ABSTRACT
A glasshouse study was conducted to evaluate the effects of high temperature growth conditions on the integrity of the photosynthetic pigment complex in maize (Zea mays L.) plants and to elucidate the influence of ascorbate and kinetin applications. Total chlorophyll content declined with age in all categories of plants. Ascorbate and kinetin applications tended to slow down the rate of chlorophyll loss even though these treatments did not increase the pigment content of treated plants significantly over those in untreated plants. Chlorophyll a:b ratio varied significantly with plant age under both ambient and high temperature conditions for both of ascorbate and kinetin treatments. These ratios were higher in heat stressed plants than in non-stressed plants. Chlorophyll: Carotenoid ratios varied significantly with plant age both under ambient and high temperature growth conditions for both ascorbate and kinetin treatments. These ratios were generally higher under ambient conditions than under heat stress conditions. Correlations between air temperature and pigment contents/ratios were mainly negative and were generally higher under high temperature than under ambient growth conditions. Similarity in the pattern of variation of both Chlorophyll a:b ratios and Chlorophyll: Carotenoid ratios observed in this study emphasized the close functional relationship between the pigments. Applied substances did not significantly influence the integrity of the photosynthetic light-harvesting complex at the concentrations used in this study.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2006.357.362
URL: https://scialert.net/abstract/?doi=ajps.2006.357.362
INTRODUCTION
Heat stress is one of the environmental factors that elicit a series of physiological and biochemical reactions in plants. These may impose many secondary consequences on metabolism, including alterations in assimilation and carbon allocation (Pollock et al., 1993). High temperature has been reported to hasten declines in photosynthetic rate and viable leaf area during maturation of wheat (Shah and Paulsen, 2005). Some of these changes including decreased chloroplast biogenesis and disruption of lamellar systems, are largely reversible, except in situations where the tolerance limit of the species has been severely transgressed (Smillie et al., 1978; Gounaris and Brai, 1983; Gounaris et al., 1983; Pollock et al., 1993; Shah and Paulsen, 2005). The degree to which these systems are affected depends on the thermal kinetic window for the particular species (Burke, 1990).
Some other crucial systems, which can be affected by heat stress, include synthesis of chlorophyll a and b light harvesting complex (Burke, 1990; Pshibytko et al., 2004), photosynthetic oxygen evolution, PSII functioning and heterogeneity (Pshibytko et al., 2003). In certain instances, for example in barley, these effects were more pronounced in younger leaves (Pshibytko et al., 2004). The age-induced decline in chlorophyll content and photosynthetic activity is a widely reported phenomenon (Camp et al., 1982; Benbella and Paulsen, 1998). Often, chlorophyll content, chlorophyll a:b ratios as well as, Carotenoid: Chlorophyll ratios have been used as indicators of the sensitivity of the photosynthetic light harvesting complex (photo-systems) in plants subjected to various stress factors (Heath 1990; Maslova et al., 2003). To protect themselves from a vide variety of irradiance and temperature conditions; plants have evolved several mechanisms including those at the level of plastid pigment complexes (Strzalka et al., 2003). Of particular importance are the roles of carotenoids in protecting photosynthetic membranes against environmental stresses (Strzalka et al., 2003).
There are several reports that applied substances have helped in restoring pigment integrity in stressed plants. For example, kinetin applications have been commonly associated with reversal of the age induced senescence and loss of chlorophyll in plants (Benbella and Paulsen, 1998). Application of cytokinins (Benzyladenine) also significantly reduced the effect of heat stress (increased thermotolerance) (Cheikh and Jones, 1994) while ABA did not. Similarly, a new class of phytohormones, the brassinosteroids, has been used to overcome pigment loss due to salt stress (Anuradha and Rao, 1994). On the other hand, tolerance to heat stress has been associated with lower endogenous gibberellin levels (Sarker et al., 1994). Although several authors have reported on the response of the photosynthetic apparatus in a wide variety of crop plants (including maize) to high temperature stress (Burke, 1990) little information is available on how these effects are modified by applied substances in maize, particularly under tropical conditions.
This research was therefore designed to evaluate the effect of ascorbate and kinetin applications on the integrity of the photosynthetic pigment complex in maize plants grown under high temperature conditions obtainable in the Nigerian Savanna.
