Research Article
Changes in Acid Invertase Activity and Sugar Contents in Lettuce During Storage at Ambient Temperature
Not Available
Toshiyuki Matsui
Not Available
Kazuhide Kawada
Not Available
Yusuke Kosugi
Not Available
Lettuce (Lactuca sativa L.) is a member of the family Compositae and is native to the Mediterranean region. It probably spreads into the rest of Europe. In the 15th century, lettuce became popular in the United States and is now the major salad crop. It is important in most European countries, Australia and Japan (Snowdon 1991). There are six edible forms of lettuce in the species L. sativa: crisphead, butterhead, cos or romaine, leaf, Latin and stem (Ryder, 2002).
Lettuce is a hardy annual popularly used as a vegetable salad. It is rich in minerals and vitamins (Janick, 1986). Lettuce head of high quality should have crisp, green outer leaves that are free from any blemishes. Yellowish outer leaves indicate that the head is on the onset of senescence. The midribs should neither be crushed nor split crosswise (Ryall and Lipton, 1979). During handling and storage, the compositional changes like sugar and organic acid contents occur that lead to senescence and quality deterioration. The high concentrations of specific carbohydrates, especially sucrose, will maintain quality of the product.
In most plants, the transported sugar is sucrose, a nonreducing disaccharide, in which glucose and fructose are linked (α1→β2). Sucrose is transported through the phloem from source organs to sink organs. It is subsequently hydrolyzed by invertase (β-D-fructofuranosidase, EC 3.2.1.26) (Richardson et al., 1990; Zrenner et al., 1996). Invertase is a hydrolase, cleaving sucrose into glucose and fructose. Invertase plays a key role in carbohydrate metabolism and in the regulation of sucrose transport in higher plants (Koch, 1996). Invertase exists in several isoforms with different biochemical properties and subcellular locations (Sturm, 1996). Most of plant species contain at least two forms of invertases. Vacuolar invertase (soluble acid invertases) is located in the vacuole lumen of this acidic compartment. Another, extracellular invertase (cell wall invertases) is ionically bound to the cell wall has also been detected. Soluble and cell wall invertases cleave sucrose most efficiently between pH 4.5 and 5.0. Thus, these are called “acid invertases” (Sturm, 1999).
Invertase appear to regulate the entry of sucrose into the different utilization pathways. Because sugars in plants are not only nutrients but also important regulators of gene expression (Koch, 1996), invertases may be indirectly involved in the control of cell differentiation and plant development.
There is little information available on the acid invertase activity and sugar contents in lettuce during storage period. Therefore, the objective of this study was to understand the changes in acid invertase activity and determine the changes in the respiration rate, weight loss, color and sugar contents in lettuce during storage at 20°C.
Plant materials: Two lettuce cultivars (‘Shizuka’ and ‘Shisuko’) were harvested from a commercially grown crop in December, 2004. Harvested lettuce heads were stored in perforated plastic bags at 20°C for 10 days. After 48 h intervals, the heads of each cultivar were taken out from storage. The leaves were separated from the midribs and stored at -30°C until analysis.
Weight loss: Weight loss was determined by weighing the heads at random after 48 h intervals until 10 days of storage at 20°C. Weight loss was revealed as percentage of the decrease in sample weights as determined every 48 h intervals.
Color assessment: Color change in lettuce heads were determined using a Chromometer (Minolta CR-200), equipped with an 8 mm measuring head. The meter was calibrated using the manufacturer’s standard white plate. Color changes were quantified in the L, a, b color spaces. L refers to the lightness of the head and ranges from black = 0 to white = 100. A negative value of a indicates green, while a positive number indicates red-purple color. A negative value of b indicates blue color, while a positive value of b indicates yellow (McGuire, 1992). Hue angle (h° = tan-1(b/a) when a>0 and b>0, or h° = 180o + tan-1(b/a) when a<0 and b>0) was calculated from the a and b values (Lancaster et al., 1997). On each head, three reading were taken from different portions.
