ABSTRACT
A random sample of 563 individual plants of Urtica urens growing in polluted and unpolluted areas were collected to determine the effects of air pollutants on the growth parameters of this medicinally important plant using Von Bertalanfi, Gompertz and Richard`s growth models. The parameters of the three growth models were calculated for plants growing in the polluted and unpolluted areas using plant height, petiole length and leaf weight as the growth determining factors. There were differences in the growth of plants growing in the unpolluted area from those growing in the polluted area in terms of growth period, maximum plant height, leaf weight and petiole length. These differences indicated that air pollutants reduced the height of plants and thus increased the growth period and increased the length of petioles and the weight of leaves of plants growing in polluted area.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2006.335.340
URL: https://scialert.net/abstract/?doi=ajps.2006.335.340
INTRODUCTION
Alteration in the morphology and architecture of plants may result from changes in the pattern of allocation of meristems to growth (Bonser and Aarssen, 2001). These changes in meristems allocation within a species can be related to the emergence of strategies that may maximize fitness in a given environment. Moreover, plant parts, which have been subjected to diversifying forces, will often end up with weak or non-existing phenotypic correlations between organs (Herrera, 2005). In this study, the growth models of Urtica urens L. (small nettle), an important medicinal plants in Jordan and many other countries in the temperate zone of both hemispheres, growing at polluted and unpolluted areas are examined. The small nettle is an annual, monoecious plant with erect stem that is branching below and covered by stinging hairs. The leaves are broadly lanceolate or ovate, subcordate or tapering at base and are acutely dentate-serrate. Their petioles are short. The plant is stipulate with spike like inflorescence and achene fruits (Zohary, 1972). The growing regions of this plant in Jordan are subject to insult by a number of air pollutants originating mainly from thermal power plants and oil refineries. Few studies were carried out to evaluate the linkage between air pollutants and the growth patterns of plants. Most of these studies concentrated on modeling growth under different stress conditions. Latore et al. (1999) examined the effects of habitat heterogeneity and dispersal strategies on population persistence in annual plants. They calculated which habitat heterogeneity gives populations the best chance of persistence. Karlberg et al. (2005) used an integrated ecosystem model to simulate growth and transpiration over a range of salinities. They tested the model on two different seasons on saline water, drip-irrigated tomato. Thornley and France (2005) applied an open-ended logistic based growth model to determine the growth of organisms under limiting growth conditions such as temperature, nutrition or irradiance. A number of studies modeled either the influence of stress and stimulus factors from air and soil on plant growth (Chen et al., 1998), the interacting effects of browsing and shading on mountain forest tree regeneration (Weisberg et al., 2005) or the optimal root-shoot allocation and water transport in clonal plants (Stuefer et al., 1998). Moreover, few other studies examined the effects of pollutants on the growth and allometry of plants. Grantz and Yang (2000) studied the ozone impacts on allometry and root hydraulic conductance and reported that ozone induces an allometric shift in carbohydrate allocation. Kruse et al. (2003) examined the effects of elevated carbon dioxide partial pressure on growth, allometry and nitrogen metabolism of poplar plants. Their results indicated that elevated carbon dioxide partial pressure increased total biomass and the root: shoot ratio at deficient nitrogen-supply plants. Muzika et al. (2004) reported that ozone (O3), nitric dioxide (NO2) and sulfur dioxide (SO2) adversely influence the growth of Picea abies and Fugus sylvatica plants. Furthermore, researchers have reported that exposure to SO2 elicit multiple events linked to defense/stress responses in the leaves of rice seedlings (Rakwal et al., 2003), affects metabolism of Mangifera indica plants (Farooq and Hans, 1999) and alters yield of soybean grain (Xiong et al., 2003).
The purpose of this research is to determine the growth parameters of small nettle plant using Von Bertalanfi, Gompertz and Richard’s growth models, in order to determine the effects of air pollution on the growth of this plant.
