ABSTRACT
Endogenous substrates and physiological ligands of Glutathione s-transferases (GSTs) have capabilities to reduce the enzyme reactions for xenobiotic substrates. In order to investigate the physiological counterparts to plant GSTs, the inhibitory effect of pumpkin (Cucurbita maxima Duch.) seedling extract on 1-chloro-2,4-dinitrobenzene (CDNB) conjugating activities of pumpkin GSTs was examined. Alcoholic extract of pumpkin seedlings showed different degrees of inhibitory effect for the three GST species namely, CmGSTU1, CmGSTU2 and CmGSTU3. In the assay system, 50% inhibition of CmGSTU1 and CmGSTU3 activities were achieved with the extracts from 178 and 96 mg fresh tissues, respectively. The activity of CmGSTU2 was decreased marginally by the extract and did not reach 50% level. To separate into constituents, the extract was applied to Sep-pac Vac C18 column and eluted with different concentrations of methanol. Inhibitory potencies of Methanol-Eluted Fractions (MEFs) against CmGSTU3 were estimated. Twenty, forty and sixty percent MEFs exhibited higher inhibitions followed by 0% MEF. CmGSTU3 activity was decreased marginally by 100% MEF. MEFs of pumpkin seedlings extract were successively analyzed by HPLC. The analyses indicated that MEFs contain a number of inhibitors with different degrees of hydrophobicity, while 0% MEF also contains hydrophilic inhibitors. The above results suggest that pumpkin seedlings contain various physiological substrates or ligands for pumpkin GSTs.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2006.345.352
URL: https://scialert.net/abstract/?doi=ajps.2006.345.352
INTRODUCTION
Plants produce an amazing diversity of secondary metabolites that have versatile physiological and protective roles (Coleman et al., 1997; Steyn et al., 2002) most are phytotoxic even to the cell that produce them until appropriate cellular localization is accomplished (Marrs, 1996; Alfenito et al., 1998). The ability to sequestrate such harmful compounds is crucial for survival of cells and plants. Storage of these toxic compounds within central vacuole, a compartment with a very low metabolic activity, is generally assumed to protect plant cells against potentially toxic effect of these substances (Klein et al., 1996). In addition, a wide variety of hormones like auxins and cytokinins are produced in plant bodies but maintaining their effective concentration in the cell is prerequisite to run specific biological processes smoothly. Hence, plants adapt various mechanisms for proper management of endogenous phytochemicals for their survival.
Plants enzymatically detoxify various exo-and endogenous toxins using a three-phase detoxification system (Neuefeind et al., 1997). Phase I (transformation): introduction of functional groups onto substrates by cytochrome P450. Phase II (formation of conjugate): conjugation of substrates with hydrophilic compounds such as glutathione (GSH). Phase III (compartmentation): transportation of conjugates into vacuole or apoplast by ATP-dependent transporters (Coleman et al., 1997; Rea, 1999; Walczak and Dean, 2000; Dixon et al., 2002). With regard to phase II, a number of GSTs (EC 2.1.5.18) that use a wide variety of herbicides as substrates for glutathione conjugation reaction have been reported in plants (Irzyk and Fuerst, 1993; Lamoureux and Rusness, 1989; Timmerman, 1989; Dixon et al., 1998; Edwards et al., 2000).
In addition to detoxification of exogenous toxins, plant GSTs are involved in the metabolism of endogenous substances like anthocyanins and some phenylpropanoids, thereby reducing their toxicity (Dean et al., 1995; Marrs et al., 1995; Alfenito et al., 1998; Mueller et al., 2000). GSTs also play a role in the plant protection system against oxidative damage (Gronwald and Plaisance, 1998; Cummins et al., 1997) and can function as GSH peroxidases (Mannervik and Danielson, 1988; Bartling et al., 1993). On the whole, however, few endogenous substrates of plant GSTs have been characterized and much remains to be done in that domain.
More recent hypotheses suggest that the main role of plant GSTs is to act as binding proteins or ligandins (Walbot et al., 2000). They are involved in intracellular transport of hydrophobic and amphipathic molecules including plant hormone like auxin (Bilang et al., 1993; Zettl et al., 1994; Bilang and Sturm, 1995; Watahiki et al., 1995), cytokinin (Gonneau et al., 1998) and auxin-like substances (Droog et al., 1995) and thus can modulate effective hormone concentration in the cell. Some plant GSTs have high affinities for tetrapyrroles and porphyrin metabolites (Lamoureux and Rusness, 1989; Dixon et al., 1999). Bindings of GST with tetrapyrroles, porphyrins and plant hormones, inhibit its own activity toward xenobiotics like CDNB, but the inhibitory ligands do not undergo conjugation with glutathione (Litwack et al., 1971; Hayes and Pulford, 1995; Lederer and Boger, 2003).