Respiration rate measurement: Respiration rate was determined using a Gas Chromatograph; GC-8A. The lettuce head was weighed and carefully placed in a 10 L glass jar held at 20°C. Carbon dioxide production was measured at 48 h intervals from an intact head sealed for 1 h by taking 10 mL gas sample from the glass jar and injected to a TCD gas chromatograph equipped with a 1 m activated charcoal column at 70°C (GC-8 AIT, Shimadzu Co. Ltd.). The result was revealed as mL CO2 kg-1h-1.
Enzyme extraction for acid invertase: Extraction of enzyme was estimated using the methods similar to Islam et al. (1996). Approximately 5 g of fresh sample from each portion were added with 1% of polyvinylpolypyrrolidone (PVPP) and 1 g of sea sand. The mixture was homogenized using a cooled mortar and pestle with 5 mL of 0.2 M citrate-phosphate buffer (C-P buffer) at pH 5.0. The resulting homogenate was filtered through four layers of cotton cloth and the filtrate was centrifuged at 12,000 x g, at 2°C for 10 min. The total supernatant was dialyzed with 0.2 M C-P buffer (pH 5.0), diluted 40 times for 12 h with frequent stirring and the inner solution was designated as ‘soluble fraction’. The residual tissues were re-extracted with 5 mL of 0.2 M NaCl C-P buffer for about 24 h. The supernatant was dialyzed as described above. The dialyzed solution was designated as ‘cell wall-bound fraction’. All extraction procedures were carried out at 4°C followed immediately by the enzyme assays.
Enzyme assay for acid invertase: The standard assay medium for acid invertase consisted of 0.1 mL of water, 0.2 mL of 0.2 M C-P buffer (pH 5.0), 0.1 mL of 0.5 M sucrose and 0.1 mL of crude enzyme solution. The blank experiment contained distilled water instead of sucrose. The assay mixture was incubated at 45°C for 15 min. After that, the assay mixture was neutralized with 0.1 N NaOH or 0.1 N HCl. A coloring Somogyi’s copper reagent was added and the mixture was heated for 10 min in boiling water. After cooling, the mixture was added with 1 mL of Nelson’s reagent. The amount of reducing sugars was estimated by Somogyi (1952) method. Soluble protein content was determined by Lowry method (Lowry et al., 1951) using bovine serum albumin as the standard. The enzyme activity was revealed as the amount of glucose produced per minute per milligram of protein.
Determination of sucrose, glucose and fructose contents by high performance liquid chromatography (HPLC): The soluble sugar contents were determined by HPLC using the methods similar to the published previously (Islam et al., 1996). Approximately 2 g of fresh sample from each portion were added 1 g of sea sand and homogenized using a cooled mortar and pestle. Ten milliliter of distilled water was added to the homogenate and centrifuged at 12,000xg, at 2°C for 10 min. The mixture was filtered through a cellulose nitrate membrane filter (0.45 μm pore size). Soluble sugars were analyzed by HPLC using a stainless steel column (10.7 mm IDx30 cm) packed with silica gel (gel pack C610). The mobile phase (filtered water) was pumped through the column at a flow rate of 1.0 mL min-1. The pressure was adjusted to 28-29 kg cm-2 and the temperature to 60°C. A refractive index monitor (Hitachi L-3300) was used to record the peak heights. Sucrose, glucose and fructose were identified by their retention times and were quantified according to standards.
Statistics: A Randomized Complete Block Design (RCBD) was adopted with three replications. The level of significance was calculated from the F-value of ANOVA. The relationship between sugar contents and acid invertase activity were described with linear correlation analysis.
Weight loss: Figure 1 shows percent weight loss of two lettuce cultivars during storage at 20°C for 10 days. In both cultivars, weight loss was high after 2 days of storage. A further gradual decrease in weight was observed until the end of the storage period in both cultivars. Comparing the two cultivars, ‘Shisuko’ has higher weight loss than ‘Shizuka’.