MATERIALS AND METHODS
Study site and species: The first site was Zarqa, which is in the vicinity of pollution sources namely a petroleum refinery, a wastewater treatment plant and a thermal power plant. The collection site was about 100 meters from the thermal power plant. Al-Hassan (1995, 2000) reported that three types of air pollutants are found at this site, sulfur dioxide (SO2) ( 0.775 ppm hourly average and 0.352 ppm daily average), hydrogen sulfide (H2S) (0.840 ppm hourly average and 0.157 ppm daily average) carbon monoxide (CO) (6.24 ppm hourly average and 5.28 ppm daily average). All pollutant levels exceeded daily and monthly limits according to the Jordanian ambient air quality standard (JS1140/99). The second site was Sukhneh, which is located 20 kilometers away from the nearest pollution source. Based on rain distribution that is mainly in winter and spring Jordan has Mediterranean bioclimatic region. The two studied sites are of Arid Mediterranean bioclimatic region that is characterized by annual rainfall that ranges from 150-300 mm. The prevailing wind direction in both sites is westerly to North West in most times of the year (Al-Esawi, 1996).
There are four vegetative regions in the Middle East, the Mediterranean, the Irano-Turanian, the Sudanian and the Saharo-Arabian (Zohary, 1973). The two sites are located in the Irano-Turanian vegetative regions which is characterized by poor, eroded calcareous (loess) soil type. The vegetation is a timber less (no trees) with mostly shrubs and bushes. Urtica urens (small nettle) is found growing throughout the temperate zones of both hemispheres worldwide (Europe, Africa, Asia and both Americas). It is used locally and in other countries as antiasthmatic, depurative, diuretic, haemostatic, hypoglycaemic, adrenal tonic, astringent, cholagogue, circulatory stimulant, expectorant, kidney tonic, mucolytic, nutritive, parturient, styptic and thyroid tonic (Saad et al., 1988). It is worth mentioning that the above ground, roots and seeds are used for medicinal purposes.
Sampling and measurements: A random sample of 563 healthy small nettle plant individuals were collected from an area of 1 km2 during the growing season from January to May from both sites polluted (Zarqa) and unpolluted site (Sukhneh). Collected samples were placed in plastic bags, sprinkled with water and returned to laboratory within few hours where their roots were submerged in water for 16 to 20 h. From each of the chosen plants one leaf was harvested by cutting the petiole at a point where it is completely separated from the main axis. Petioles were excised from leaf blade and their lengths were measured. The blades and their petioles were dried at 70°C to constant weight then their dry weight was then determined. Plant height for each sample in addition to stem width was calculated Chang et al. (2004).
Statistical analysis: Measured plants were divided into size groups of plant height intervals calculated using histogram plots that show normal distribution of plant height. This calculation was carried out using computer based statistical software (STATISTICA software for windows. StatSoft, Inc., Tulsa, OK, USA) to compute the number of possible cohort sizes that best fits the normal distribution (Ismail and Elkarmi, 1999). Plant height was used to represent plant growth and was theoretically calculated using Gompertz (Aiba and Kohyama, 1996 and Elkarmi, 1998): Ht = ![]() [e (-A*e(-kt))], Richard’s: Ht =
[e (-A*e(-kt))], Richard’s: Ht = ![]() [1-A*e-kt] and Von Bertalanffy growth models:
[1-A*e-kt] and Von Bertalanffy growth models:
Ht = ![]() [1-e-k(t - t0)] (Ismail and Elkarmi, 2000) where Ht is the plant height at age t,
[1-e-k(t - t0)] (Ismail and Elkarmi, 2000) where Ht is the plant height at age t, ![]() is the plant height at age
is the plant height at age ![]() , A is a constant that is calculated as:
, A is a constant that is calculated as:
A= 1- (minimum height/ maximum height), k is the growth coefficient and t0 is the age at which the length is theoretically nil. The constants H∞, k and t0 were calculated using the quasi-Newton method (Ostle and Mensing, 1975). The same models were also applied to petiole length Leaf width and petiole weight.
RESULTS
The data show that on the bases of height the plants growing in the unpolluted area have a growth period of four months and those growing in the polluted area have a growth period of five months (Fig. 1). As discussed in the method section, this finding is based on the assumption that plant height is normally distributed (Ismail and Elkarmi, 1999).