The endogenous substrates and ligands for GSTs often can decrease GST activity towards xenobiotics. In this way, the inhibition study of GSTs could be the valuable tool for searching physiological substrates and non-substrate ligands of the enzymes.
Plant GSTs are abundant both in quantity (up to 1% of total soluble protein) and in diversity (42 distinct GSTs have been found in corn) (McGonigle et al., 2000). Pumpkin plant contains high levels of glutathione (Nakagawa et al., 1986) and the young seedlings as well as the culture cells induced from fruit expresses high GST activity particularly under stressful conditions (Fujita et al., 1994, 1995; Fujita and Hossain, 2003a). Therefore, we used pumpkin seedlings in our inhibition studies. Previously, four cDNA genes of pumpkin GSTs, CmGSTU1, CmGSTU2, CmGSTU3 and CmGSTF1 have been cloned from cDNA libraries of pumpkin callus and flowers and their expressions in E. coli were established (Fujita and Hossain, 2003b; Hossain and Fujita, 2002). In order to identify inhibitors of pumpkin GSTs, a small progress has been achieved in the past (Fujita and Hossain, 2003a) but at present no research findings published yet regarding the physiological inhibitors of the enzyme. However, studies on endogenous GST inhibitors are important for better understanding of their physiological roles and possible interactions with GSTs. Hence, we made an attempt to search endogenous substances in pumpkin seedlings which inhibit the activity of individual pumpkin GSTs towards CDNB. This study will not give only the way of searching the intrinsic substrates or physiological ligands of pumpkin GSTs but will also provide a basis for understanding the physiological role of the compounds in plant cells.
MATERIALS AND METHODS
Plant material: Mature pumpkin seeds were sown in vermiculite saturated with deionized water and incubated in the dark at 25°C. Six to seven-day old seedlings were used for extraction.
Extraction and fractionation: After removal of cotyledons, 25 g fresh Pumpkin Seedlings (PS) were submerged twice in each 100 mL of methanol : chloroform : water (12:5:3, v/v/v) and once in the same volume of 70% ethanol at -20°C as described by Lancaster and Shaw (1989). The first two extractants were combined and then separated into two phases with the addition of 90 mL chloroform and 110 mL water. After removal of chloroform (lower) phase, the third one (100 mL extractant of 70% ethanol) was added to the methanol-water (upper) phase and thereafter evaporated under 40°C. Dried substances were then dissolved in 10 mL distilled water, stored at -8°C until use as alcoholic extract of PS.
The alcoholic extract of 7.8 mL was put into a solid phase extraction kit (Sep-Pak Vac 12 mL, C18 column, Waters, Ireland) and eluted with 20 mL of distilled water (0% methanol) followed by the same volume of 20, 40, 60 and 100% methanol in stages. The collected liquids were evaporated to dryness, dissolved in 0.8 mL of distilled water and used as Methanol Eluted Fractions (MEFs) of PS.
Preparation of enzyme: E. coli cells, containing CmGSTU1, CmGSTU2 and CmGSTU3 cDNAs in pBluescripts [SK(-)] were cultivated separately for around 16 h at 37°C in Luria-Bertani liquid media with ampicillin (50 μL mL-1) and 1 mM isopropyl-β-D-thiogalactopyranoside (IPTG). For expression of CmGSTU3, IPTG was not included in the medium. After incubation, the cells were centrifuged at 2400xg for 10 min. The cells’ pellete was homogenized in 25 mM Tris-HCl buffer (pH 8.0) containing 1 mM EDTA, 1% (w/v) ascorbate and 10%(v/v) glycerol with a mortar and pestle. A small amount of sea sand was added to make grinding easier. Cellular debris was precipitated by centrifugation (10000xg at 4°C for 10 min) and the supernatant was used as enzyme solution.