Color assessment: Figure 2 shows hue angle value in both cultivars continuously declined throughout the 10 days storage period. The decline in hue angle value during storage was exhibited by a corresponding decrease in greenness. ‘Shizuka’ has higher hue angle value than ‘Shisuko’.
Respiration rate: In ‘Shizuka’ cultivar, respiration rate rapidly declined after 2 and 4 days of storage. After that, the rate increased until the end of storage. On the other hand, a continuous decrease in respiration rate was observed in ‘Shisuko’ until the end of the 10 days storage period. ‘Shisuko’ produced higher rate of CO2 than ‘Shizuka’ (Fig. 3).
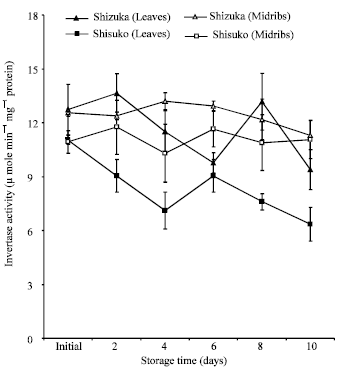
Acid invertase activity in soluble fraction: Figure 4 shows the acid invertase activity in the soluble fraction. In both cultivars and portions, the activity decreased throughout the storage period with few fluctuations except for a transient increase after 48 h of storage. The midribs portion of two cultivars showed higher activity than the leaves portion. The midribs of ‘Shizuka’ have higher acid invertase activity than ‘Shisuko’. However, there was no significant difference observed in the leaves portion between two cultivars.
Acid invertase activity in cell wall-bound fraction: In both cultivars, invertase activity decreased with some fluctuations in the leaves and midribs until the end of the storage period (Fig. 5).
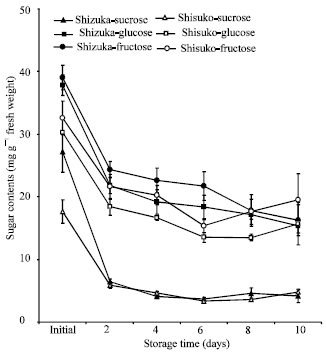
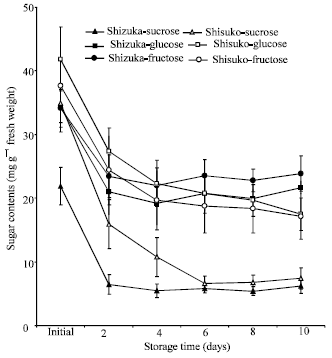
Soluble sugar contents: Sucrose content rapidly declined after 2 days of storage in the leaves and midribs of two cultivars and remained almost unchanged until the end of the 10 days storage period (Fig. 6 and 7).
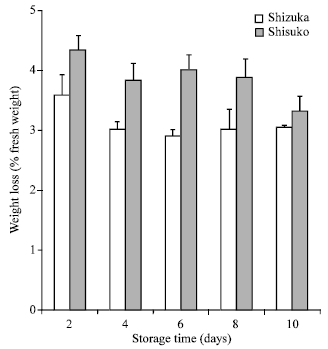 | |
| Fig. 1: | Weight loss of two lettuce cultivars (‘Shizuka’ and ‘Shisuko’) during storage at 20oC for 10 days. Each point represents the mean of 3 replications. Vertical bars indicate Standard Errors (SE) |
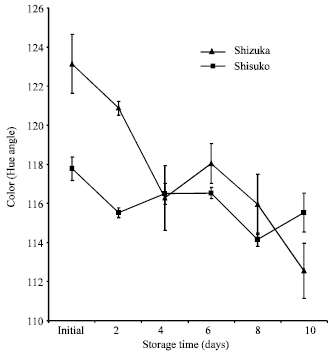 | |
| Fig. 2: | Color change of two lettuce cultivars (‘Shizuka’ and ‘Shisuko’) during storage at 20oC for 10 days. Each point represents the mean of 3 replications. Vertical bars indicate Standard Errors (SE) |
Fructose and glucose contents also declined with few fluctuations throughout the experimental period. A higher amount of three sugars (sucrose, glucose and fructose) were observed in the midribs than in leaves.