The three models successfully calculated the growth parameters of Urtica urens growing in the unpolluted and polluted areas. Table 1-3 lists these parameters for plant height, petiole length and leaf weight.
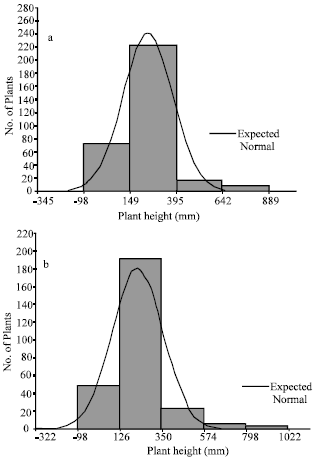 | |
| Fig. 1: | Frequency distribution according to plant height of estimated length groups showing expected normal distribution for plants growing in (a) unpolluted and (b) polluted areas |
| Table 1: | Results of the Richard’s growth model |
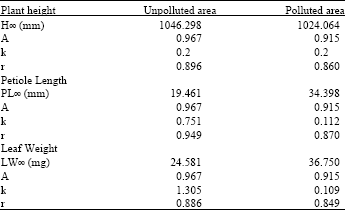 | |
For example Richards model predicted the growth of the plants in the unpolluted area calculated on the bases of plant height to be as:
| Table 2: | Results of the Von Bertalanfi’s growth model |
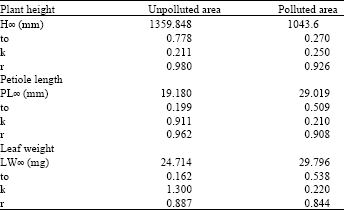 | |
| Table 3: | Results of the Gompertz growth model |
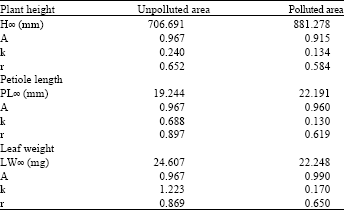 | |
PH = 1046.298 * (1- 0.967* e -0.2*t)
For the same parameter the Von Bertalanfi’s model is:
PH = 1359.848 * (1-e –0.211*(t-0.7782))
Moreover, the Gompertz model predict the growth of the plant to be as:
PH = 706.691 * [ e (-0.967* e (-0.24*t)) ]
The relationships between plant height, petiole length and leaf weight with the growing period for plants growing in polluted and unpolluted areas are shown in Fig. 2 and 3, respectively.
The three models used showed that the maximum height of the plants growing in the unpolluted area is more than the plant growing in the polluted area. These results are listed in Table 1-3. For example Richard’s growth model calculated the maximum height of Urtica urens growing in the unpolluted area to be 1046.298 mm and in the polluted area to be 1024.064 mm.
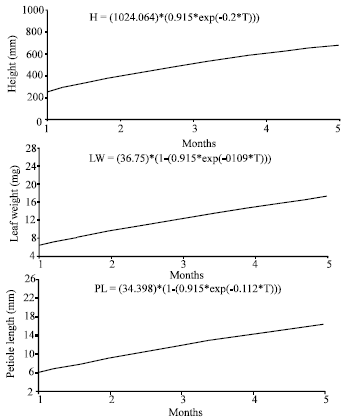 | |
| Fig. 2: | The relationships between plant height, leaf weight and petiole length for plants growing in the polluted area according to Richard’s growth model |
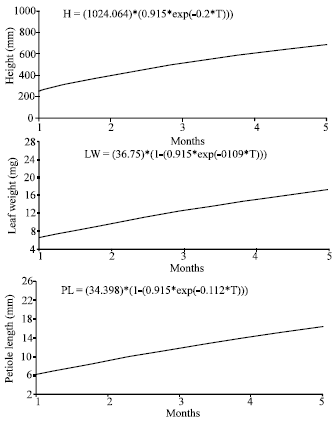 | |
| Fig. 3: | The relationships between plant height, leaf weight and petiole length for plants growing in the unpolluted area according to Richard’s growth model |
On the other hand, the three models showed that the maximum petiole length and leaf weight in the unpolluted areas are less than that in the polluted areas. For example, according to Richard’s model the maximum petiole length in the unpolluted area is 19.461 mm while in the polluted area is 34.398 mm.
