ABSTRACT
The effect of three plant growth regulators, N6-furfuryladenine (Kinetin-KIN), 6-γ-γ-dimethylaminopurine (2-iP) and 6-benzylaminopurine (BA) on the culture establishment and shoot proliferation of two grape genotypes, Vitis ficifolia var. ganebu and Kadainou R-1 (Vitis ficifolia var. ganebu x Vitis vinifera L.) and the effect of different concentrations of indole 3-butyric acid (IBA) and charcoal on in vitro rooting were investigated. Among the cytokinins tested for the culture establishment, KIN gave the highest culture establishment for Vitis ficifolia var. ganebu grape and Kadainou R-1 grape at 1.0 and 0.1 μM concentrations, respectively. For shoot proliferation, BA at 5.0 μM concentration resulted in the longest shoots in both genotypes. Shoot numbers were significantly higher at higher concentrations (5.0 and 10.0 μM) of BA in both genotypes. Rooting frequency did not differ significantly with different concentrations of IBA but root number increased significantly at higher concentrations (2.0 and 4.0 mg L-1) for both genotypes. Activated charcoal (AC) alone or with IBA did not improve rooting frequency but significantly increased root length for both genotypes.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2005.466.471
URL: https://scialert.net/abstract/?doi=ajps.2005.466.471
INTRODUCTION
Vitis ficifolia var. ganebu (hereafter referred to as Ryuukyuuganebu) is a wild grape native to Japan which is distributed in Amami, Ryuukyuu and Yaeyama Islands[1] and contains a high amount of anthocyanin[2]. Ryuukyuuganebu has no bud dormancy[3] and also has ever-bearing habit. Due to its novel characteristics, it has been utilized as a valuable resource for breeding programs. Kadainou R-1, derived from two Vitis genotypes i.e. Vitis ficifolia var. ganebu and Vitis vinifera L. (Muscat of Alexandria), is a new wine- type hybrid grape cultivar developed in Kagawa University, Japan. Kadainou R-1, which resembles Ryuukyuuganebu, also has no bud dormancy, with the possibility of year-round production.
Grape (Vitis sp.) categorized as a woody species, is mainly propagated through hardwood cuttings. Ryuukyuuganebu is difficult to root under ordinary conditions. Even use of indole 3-bytyric acid did not improve the rooting frequency for both hard-wood and soft-wood cuttings (data not published). However, this method requires large amount of materials for propagation and takes considerable time when a newly released hybrid, mutant or selection is to be multiplied in large numbers for commercial growing. Thus, micropropagation is considered to be a better alternative for both difficult to root species and new cultivars.
In vitro propagation of grape is affected by various factors like sucrose and NAA (α-naphthalene acetic acid) concentration[4], photoperiod[5], axillary bud position[6] and cytokinin and auxin concentration[7,8]. Isolated grapevine meristems show strong hormonal regulation in in vitro development and require cytokinins for their growth and development[9-11]. Different plant growth regulators at varying concentrations show considerable variability in in vitro regeneration of different species or cultivars. Plant growth regulators effective for in vitro regeneration of one species may not be equally effective for another cultivar or species[10-14]. Therefore, the selection of suitable plant growth regulators at their optimum concentrations will be dependent on the particular characteristics of the species, cultivar or selection to be multiplied in vitro. This is the first report that demonstrates that KIN is effective for culture establishment of grape genotypes. In addition, here, we report the effect of 3 plant growth regulators on in vitro propagation of Ryuukyuuganebu and Kadainou R-1 grapes and the effect of IBA and AC on in vitro rooting.
MATERIALS AND METHODS
Plant materials and explant preparation: Kadainou R-1 and Ryuukyuuganebu were used as sources of explants. Healthy, vigorous buds were collected from the University Farm vineyards of Kagawa University, located at Sanuki City, Kagawa, Japan from September 1 to 21, 2004. To reduce the heterogeneity of the explant materials, buds were removed from the middle portion of the branch, i.e. above the fifth node from the base and below the fourth node from the tip. Explants were collected after two sunny days, if following a rainy day, in order to avoid field contamination. The collected buds were rinsed with tap water for half an hour, rinsed for 30 sec with 70% ethanol and agitated for 15 min in 1% sodium hypochlorite and 2-3 drops of surfactant (Approach BI, Kao Co., Ltd., Japan) per 100 mL solution. Surface-sterilized buds were washed 5 times with double distilled water under a laminar airflow hood.
Meristems were excised from the buds and then left immersed for one month in 180x15 mm test tubes containing 10 mL liquid medium under sterilized condition.