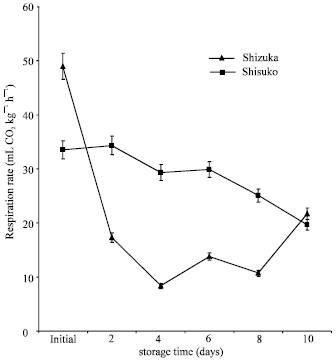 | |
| Fig. 3: | Respiration rate of two lettuce cultivars (‘Shizuka’ and ‘Shisuko’) during storage at 20oC for 10 days. Each point represents the mean of 3 replications. Vertical bars indicate standard errors (SE) |
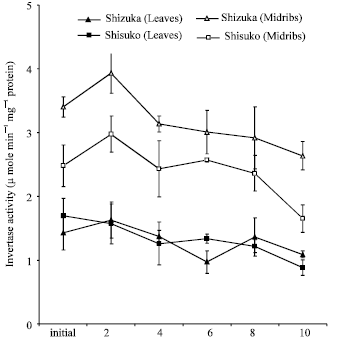 | |
| Fig. 4: | Change in acid invertase activity in the soluble fraction in the leaves and midribs of two lettuce cultivars (‘Shizuka’ and ‘Shisuko’) during storage at 20oC for 10 days. Each point represents the mean of 3 replications. Vertical bars indicate standard errors (SE) |
Among the three sugars, the level of fructose was higher than that of glucose and sucrose in the leaves of ‘Shizuka’ than ‘Shisuko’.
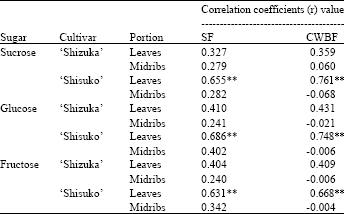
Correlation coefficients (r) between acid invertase activity and sugar contents: Table 1 shows the correlation coefficient (r) between the activities of acid invertase and sugar contents in the leaves and midribs of two cultivars. There was a highly significant positive correlation observed between the acid invertase activity in the soluble and cell wall-bound fractions and the sugar contents (sucrose, glucose and fructose) in the leaves of ‘Shisuko’ cultivar. No significant correlation was found between the invertase activity and sugar contents in both portions of ‘Shizuka’ cultivar.
Physiological and biochemical changes were observed in lettuce during storage at 20°C for 10 days. Weight loss was highest after 2 days of storage (Fig. 1) which could be attribute to loss in water and reducing substances. Water loss can be one of the main causes of deterioration during postharvest storage (Kader, 1986). It caused not only shrinkage, drying and softening of the commodity, but lead to accelerated senescence (Ben-Yehoshua, 1985). In addition, loss of green color is the most common postharvest defects (Cantwell et al., 1998). We observed that hue angle value continuously declined throughout the storage period (Fig. 2). Yellowing is the most important symptom of postharvest senescence in broccoli which is regulated by ethylene (King and Morris, 1994). A continuous decrease in respiration rate was observed in ‘Shisuko’ (Fig. 3). On the other hand, respiration rate of ‘Shizuka’ rapidly declined after 2 days but increased at the end of the storage period. This result is in agreement with the finding of Platenius (1942) that the respiration rate of lettuce during storage at 24°C declined after the first few days and a subsequent rise in the rate occurred toward the end of the storage period. This second rise in CO2 production might be attributable to the growth of microorganisms.