DISCUSSION
The results show an apparent difference in the growth of plants growing in the unpolluted area from those growing in the polluted area. In addition to growth periods, maximum plant height, leaf weight and petiole length are different in the plants growing in the two areas. It is interesting to note that although maximum plant heights predicted by all three models in the unpolluted area are more than those in the polluted area, petiole length and leaf weight showed opposite behaviors. It seems that the effects of the pollutants resulted in reduced height and thus increased the growth period. On the other hand air pollutants stimulated leaf weight and petiole length increase possibly to counter the effects of pollution on leaves. A number of studies indicated that there are various effects of air pollutants on plant leaves. Vu (2005) reported that leaves of plants of A. hypogaea growing under elevated CO2 had higher photosynthetic CO2 exchange rate, lower transpiration and stomatal conductance and higher water-use efficiency compared to those grown at ambient CO2. Ribas et al. (2005) showed that ozone exposure induces the activation of leaf senescence-related processes and morphological and growth changes in seedlings of Mediterranean tree species. Other studies showed that growth of different plant parts can change as a result of environmental influences. Muzika et al. (2004) indicated that NO2 and SO2 had a negative effect on growth of plants. This was also reported by Deepak and Agrawal (2001) who reported that SO2 exposure resulted in reductions in plant growth, biomass and yield. Olszyk (1989) reported that SO2 reduced number and size of fruits. The results of the physiological and biochemical studies that were carried out (Perez et al., 2005; Hamerlynck et al., 2002; Rakwal et al. 2003) to examine the effects of pollutants on leaves of plants showed that there are effects of air pollutants on the physiology of leaves. Moreover, Collins and Cunningham (2005) after modeling the fate of sulfur in crops stated that sulfur as an air pollutant can be readily assimilated by vegetation. These reports indicate that plants when subjected to high concentration of atmospheric sulfur will tend to use it as a source of sulfur instead of soil sulfur. This uptake may be the physiological bases for the effects of air pollutants of the growth patterns of plants. Huber et al. (1999) indicated that phenotypic plasticity or the environmental effects on morphology and architecture can result in major changes. These include changes in size, structure and spatial positioning of plant organs including changes in internode and petiole length, in meristem utilization, timing of meristem outgrowth and the fate of the meristems. These reports are mainly in agreement with our results that air pollutants can influence the growth pattern of plants and affect the growth of different plant parts.
The results indicate that air pollutants reduced the height of plants and thus increased the growth period of plants growing in polluted areas while it increased the length of petioles and increased the weight of leaves possible to mitigate the effects of air pollution. Further studies are needed to examine the biochemical and physiological bases of the reported effects of pollutants on various plant parts.
ACKNOWLEDGMENTS
This research was funded by a grant from the Deanship of Scientific research and Graduate Studies at the Hashemite University, Jordan. The authors wish to thank Miss Maysoon Herzallah for her assistance in sample collection and measurements.
REFERENCES
- Aiba, S. and T. Kohyama, 1996. Tree stratification in relation to allometry and demography in warm-temperate rain forest. J. Ecol., 84: 207-218.
Direct Link - Chen, C.W., W.T. Tsai and A.A. Lucier, 1998. A model of air-tree-soil system for ozone impact analysis. Ecol. Model, 111: 207-222.
CrossRefDirect Link - Collins C. and N. Cunningham, 2005. Modelling the fate of sulphur-35 in crops. 1. Calibration data. Environ. Pollut., 133: 431-437.
CrossRefDirect Link - Deepak, S. and M. Agrawal, 2001. Infuence of elevated CO2 on the sensitivity of two soybean cultivars to sulphur dioxide. Environ. Exp. Bot., 46: 81-91.
CrossRefDirect Link - Farooq, M. and R.K. Hans, 1999. Metabolic effects of sulfur dioxide fumigation on Mangifera indica plants. Bull. Environ. Contam. Toxicol., 63: 774-781.