Culture establishment and shoot proliferation: For culture initiation, half strength MS[15] medium was supplemented with 30 g L-1 sucrose, 0.01 mg L-1 NAA and three types of cytokinins at different concentrations. The cytokinins used were BA, 2-iP and KIN. BA and 2-iP were used at 0.1, 0.5, 1.0, 5.0 and 10.0 μM; and KIN at 0.01, 0.1, 0.3, 0.5 and 1.0 μM.
After one month, contamination percentage, survival percentage and response were recorded. The surviving meristems were further proliferated 2 times to get a mass of nodal shoots for further experiments using the best medium achieved in the previous experiment but added with 8.0 g L-1 agar. The proliferated shoots were used to study shoot proliferation potential.
Single node micro-cuttings (7 mm long) containing one leaf were used to study proliferation potential. Nodal micro-cuttings were excised from the mass of shoots and implanted vertically in a test tube (180x15 mm) containing 10 mL solid medium with the same concentrations of cytokinins as in the culture initiation media but without auxin. Shoot length, leaf and shoot numbers were recorded after one month.
Rooting: The rooting media consisted of half strength MS Salts, 30 g L-1 sucrose, 8 g L-1 agar, IBA at 0.2, 2.0 and 4.0 mg L-1 with or without 200 mg L-1 AC.
Fifteen mm long shoots excised from the mass of shoots prepared as described previously, were transplanted into a test tube containing 10 mL rooting medium. The number of primary and secondary roots and the longest root length were recorded after one month.
The pH of all the media used for culture establishment, shoot proliferation and rooting was adjusted to 5.7 prior to autoclaving. All the media were autoclaved for 20 min at 1.1 kg cm-2 and 121°C.
Hardening and acclimatization: For hardening, a glass flask (200 mL) filled with Kanumatsuchi soil (Kumeya Co. Japan) that was moistened with 1 ml L-1 Hyponex® (Hyponex Japan Co., Japan), capped with aluminum foil and autoclaved was employed. Well-rooted plantlets, obtained after one month of rooting, were washed thoroughly with distilled water to remove agar from the root surface and transferred to the hardening medium. After 3 weeks, the aluminum caps were loosened gradually day by day and removed completely at the end of a one-week period. Survival percentage was noted after 3 months.
Culture conditions: Cultures in the culture initiation, shoot proliferation, rooting and hardening experiments were kept in a growth room for a 16 h photoperiod at 25±1°C. The light source was provided by cool white florescent tubes at 30 μmol m-2 s-1.
Statistical analysis: The experimental design for all the experiments was completely randomized with 10 units per treatment, replicated three times. Data were subjected to analysis of variance (ANOVA) and the differences between means were tested at 5% level using Tukey test[16]. Percentage data were subjected to arc Sin square root transformation before analysis and later changed to original values for presentations.
RESULTS AND DISCUSSION
Effect of cytokinin on culture establishment: The three cytokinins tested in this study differed in their effect on culture initiation at different concentrations. KIN was found more effective than 2-iP and BA for culture establishment of Ryuukyuuganebu grape. Individually, the most responsive concentration of KIN was 1.0 μM followed by 2-iP at 0.5 μM i.e. 75 and 70%, respectively. The meristems of Ryuukyuuganebu grape did not survive BA concentrations above 0.1 μM. The survival rate of the meristems gradually decreased as the concentration of 2-ip increased above 0.5 μM (Table 1). For Ryuukyuuganebu, the phenylurea group of cytokinins resulted in better survival rate than the amino purine group of cytokinins.
