accumulation of Ca2+ and K+ and on the influx and translocation of K+ in wheat of low and high K+ status. Physiol. Plant., 84: 94-100.
Direct Link - Tukendorf, A. and T. Baszynski, 1985. Partial purification and characterization of copper-binding protein from roots of Avena sativa grown on excess copper. J. Plant Physiol., 120: 57-63.
Direct Link - Wu, L., D.A. Thurman and A.D. Bradshaw, 1975. The uptake of copper and its effect upon respiratory processes of roots of copper-tolerant and non-tolerant clones of Agrostis stolonifera. New Phytol., 75: 225-229.
Direct Link - Zheng, S.J., J.F. Ma and H. Matsumoto, 1998. High aluminium resistance in buckwheat: I. Al-induced specific secretion of oxalic acid from root tips. Plant Physiol., 117: 745-751.
Direct Link