CrossRefDirect Link - Grantz, D. and S. Yang, 2000. Ozone impacts on allometry and root hydraulic conductance are not mediated by source limitation nor developmental age. J. Exp. Bot., 51: 919-927.
Direct Link - Huber, H., S. Lukacs and M.A. Watson, 1999. Spatial structure of stoloniferous herbs: An interplay between structural blue-print, ontogeny and phenotypic plasticity. Plant Ecol., 141: 107-115.
CrossRefDirect Link - Hamerlynck, E.P., T.E. Huxman, T.N. Charlet and S.D. Smith, 2002. Effects of elevated CO2 (FACE) on the functional ecology of the drought-deciduous Mojave Desert shrub, Lycium andersonii. Environ. Exp. Bot., 48: 93-106.
Direct Link - Herrera, J., 2005. Phenotypic correlations among plant parts in Iberian Papilionoideae (Fabaceae). Ann. Bot., 95: 345-350.
CrossRefDirect Link - Ismail, N.S. and A.Z. Elkarmi, 1999. Age, growth and shell morphometrics of limpet Cellana radiate (Born, 1778) from the Gulf of Aqaba, Red Sea. Venus Jap. J. Malacol., 58: 61-69.
Direct Link - Karlberg, L., A. Ben-Gal, P.E. Jansson and U. Shani, 2006. Modelling transpiration and growth in salinity-stressed tomato under different climatic conditions. Ecol. Modell. 190: 15-40.
Direct Link - Latore, J., P. Gould and A.M. Mortimer, 1999. Effects of habitat heterogeneity and dispersal strategies on population persistence in annual plants. Ecol. Modell., 123: 127-139.
CrossRefDirect Link - Muzika, R.M., R.P. Guyette, T. Zielonka and A.M. Liebhold, 2004. The influence of O3, NO2 and SO2 on growth of Picea abies and Fagus sylvatica in the Carpathian Mountains. Environ. Pollut., 130: 65-71.
Direct Link - Perez, P., R. Morcuende, I.M.D. Molino and R. Martinez-Carrasco, 2005. Diurnal changes of Rubisco in response to elevated CO2, temperature and nitrogen in wheat grown under temperature gradient tunnels. Environ. Exp. Bot., 53: 13-27.
Direct Link - Ribas, A., J. Penuelas, S. Elvira and B. Gimeno, 2005. Ozone exposure induces the activation of leaf senescence-related processes and morphological and growth changes in seedlings of mediterranian tree species. Environ. Pollut., 134: 291-300.
Direct Link - Stuefer, J.F., H.J. During and F. Schieving, 1998. A model on optimal root-shoot allocation and water transport in clonal plants. Ecol. Modell., 111: 171-186.
Direct Link - Thornley, J.H.M. and J. France, 2005. An open-ended logistic-based growth function. Ecol. Modell., 184: 257-261.
CrossRefDirect Link - Weisberg, P.J., F. Bonavia and H. Bugmann, 2005. Modeling the interacting effects of browsing and shading on mountain forest tree regeneration (Picea abies). Ecol. Modell., 185: 213-230.
Direct Link - Xiong, X., G. Allinson, F. Stagnitti, F. Murray, X. Wang, R. Liang and J. Peterson, 2003. Effects of simultaneous exposure to atmospheric sulfur dioxide and heavy metals on the yield and metal content of soybean grain (Glycine max [L.] Merr.). Bull. Environ. Contam. Toxicol., 71: 1005-1010.
CrossRefDirect Link - Vu, C.V., 2005. Acclimation of peanut (Arachis hypogaea L.) leaf photosynthesis to elevated growth CO2 and temperature. Environ. Exp. Bot., 53: 85-95.
CrossRefDirect Link - Rakwal, R., G.K. Agrawal, A. Kubo, M. Yonekura, S. Tamogami, H. Saji and H. Iwahashi, 2003. Defense/stress response elicited in rice seedlings exposed to the gaseous air pollutant sulphur dioxide. Environ. Exp. Bot., 49: 223-235.
CrossRef