Interestingly, acid invertase activity in soluble and cell wall-bound fractions decreased (Fig. 4 and 5) while the soluble sugar contents (Fig. 6 and 7) declined throughout the storage period with few fluctuations in both cultivars and portions. It is likely that there are some factors, such as enzyme inhibitors, which bring about the low activity of acid invertase during storage in the lettuce head. Matsuura-Endo et al. (2004) reported that in potato tuber ‘White Fryer’ cultivar has very low invertase activity and the sucrose content decreased during storage.
 | |
| Fig. 5: | Change in acid invertase activity in the cell wall-bound fraction in the leaves and midribs of two lettuce cultivars (‘Shizuka’ and ‘Shisuko’) during storage at 20oC for 10 days. Each point represents the mean of 3 replications. Vertical bars indicate Standard Errors (SE) |
 | |
| Fig. 6: | Change in soluble sugar contents in the leaves of two lettuce cultivars (‘Shizuka’ and ‘Shisuko’) during storage at 20oC for 10 days. Each point represents the mean of 3 replications. Vertical bars indicate Standard Errors (SE) |
 | |
| Fig. 7: | Change in soluble sugar contents in the midribs of two lettuce cultivars (‘Shizuka’ and ‘Shisuko’) during storage at 20oC for 10 days. Each point represents the mean of 3 replications. Vertical bars indicate standard errors (SE) |
Activities of acid invertases are inhibited by their reaction products, with glucose acting as a non-competitive inhibitor and fructose as a competitive inhibitor (Sturm, 1999). It was further observed in the leaves of ryegrass that sucrose was broken down and the fructose was not used, which subsequently tend to inhibit hydrolysis (Foyer et al., 1997). In soluble fraction, the midribs of two cultivars showed higher invertase activity than leaves (Fig. 4). The difference in enzyme activities could be attributed to tissue types as observed in the branchlets and florets of broccoli (Baclayon et al., 2004; Pramanik et al., 2004). The activity was much higher in developing leaves than in mature leaves (Pavlinova et al., 2002). The acid invertase in the cell wall-bound fraction showed a higher activity than the soluble fraction. Likewise, this pattern was reported by Pramanik et al. (2004) in the broccoli stored at 20°C and Kassinee et al. (2004) in the vegetable soybean.
Sucrose content declined more rapidly than fructose and glucose (Fig. 6 and 7). Similar results were shown by Pogson and Morris (1997), who found that the sucrose content in broccoli florets declined more rapidly during storage than the content of glucose and fructose. In addition, Enninghorst and Lippert (2003) reported that during storage of Lamb’s lettuce at 20°C, the contents of glucose and fructose were almost constant while sucrose declined rapidly. The loss of sugar during storage is probably due to its transformation to cell wall material, lignin and other structural substances (Hansen and Weichmann, 1987).
| Table 1: | Correlation coefficients (r) between acid invertase activity and sugar concentrations in soluble and cell wall bound-fractions of lettuce leaves and midribs of two cultivars |
 | |
| SF = soluble fraction, CWBF = cell wall-bound fraction, | |
| *, **denote significant correlation at 0.01<p≤0.05 and p≤0.01, respectively, n = 18 | |
There was a highly significant positive correlation between acid invertase activity and sugar contents in the leaves of ‘Shisuko’ (Table 1), which suggest that the decrease of sugar contents was associated with the decrease in the activity of invertase. Eldan and Mayer (1974) reported that sucrose content immediately declined with the onset of germination in lettuce seeds, whereas acid invertase activity was found after 10-15 h of germination. Hence, acid invertase is apparently not the only enzyme which takes part in the hydrolysis of sucrose.
In the present study, it is concluded that there are physiological and biochemical changes occurred in lettuce during storage. These variabilities contributed to postharvest senescence of the commodity. ‘Shisuko’ showed higher percent of weight loss, degree of yellowing, respiratory metabolism and has low sugar contents than ‘Shizuka’. Further work is required to study the relationship between acid invertase activity and other sugar related enzymes in lettuce heads during storage.
The authors are grateful to the Government of Japan through its Ministry of Education, Culture, Sports, Science and Technology (Monbukagakusho: MEXT) for the financial support.