Enzyme inhibition: GST activity was determined spectrophotometrically by the method of Booth et al. (1961) with some modifications. The reaction mixture contained 100 mM potassium phosphate buffer (pH 6.5), 1.5 mM reduced glutathione, 1 mM CDNB, a fixed volume of enzyme solution and various amount of alcoholic extract. The final volume of the reaction mixture was maintained at 0.7 mL by adjusting volume of distilled water. The enzyme reaction was initiated by the addition of CDNB and A340 was monitored at 25°C for 1 min. The effects of MEFs as well as HPLC eluates of PS extract were measured only for CmGSTU3 using the same assay condition. Remaining GST activity was expressed as percentage of the activity of the system without inhibitor.
HPLC analysis: MEFs obtained from Sep-Pak Vac C18 column were analyzed on a LC-6AD Liquid Chromatograph (Shimadzu, Japan) fitted with UV-VIS detector (SPD-6AV) and C-R6A Chromatopac. Separation was performed on to a Shim Pack CLC-ODS column (4.6 mm i.d.x250 mm, Shimadzu, Japan). The flow rate was 0.6 mL min-1 and detection was carried out at 220 nm. For 0%MEF, the column was eluted with water (0% methanol) and for 20, 40 and 60% MEFs, with linear gradients of 10-30, 30-80 and 50-80% methanol, respectively.
Activity profiling: Fifty micro liters of each MEF of PS extract (corresponding to extract from 1.22 g fresh tissues) was injected on to the column. The effluent from the column was fractionated according to each major or some adjacent minor peaks or after some intervals (when no peak appeared). The solvent was removed by evaporation and the dried substances were redissolved in 250 μL distilled water. Inhibition of CmGSTU3 (CDNB) activity was assayed for individual fractions.
All studies were conducted at the laboratory of plant stress responses, Faculty of Agriculture, Kagawa University, Japan during January to November, 2005.
RESULTS
Inhibition of pumpkin GSTs by alcoholic extract of PS: The inhibitory effect of alcoholic extract of PS on three pumpkin GSTs, namely CmGSTU1, CmGSTU2 and CmGSTU3 (Fujita et al., 1994, 1998) was assayed towards CDNB and presented in Fig. 1. PS extract showed the highest inhibitory effect on CmGSTU3 closely followed by CmGSTU1. In the assay system, 50% inhibition of CmGSTU1 and CmGSTU3 was achieved with the extracts from 178 and 96 mg of fresh PS tissues, respectively. The extract showed only a weak inhibitory effect on CmGSTU2 and the intensity of inhibition never reached 50% level in this experiment. In the case of CmGSTU2, the assay system always contained high concentration of protein since the prepared solution of CmGSTU2 was low in specific activity (352 nmol min-1 mg-1 protein) as compared with CmGSTU1 and CmGSTU3 (3, 708 and 5, 501 nmol min-1 mg-1 protein, respectively).
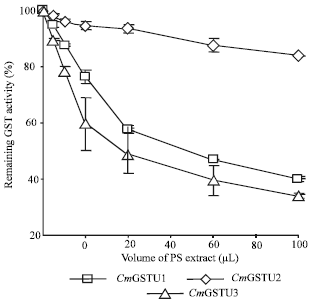 | |
| Fig. 1: | Inhibition of pumpkin GSTs activities towards CDNB by Pumpkin Seedling (PS) extract. Results were obtained from two independent experiments and bars indicate standard error. Each μL extract corresponds to the amount of the substances present in 2.5 mg of fresh PS tissues |
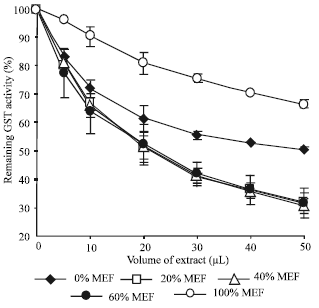 | |
| Fig. 2: | Inhibition of CmGSTU3 activity towards CDNB by Methanol Eluted Fractions (MEFs) of PS extract. Results were obtained from two independent experiments and bars indicate standard error. Each μL extract corresponds to the amount of the substances present in 24.4 mg of fresh tissue |
Previously, Fujita and Hossain (2003b) also reported lower specific activity of CmGSTU2 than CmGSTU1 and CmGSTU3 purified from E. coli cells. To check whether the variation in inhibition among GST species is caused by different amount of various proteins in reaction mixture or by different affinities between inhibitors and GST species, we carried out an inhibition study adding Bovine Serum Albumin (BSA) as an extra protein.