| Table 1: | Effect of different cytokinins on culture establishment of two grape genotypes |
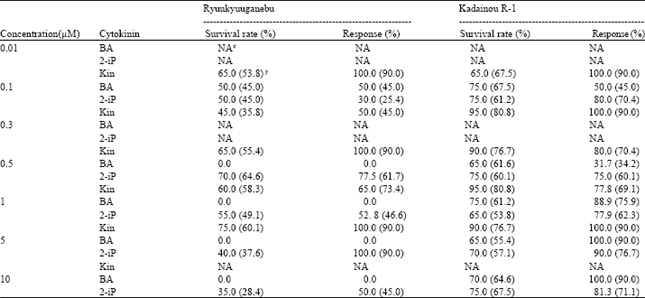 | |
| Data are means of 10 unit per treatment replicated three times, z: NA, indicates not applicable, y: Data on parenthesis indicate means of transformed values | |
| Table 2: | Effect of KIN on shoot proliferation of two grape genotypes |
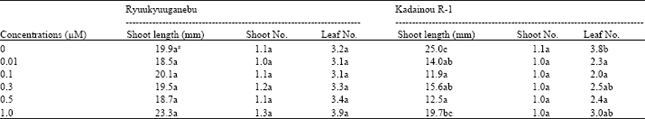 | |
| Data are means of 10 unit per treatment replicated three times, z: Different letter (s) within columns indicate significant difference at p<0.05 by the Tukey’s test | |
| Table 3: | Effect of 2-iP on shoot proliferation of two grape genotypes |
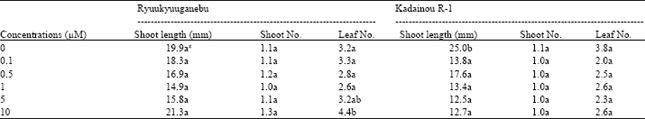 | |
| Data are means of 10 unit per treatment replicated three times z: Different letter (s) within columns indicate significant difference at p<0.05 by the Tukey’s test | |
| Table 4: | Effect of BA on shoot proliferation of two grape genotypes |
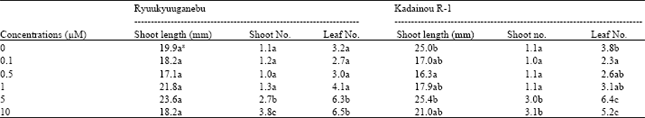 | |
| Data are means of 10 unit per treatment replicated three times z: Different letter(s) within columns indicate significant difference at p<0.05 by the Tukey’s test | |
| Table 5: | In vitro rooting of two grape genotypes |
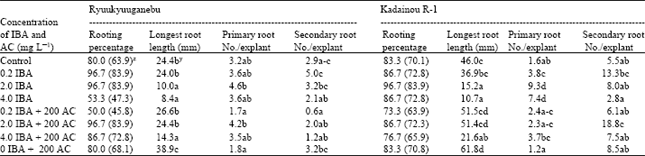 | |
| Data are means of 10 unit per treatment replicated three times, z: Data on parenthesis indicate mean of transformed values y: Different letter (s) within columns indicate significant difference at p<0.05 by the Tukey’s test | |
For Kadainou R-1 grape, KIN at 0.1 and 0.5 μM gave the highest survival rate (95%). Response was highest with lower concentrations (0.01 and 0.1 μM) of KIN and higher concentrations of BA (5 and 10 μM). Survival percentage was always poor when BA was used; however, quick and vigorous growth was observed in higher concentrations. No significant difference (p<0.05) was observed among the treatments in terms of survival percentage and response for both genotypes.
Low levels of cytokinin resulted in higher survival rates for both genotypes; however, the growth and response was delayed. Although, earlier studies had proved the superiority of BA on in vitro culture establishment of grapevines[9-11], the present study showed the effectiveness of KIN for the highest culture establishment. This may be attributed to the genotype or concentrations used. Goldy and Goldy[6] reported growth regulator and axillary bud position effects on in vitro establishment of Vitis rotundifolia. In their study, Thidiazuron (TDZ) alone or in combination with BA or KIN was found effective for establishing axillary buds. Furthermore, the study showed that explants originating from the 10 basal nodes of a shoot with at least 25 nodes gave better shoot proliferation than explants originating from the 10 distal nodes. In this experiments, KIN alone gave good results. So as to attain a more homogeneous explants sample, we obtained our explant materials as described earlier.
Effect of cytokinin on shoot proliferation: For shoot proliferation of Ryuukyuuganebu grape, BA at 5.0 μM gave the longest shoots, followed by KIN at 1.0 μM; however, there was no significant difference (p<0.05) between the two levels (Table 2 and 4). 2-iP did not significantly enhance the shoot length and number but significantly improved leaf number at 5.0 and 10.0 μM compared to lower concentrations (p<0.05) (Table 3). The shoot length increased as the BA concentration increased except at 10.0 μM (Table 4). In addition, shoot number increased as the BA concentration increased except at 0.5 μM concentration but no significant difference (p<0.05) was observed among the shoot numbers obtained in all treatments with the exception of that obtained in the treatment using BA at higher concentrations (5.0 and 10.0 μM).
Shoot length of Kadainou R-1 grape produced in KIN-supplemented media were comparable with the control (cytokinin-free media) (Table 2) but control shoots were significantly longer than those produced at 2-iP-supplemented media (p<0.05) (Table 3). Additionally, shoot length decreased as the 2-iP concentration increased above 0.5 μM (Table 3). The effect on shoot proliferation of BA on Kadainou R-1 was similar to that on Ryuukyuuganebu grape (Table 4). In the case of Kadainou R-1 grape, BA at 5.0 μM gave the longest shoot followed by cytokinin-free media; however, no significant difference was observed between the two treatments. Among the cytokinins tested, only BA above 5.0 μM concentration significantly affected the shoot and leaf numbers (p<0.05); mostly single shoots were observed with KIN and 2-iP for both genotypes even though proliferation period was prolonged to 6 weeks. As the proliferation period was prolonged, mostly vitrified shoots were observed at higher concentrations of BA.