MATERIALS AND METHODS
Plant culture and treatments: The experiment was conducted at the experimental gardens of the Department of Biological Sciences, Ahmadu Bello University, Samaru and Zaria. Zaria is located on Lat. 11°11’ N, 7°38’ E and altitude 686 m, in the Northern Guinea Savanna Region of Nigeria, West Africa. Seeds of an open-pollinated variety of maize (Zea mays L.) were sown in earthenware pots of approximately 38 cm depth and 30 cm diameter. The pots were filled with about 155 cm3, garden soil to within 3-4 cm of maximum capacity. Fertilizer (NPK 15:15:15) was applied at computed rate equivalent to NAERLS (1994) recommendation.
Plants subjected to heat stress were exposed to a mean air temperature of about 7.0°C higher than ambient and a mean soil temperature of about 5.0°C higher than ambient for 8-12 h each day, throughout the duration of the experiment. Maximum mean air temperature to which heat stressed plants were exposed during this period was 41.0°C in the air and 35.8°C in the soil. Mean day temperature was 37.3°C (air) and 32.2°C (soil). For plants grown under ambient conditions, maximum temperatures during this period were 32.0°C (air) and 29.0°C (soil). Mean day temperatures were 30.3°C (air) and 27.3°C (soil).
After five weeks of growth sampling for pigment contents started and this was maintained at weekly intervals for the next five weeks. After the first two samplings, freshly prepared solutions of ascorbate and kinetin (6-benzylaminopurine) were applied as foliar sprays using a simple atomizer. Concentrations of 100, 200 and 300 ppm of each were applied to appropriately labelled pots. Twenty to fifty (20-50) mL of each were applied till the leaf surfaces were properly wet and dripping. No wetting agents were used (Krishnayya and Date, 1996). For each treatment, plants were thinned to three plants per pot and they were grown and maintained to maturity. Each treatment was replicated three times in a completely randomised design.
Sampling and analyses: Leaf pigment content of the uppermost fully expanded leaf was estimated by collecting about 0.5 g of fresh leaf materials from the sampled plants by cutting basal and median sections from they blades with a clean pair of scissors, cleaning them and then homogenizing in 7 mL of 95% ethanol using a clean pestle and mortar under reduced light intensity. The homogenized material was then placed in clean specimen bottles and left in the dark for 24 h. according to methods described by Carreras (1996). Absorbance of the green, clear supernatant liquid obtained after this incubation period was then read in a corning Colorimeter (model EEL 253) with the wavelength set at 665, 650 and 470 nm for chlorophylls ‘a’, ‘b’ and carotenoids, respectively. Figures for absorbance of the solution obtained from this were converted into mg g-1 fr.wt. using the following formulae:
Where:
| V | = | Final volume of extracting solution |
| W | = | Weight of leaf material used |
| A | = | Absorbance readings at the wavelengths indicated |
[12.7, 2.69, 22.9, 4.68] = Absorption coefficients as outlined by Witham et al. (1970).
Carotenoid content was calculated from formula given by Clarke and Switzer (1977) as:
Where:
| A | = | Absorbance at the given wavelength |
| C | = | Concentration of Carotenoids |
| E1%1cm | = | Extinction coefficient of a 1% solution in 1 cm tube. |
Data obtained were used to compute pigment ratios. Figures obtained from this were statistically analysed by ANOVA (Parker, 1980). Correlation coefficients between air temperature data and leaf pigment data were also computed.
RESULTS
Total chlorophyll content varied significantly with age both under ambient and high temperature conditions for plants treated with both ascorbate and kinetin (LSD(0.05) = 0.68 and 0.72, respectively). Generally, chlorophyll contents peaked at the seventh week and tended to decline thereafter, though these declines were sharper in plants to which neither ascorbate nor kinetin was applied (Table 1 and 2). Chlorophyll content in plants to which either of ascorbate or kinetin was applied, tended to increase or remained steady, especially toward the later part of the growth period. Although significant differences occurred among treatments, no treatment showed significantly higher chlorophyll than controls at both ambient and high temperature for each of ascorbate or kinetin treatments.