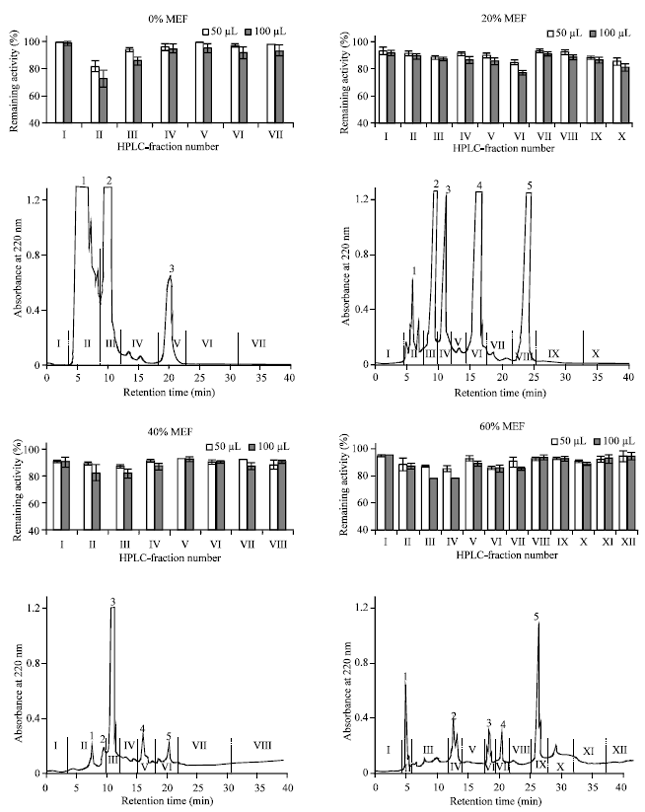 | |
| Fig. 3: | HPLC-based activity profiling with different MEFs of PS extract for CmGSTU3 activity towards CDNB. The HPLC chromatograms show the absorbance of compounds detected at 220 nm and bar graphs above each chromatogram show the inhibitory effect of corresponding HPLC-fractions. Fifty micro liters of extract (corresponding to extract from 1.22 g fresh PS tissues) was injected on to HPLC column. Chromatographic conditions are described in “materials and methods” section. The solvent of each collected fractions was removed by evaporation and the dried substances were redissolved in 250 μL distilled water. Inhibitory potency was checked for each 50 and 100 μL. Each experiment was repeated two times and bars indicate standard error. Each HPLC-chromatogram shows the typical result obtained from one experiment |
The addition of BSA to the assay systems of CmGSTU1, CmGSTU3 did not show any significant variation in inhibition caused by PS extract (data not shown), suggesting that the differences of inhibition among different GSTs are the specific characteristics for the respective species.
Inhibition of CmGSTU3 by MEFs of PS extract: We separated the alcoholic extract of PS into five fractions through Sep-Pak Vac C18 column chromatography with various concentrations of methanol (as stated in “materials and methods”). To assay the inhibitory effects of MEFs obtained, we selected CmGSTU3 since it exhibited the highest activity among the three GST enzyme solutions. Twenty, forty and sixty percent MEFs showed similar high inhibitions followed by 0% MEF, while 100% MEF inhibited marginally (Fig. 2). These results suggest that PS contains hydrophobic as well as hydrophilic inhibitors.
HPLC analysis: In order to obtain further information about inhibitory properties of constituents in PS extract, we also carried out HPLC analyses of each MEF except for 100% MEF as it exhibited small inhibitory effect. Fifty micro liters of each sample were loaded on to a Shim Pack CLC-ODS column. To get good separations, the column was eluted with proper gradients of methanol as described in “materials and methods”. HPLC elution profiles of different MEFs and the inhibitory effects of obtained fractions are shown in Fig. 3.
Figure 3 indicates that 0% MEF of PS extract contains some potent inhibitors in HPLC-fraction number-II (f-II). Probably, a number of hydrophilic substances are eluted in the fraction as it contains the effluents of peak-1 including some adjacent minor peaks. In case of 20% MEF, f-VI (peak-4) showed high degree of inhibitory potency. By NMR and Mass Spectrophotometries, the chemical structure of the major compound in f-VI was identified as tryptophan but authentic tryptophan was found to be a weak inhibitor and its degree of inhibition was very low as compared with that of f-VI (data not shown), suggesting that the fraction might contain some other inhibitory compounds along with tryptophan, which have no absorption at 220 nm. Some constituents corresponding to peak-1, 2 and 3 for 40% MEF and most of the moderate peaks detected at 220 nm for 60% MEF also showed small inhibitory effects. This experiment strongly suggests that HPLC-fractions with high inhibitory potencies for a particular MEF contain some potent inhibitors towards CmGSTU3.
DISCUSSION
The GST inhibitors can be grouped into three classes based on binding site on GST protein and mechanism of inhibition (Lyon et al. 2003). The first is the analogues of electrophilic substrates, which bind in the hydrophobic region of the active site (H-site) and competitively inhibit binding of hydrophobic electrophiles. The second is the glutathione conjugates, which occupy both the glutathione binding site (G-site) and a part of the H-site and are typically competitive with respect to both glutathione and hydrophobic substrates. Third, a collection of compounds referred to as non-substrate ligands which bind at a different site (L-site) for the purpose of intracellular transportation are noncompetitive inhibitors of GSTs that act as ligandins.
It has been well documented that substrates of GSTs (fall under the first class inhibitors) are typically hydrophobic (Koehler et al., 1997) while non-substrate ligands (fall under the third class inhibitors) are either hydrophobic or amphipathic substances (Ketley et al., 1975; Reinemer et al., 1991; Bilang et al., 1993; Lyon and Atkins, 2002). In regard to the second class inhibitors, considerable research reports indicated that GSH-congugates are potential inhibitors of animal and plant GSTs (Lucenta et al., 1998). Reportedly, glutamyl-COOH group of GSH-conjugate is very hydrophilic and is responsible for binding to G-site (Burg et al., 2002). It is also reported that longer S-alkyl chain or larger aromatic groups of GSH-conjugates show relatively higher hydrophobicity as well as higher inhibitory potencies as compared to shorter or smaller ones (Koehler et al., 1997; Ortiz-Salmeron et al., 2001).
The endogenous substances in pumpkin seedling that inhibit CDNB conjugating activity of CmGSTU3 might be physiological substrates or non-substrate ligands for the enzyme. The potential substances present in 20, 40 and 60% MEFs, seem to be amphipathic and/or hydrophobic inhibitors. Most of the research reports addressed some secondary sulphur compounds like glutathione derivatives and its structurally related substances as amphipathic inhibitors (Neuefeind et al., 1997; Lucenta et al., 1998) that are common in most of the plant cells, particularly abundant in Allium spp. (Lancaster and Shaw, 1989; Jones et al., 2004). A considerable number of research findings indicated that plant natural products with different degrees of hydrophobicity exhibit significant interaction with GSTs. These include phytoalexin medicarpin (Li et al., 1997), anthocyanin, cyanidin-3-glucoside (Marrs et al., 1995; Mueller et al., 2000), cinnamic acid, coumaric acid and some of their derivatives (Dean et al., 1995), many other flavonoids, phenol and α, β-unsaturated carbonyl compounds (Gronwald and Plaisance, 1998; Yu and Abo-Elghar, 2000), phorphyrins and tetrapyrrole metabolites (Lederer and Boger, 2003) and a wide variety of plant hormones and related substances (Watahiki et al., 1995). It is possible that pumpkin seedlings contain some of these phytochemicals which showed inhibitory effects on pumpkin GSTs either by formation of conjugate with glutathione or by acting as inhibitory ligands.
The potential substances in 0% MEF are might be some water soluble organic acids and/or acidic carbohydrates. Another possibility is that pumpkin seedlings contain glutathione conjugates that formed previously in course of normal cellular detoxification. These conjugates are very hydrophilic and are capable of decrease GST activity by reducing the formation of further glutathione conjugates (Rea et al., 1998). However, to the best of our knowledge no hydrophilic compounds are detected as inhibitors of GSTs in the past. Interestingly, our results indicated a number of hydrophilic compounds in 0% MEF of PS extract where at least one might be a potent inhibitor.
Finally, we conclude that tissues of pumpkin seedling contain a number of inhibitors, most of which might represent physiological substrates or non-substrate ligands of pumpkin GSTs. Some of the inhibitors are water soluble and the others are different in hydrophobicity. Present research however, will provide a basis for better understanding the physiological role of endogenous compounds of plant tissue and their possible interactions with GSTs. In addition, a new idea about hydrophilic inhibitors of GSTs will also be established. However, we are now trying to develop an appropriate method to separate and identify the structures of individual compounds of pumpkin seedling extract that will lead to establishment of the inhibition mechanism of the compounds.