Chlorophyll a:b ratio varied significantly with plant age under both ambient and high temperature conditions for both of ascorbate and kinetin treatments (LSD(0.05) = 0.007 and 0.008, respectively).
| Table 1: | Total chlorophyll content (mg g-1 fr.wt.) in leaves of maize (Zea mays L.) plants grown under ambient (B) and high temperature (A) conditions as influenced by ascorbate treatments |
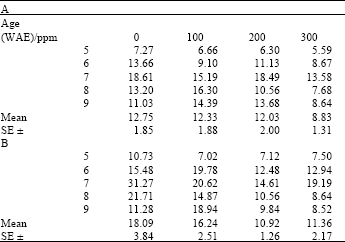 | |
| Table 2: | Total chlorophyll content (mg g-1 fr.wt.) in leaves of maize (Zea mays L.) plants grown under ambient (B) and high temperature (A) conditions as influenced by kinetin treatments |
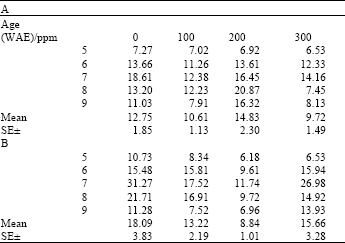 | |
After an initial increase, this ratio remained more or less stable during the later parts of the growth period (Table 3 and 4). Chlorophyll a:b ratios were significantly higher in heat stressed plants than in non-stressed plants. On the other hand, differences between kinetin treatments at both ambient and high temperatures were not significant (Table 4).
Chlorophyll: Carotenoid ratios varied significantly with plant age both under ambient and high temperature growth conditions for both ascorbate and kinetin treatments (LSD (0.05) = 0.20 and 0.16, respectively). After an initial increase, this ratio remained fairly stable during the later parts of the growth period (Table 5 and 6). Chlorophyll: Carotenoid ratios were generally higher under ambient conditions than under heat stress conditions. Among the treatments, Chlorophyll: Carotenoid ratios in treated plants were not significantly higher than in controls both for ascorbate (Table 5) and kinetin (Table 6) treatments.
| Table 3: | Chlorophyll: Carotenoid ratio in leaves of maize (Zea mays L.) plants grown under ambient (B) and high temperature (A) conditions as influenced by ascorbate treatment |
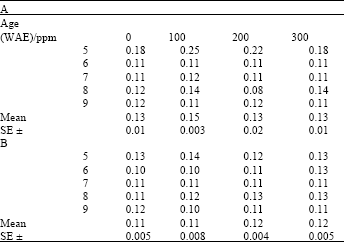 | |
| Table 4: | Chlorophyll: Carotenoid ratio in leaves of maize (Zea mays L.) plants grown under ambient (B) and high temperature (A) conditions as influenced by kinetin treatments |
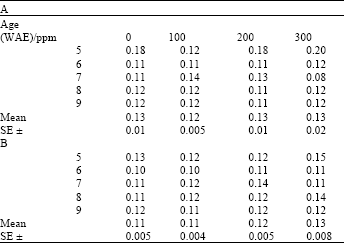 | |
| Table 5: | Chlorophyll a:b ratio in leaves of maize (Zea mays L.) plants grown under ambient (B) and high temperature (A) conditions as influenced by ascorbate treatments |
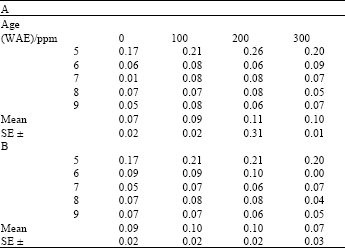 | |
| Table 6: | Chlorophyll a:b ratio in leaves of maize (Zea mays L.) plants grown under ambient (B) and high temperature (A) conditions as influenced by kinetin treatments |
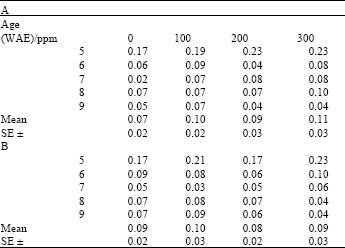 | |
| Table 7: | Correlation coefficients between pigment contents, ratios and air temperature in leaves of maize (Zea mays L.) plants grown under ambient and high temperature conditions as affected by ascorbate and kinetin treatments |
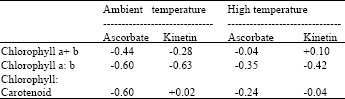 | |
| p = 0.63; N = 10 | |
Correlations between air temperature and pigment contents/ratios were mainly negative (Table 7) and were generally higher under high temperature than under ambient growth conditions.