REFERENCES
- Alfenito, M.R., E. Souer, C.D. Goodman, R. Buell, J. Mol, R. Koes and V. Walbot, 1998. Functional complementation of anthocyanin sequestration in the vacuole by widely divergent glutathione S-transferases. Plant Cell., 10: 1135-1149.
Direct Link - Bartling, D., R. Radzio, U. Steiner and E.W. Weiler, 1993. A glutathione S-transferase with glutathione- peroxidase activity from Arabidopsis thaliana. Eur. J. Biochem., A216: 579-586.
PubMedDirect Link - Bilang, J. and A. Sturm, 1995. Cloning and characterization of a glutathione S-transferase that can be photolabeled with 5-azido-indole-3-acetic acid. Plant Physiol., 109: 253-260.
PubMedDirect Link - Bilang, J., H. Macdonald, P.J. King and A. Sturm, 1993. A soluble auxin-binding protein from Hyoscyamus muticus is a glutathione S-transferase. Plant Physiol., 102: 29-34.
PubMedDirect Link - Burg, D., L. Hameetman, D.V. Filippov, G.A. van der Marel and G.J. Mulder, 2002. Inhibition of glutathione S-transferase in rat hepatocytes by a glycine-tetrazole modified S-alkyl-GSH analogue. Bioorganic Med. Chem. Lett., 12: 1579-1582.
PubMedDirect Link - Coleman, J.O.D., M.M.A. Blacke-Kalff and T.G.E. Davies, 1997. Detoxification of xenobiotics by plants: Chemical modification and vacuolar compartmentation. Trends Plant Sci., 2: 144-151.
CrossRefDirect Link - Cummins, I., D.J. Cole and R. Edwards, 1997. Purification of multiple glutathione transferases involved in herbicide detoxification from wheat (Triticum aestivum L.) treated with the safener fenchlorazole-ethyl. Pesticide Biochem. Physiol., 59: 35-49.
Direct Link - Dean, J.V., T.P. Devarenne, I.S. Lee and L.E. Orlofsky, 1995. Properties of a maize glutathione S-transferase that conjugates coumaric acid and other phenylpropanoids. Plant Physiol., 108: 985-994.
PubMedDirect Link - Dixon, D.P., A. Lapthorn and R. Edwards, 2002. Protein family review, plant glutathione transferases. Genome Biol., 3: 3004.1-3004.10.
PubMedDirect Link - Dixon, D.P., D.J. Cole and R. Edwards, 1999. Dimerization of maize glutathione transferases in recombinant bacteria. Plant Mol. Biol., 40: 997-1008.
CrossRefDirect Link - Dixon, D.P., I. Cummins, D.J. Cole and R. Edwards, 1998. Glutathione-mediated detoxification system in plants. Curr. Opin. Plant Biol., 1: 258-266.
CrossRefDirect Link - Droog, F.N.J., P.J.J. Hooykaas and B.J. van der Zaal, 1995. 2,4-dichlorophynoxyacetic acid and related chlorinated compounds inhibit two auxin-regulated type-III tobacco glutathione S-transferases. Plant Physiol., 107: 1139-1146.
PubMedDirect Link - Edwards, R., D.P. Dixon and V. Walbot, 2000. Plant glutathione S-transferases: Enzymes with multiple functions in sickness and in health. Trends Plant Sci., 5: 193-198.
PubMedDirect Link - Fujita, M. and M.Z. Hossain, 2003. Modulation of pumpkin glutathione S-transferases by aldehydes and related compounds. Plant Cell Physiol., 44: 481-490.
Direct Link - Fujita, M. and M.Z. Hossain, 2003. Molecular cloning of cDNAs for three tau-type glutathione S-transferases in pumpkin (Cucurbita maxima) and their expression properties. Physiol. Plant., 117: 85-92.
Direct Link - Fujita, M., Y. Adachi and Y. Hanada, 1994. Preliminary characterization of glutathione S-transferases that accumulate in callus cells of pumpkin (Cucurbita maxima Duch.). Plant Cell Physiol., 35: 275-282.