DISCUSSION
Negative correlations between pigment contents/ratios and air temperatures were expected, as temperature is known to severely affect the structure and functioning of the photosynthetic apparatus However, the weak but positive correlation between total chlorophyll and high temperature for kinetin applications and for ascorbate applications at ambient temperature indicated some limited level of enhancement effects of kinetin and ascorbate. Among other physiological effects, high temperatures enhance loss of chlorophyll and photosynthetic enzymes (Al-Khatib and Paulsen, 1984; Xu et al., 1995). Growth at temperatures outside the thermal kinetic window of a particular species results in both structural and functional changes within the pigment complex, including inhibition of chloroplast biogenesis and disorientation of lamellar systems (Smillie et al., 1978). Reversible rearrangements of thylakoid membranes also occur (Sunby and Andersson, 1985). Functionally, the heat induced dissociation of the Light-harvesting Complex (LHC), results in decreased quantum yields of electron transport at limiting light intensities, followed by a loss of electron transport activity at rate saturating light intensities (Weis, 1985).
That total chlorophyll content declined with age in all categories of plants is simply an expression of the age-induced senescence in these plants. Ascorbate and kinetin applications tended to slow down the rate of chlorophyll loss even though these treatments did not increase the pigment content of treated plant significantly over those in untreated plants. The lack of pronounced enhancements of pigment content by applied substances could be due to the fact that only single applications were given. These results support the findings of Al-Khatib and Paulsen (1985) and Benbella and Paulsen (1998) in wheat, where although kinetin applications delayed senescence induced by heat stress, repeated applications were necessary to achieve full effect.
That chlorophyll a:b ratios were higher in heat stressed plants than in non-stressed plants meant that there was relatively more of chlorophyll a than b, in the stressed plants. On the other hand, higher chlorophyll: carotenoid ratios in plants grown under ambient conditions meant that in plants grown under ambient temperature, there was more chlorophyll than carotenoid, as opposed to those grown under high temperature, which had more carotenoids than chlorophyll. Both chlorophyll concentration and chlorophyll a:b ratios indicate the rate of photosynthesis of the LHC, which proceeds maximally only within the Thermal Kinetic Window (TKW) of the species (Smillie et al., 1978). These results imply that chlorophyll b may be more stable at the temperatures employed here than chlorophyll a. Although chlorophyll a is dominant and more widely distributed in higher plants (Richter, 1978), these results indicate that it may have been more readily degraded under the conditions of this investigation. On the other hand, carotenoids seemed to have been more stable than the chlorophylls, as was indicated by the chlorophyll: carotenoid ratios of the heat stressed plants. Carotenoids are traditionally documented as pigments that act both as antennae to the LHC at low light intensities, as well as photo-protectors of the chlorophylls at high light intensities. They play their role as photo-protectors by modulating membrane physical properties and quenching of free radicals (reactive oxygen species) generated by stress conditions, including high temperature (Strzalka et al., 2003). Similarity in the pattern of variation of both chlorophyll a:b ratios and chlorophyll: carotenoid ratios observed in this study emphasize the close functional relationship between these pigments. Furthermore, the stabilizing of these ratios in plant leaves during the later parts of the growth cycle suggested an early acclimation response in the plants and this could indicate that the plants did not experience any appreciable period outside their TKW at the temperatures encountered during the course of this investigation.
Lack of clear difference (except for chlorophyll a:b ratio at 200 and 300 ppm in plants under ambient conditions), between treatments and concentrations of applied substances, indicated that these substances did not significantly influence the integrity of the photosynthetic light-harvesting complex at the concentrations used in this study.
REFERENCES
- Al-Khatib, K. and G.M. Paulsen, 1984. Mode of high temperature injury to wheat during grain development. Physiol. Plant., 61: 363-368.
CrossRefDirect Link