Direct Link - Fujita, M., Y. Adachi and Y. Hanada, 1995. Glutathione S-transferases predominantly accumulate in pumpkin culture cells exposed to excessive concentrations of 2,4-dichlorophenoxyacetic acid. Biosci. Biotechnol. Biochem., 59: 1721-1726.
Direct Link - Fujita, M., Y. Hanada, H. Tomita, N. Takao and N. Taniguchi, 1998. Preliminary studies on organ specificities of 2,4-dichlorophenoxyacetic acid-inducible glutathione S-transferases and new glutathione S-transferases preferential to developing organs in pumpkin (Cucurbita maxima Duch.). Kagawa Daigaku Nogakubu Gakujutsu Hokoku, 50: 105-113.
Direct Link - Gonneau, J., R. Mornet and M.A. Laloue, 1998. A Nicotiana plumbaginifolia protein labeled with an azido cytokinin agonist is a glutathione S-transferase. Physiol. Plant., 103: 114-124.
Direct Link - Gronwald, J.W. and K.L. Plaisance, 1998. Isolation and characterization of glutathione S-transferase from sorghum. Plant Physiol., 117: 877-892.
PubMedDirect Link - Hayes, J.D. and D.J. Pulford, 1995. The glutathione S-transferase supergene family: Regulation of gst and the contribution of the isoenzymes to cancer chemoprotection and drug resistance. Crit. Rev. Biochem. Mol. Biol., 30: 445-520.
CrossRefPubMedDirect Link - Hossain, M.Z. and M. Fujita, 2002. Purification of a phi-type glutathione S-transferase from pumpkin flowers and molecular cloning of the cDNA. Biosci. Biotechnol. Biochem., 66: 2068-2076.
Direct Link - Irzyk, G.P. and E.P. Fuerst, 1993. Purification and characterization of a glutathione S-transferase from benoxacor-treated maize (Zea mays). Plant Physiol., 102: 803-810.
Direct Link - Jones, M.G., J. Hughes, A. Tregova, J. Milne, A.B. Tomsett and H.A. Collin, 2004. Biosynthesis of flavour precursors of onion and garlic. J. Exp. Bot., 55: 1903-1918.
Direct Link - Ketley, J.N., W.H. Habig and W.B. Jakoby, 1975. Binding of nonsubstrate ligands to the glutathione S-transferases. J. Chem., 250: 8670-8673.
Direct Link - Klein, M., G. Weissenbock, A. Dufaud, C. Gaillard, K. Kreuz and E. Martinoia, 1996. Different energization mechanisms drive the vacuolar uptake of a flavonoid glucoside and a herbicide glucoside. J. Biol. Chem., 271: 29666-29671.
Direct Link - Koehler, R.T., H.O. Villar, K.E. Bauer and D.L. Higgins, 1997. Ligand-based protein alignment and isozyme specificity of glutathione S-transferase inhibitors. Proteins, 28: 202-216.
PubMedDirect Link - Lamoureux, G.L. and D.G. Rusness, 1989. The Role of Glutathione and Glutathione S-transferases in Pesticide Metabolism, Selectivity and Mode of Action in Plants and Insects. In: Glutathione: Chemical, Biochemical and Medical Aspects, Dolphin, D., R. Poulson and O. Avramovic (Eds.). John Wiley and Sons, New York, pp: 153-196.
- Lancaster, J.E. and M.L. Shaw, 1989. γ-Glutamyl peptides in the biosynthesis of S-alk(en)yl-L-cysteine sulphoxides (flavour precursors) in Allium. Phytochemistry, 28: 455-460.
CrossRefDirect Link - Lederer, B. and P. Boger, 2003. Binding and protection of porphyrins by glutathione S-transferases of Zea mays L. Biochim. Biophys. Acta, 1621: 226-233.
PubMedDirect Link - Litwack, G., B. Ketterer and I.M. Arias, 1971. Ligandin: A hepatic protein which binds steroids, bilirubin, carcinogens and a number of exogenous organic anions. Nature, 234: 466-467.
Direct Link - Li, Z.S., M. Alfenito, P.A. Rea, V. Walbot and R.A. Dixon, 1997. Vacuolar uptake of the phytoalexin medicarpin by the glutathione conjugate pump. Phytochemistry, 45: 689-693.
Direct Link - Lucente, G., G. Luisi and F. Pinnen, 1998. Design and synthesis of glutathione analogues. Il Farmaco., 53: 721-735.
CrossRefDirect Link - Lyon, R.P. and W.M. Atkins, 2002. Kinetic characterization of native and cysteine 112-modified glutathione S-transferase A1-1: Reassessment of non substrate ligand binding. Biochemistry, 41: 10920-10927.
CrossRefDirect Link - Lyon, R.P., J.J. Hill and W.M. Atkins, 2003. Novel class bivalent glutathione S-transferase inhibitors. Biochemistry, 42: 10418-10428.
CrossRefDirect Link - Mannervik, B. and U.H. Danielson, 1988. Glutathione transferases-structure and catalytic activity. Crit. Rev. Biochem., 23: 283-337.
PubMedDirect Link - Marrs, K.A., 1996. The functions and regulation of glutathione S-transferases in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol., 47: 127-158.
CrossRefDirect Link - Marrs, K.A., M.R. Alfenito, A.M. Lloyd and V. Walbot, 1995. A glutathione S-transferase involved in vacuolar transfer encoded by the maize gene Bronze-2. Nature, 375: 397-400.
CrossRefDirect Link - McGonigle, B., S.J. Keeler, S.M.C. Lau, M.K. Koeppe and D.P. O'Keefe, 2000. A genomic approach to the comprehensive analysis of the glutathione S-transferase gene family in soybean and maize. Plant Physiol., 124: 1105-1120.
Direct Link - Mueller, L.A., C.D. Goodman, R.A. Silady and V. Walbot, 2000. AN9, a petunia glutathione S-transferase required for anthocyanin sequestration, is a flavonoid-binding protein. Plant Physiol., 123: 1561-1570.
Direct Link - Neuefeind, T., R. Huber, H. Dasenbrock, L. Prade and B. Bieseler, 1997. Crystal-structure of herbicide-detoxifying maize glutathione S-transferase-I in complex with lactoylglutathione: Evidence for an induced-fit mechanism. J. Mol. Biol., 274: 446-453.
PubMedDirect Link - Ortiz-Salmeron, E., Z. Yassin, M.J. Clemente-Jimenez, F.J.L. Heras-Vazquez, F. Rodriguez-Vico, C. Baron and L. Garcia-Fuentes, 2001. A calorimetric study of the binding of S-alkylglutathiones to glutathione S-transferase. Biochim. Biophy. Acta, 1548: 106-113.
PubMedDirect Link - Rea, P.A., 1999. MRP subfamily ABC transporters from plants and yeast. J. Exp. Bot., 50: 895-913.
Direct Link - Reinemer, P., H.W. Dirr, R. Ladenstein, J. Schaffer, O. Gallay and R. Huber, 1991. The three-dimensional structure of class pi glutathione S-transferase in complex with glutathione sulfonate at 2.3 A resolution. EMBO J., 10: 1997-2005.
PubMedDirect Link - Steyn, W.J., S.J.E. Wand D.M. Holcroft and G. Jacobs, 2002. Anthocyanins in vegetative tissues: A proposed unified function in photoprotection. New Phytol., 155: 349-361.
Direct Link - Walbot, V., L. Mueller, R.A. Silady and C.D. Goodman, 2000. Do Glutathione S-Transferases Act as Enzymes or as Carrier Proteins for Their Natural Substrates?. In: Sulfur Metabolism in Higher Plants-Molecular, Biochemical and Physiological Aspects, Brunold, C., H. Rennenberg, L.J. De Kok, I. Stulen and J.C. Davidian (Eds.). Paul Haupt Publisher, Berne, pp: 155-165.
- Watahiki, M.K., H. Mori and K.T. Yamamoto, 1995. Inhibitory effects of auxins and related substances on the activity of an Arabidopsis glutathione S-transferase isozyme expressed in Escherichia coli. Physiol. Plant., 94: 566-574.
Direct Link - Yu, S.J. and G.E. Abo-Elghar, 2000. Allelochemicals as inhibitors of glutathione S-transferases in the fall armyworm. Pesticide Biochem. Physiol., 68: 173-183.
CrossRefDirect Link - Zettl, R., J. Schell and K. Palme, 1994. Photoaffinity labeling of Arabidopsis thaliana plasma membrane vesicles by 5-azido-[7-3H]indole-3-acetic acid: Identification of a glutathione S-transferase. Proc. Natl. Acad. Sci. USA., 91: 689-693.
PubMedDirect Link