The superiority of BA on in vitro shoot proliferation of grapevines was earlier reported by many researchers[8,10,11,17] although the concentrations used and the genotypes studied were different. The present study agrees with the results of previous studies on BA that resulted superior in terms of increasing shoot length, shoot number and leaf number. Kataeva et al.[18] also reported the vitrification effect when BA was used above 4.4 μM with Gerbera jamesonii and Malus domestica, similar to that observed in our experiment when proliferation period was prolonged to more than 6 weeks using the same medium (data not shown). The authors attributed vitrification to the excessive amount of cytokinins, which results in rapid division of cells in the meristems when subjected to an atmosphere of high humidity. Ibañez et al.[9] suggested to limit the multiplication cycles for Vitis vinifera L. cv. Napoleon to three since further transfers induced the vitrification phenomenon and degeneration. In these experiments, vitrification was not observed in short term culture, i.e. up to 4 weeks, which suggests that excess cytokinin along with prolonged proliferation period, might be associated with the vitrification process.
Shoot length did not differ significantly regardless of cytokinin type and concentrations, except for 2-iP in all concentrations for Kadainou R-1, as compared to the control in both genotypes (p<0.05). In the case of Kadainou R-1, the shoot length produced in cytokinin-free media was comparable to that produced in all other treatments but significantly longer (p<0.05) than that produced in 2-iP-supplemented media, which suggests that grape shoot length can be increased even in hormone-free media after several subculturings; however, cytokinin might be essential if shoot and leaf number are to be increased. Cytokinin was reported as the primary plant cell division hormone[19] . However, many tissues may not require cytokinin to proliferate in culture. Although some tissues may require cytokinin, after prolonged culture involving many subculturings, they lose their cytokinin requirement and will continue to grow without the addition of the hormone to the culture medium. It is likely that cytokinin synthesis is regulated in the plant and this regulatory mechanism can be bypassed in some cases, leading to unscheduled cytokinin synthesis, allowing the tissue to proliferate as long as its nutritional requirements are met.
Effect of IBA and AC on in vitro rooting: Low levels of IBA were effective in enhancing rooting percentage for Ryuukyuuganebu grape but drastically reduced rooting percentage of both genotypes at higher concentration (4.0 mg L-1). When IBA was used alone, the highest rooting percentage (96%) was achieved with 0.2 and 2.0 mg L-1 concentrations for Ryuukyuuganebu grape (Table 5); 2.0 mg L-1 concentration was required to get the best rooting percentage (96.7%) for Kadainou R-1 (Table 5). AC did not increase the rooting percentage but increased the root length of both genotypes. When AC alone or low concentration of IBA (0.2 mg L-1) with or without AC was used, mostly single roots were observed, which renders the explants not suitable for transfer to soil (Table 5). IBA not only enhanced the rooting percentage but also increased the primary root numbers (Table 5). IBA at ≥2.0 mg L-1 concentration also gave thicker roots compared to AC alone or a low level of IBA or without IBA and AC.
Plants produced in vitro were successfully acclimatized and hardened with Kanamatsuchi soil supplemented with 1 ml L-1 Hyponex. Survival rate did not differ significantly between plants produced using with or without charcoal for both genotypes. One hundred percent survival of plants was observed in both genotypes (data not presented).
Present result on the effect of IBA on in vitro rooting of grapevines were comparable to the result of Heloir et al.[20], which indicated that the intermediate concentration (2.0 mg L-1) of IBA gave the best rooting percentage for both genotypes. Singh et al.[11] on the other hand, reported that when AC was added along with IBA, it increased the rooting percentage. This result does not agree with the present study which showed that the rooting percentage decreased with the lower concentration of IBA and increased with the higher concentration of IBA in combination with AC. This may be attributed to the different genotype used. Furthermore the present study suggests that the AC absorbs the IBA from the media which eventually reduced the rooting percentage. Even if AC was used alone, rooting percentage did not increase as compared to IBA-free medium. In the present study, AC greatly reduced the primary root numbers. AC was responsible for increasing the root length and mostly showed an inhibitory or no effect on rooting frequency.
REFERENCES
- Jang, G., K. Kim and R. Park, 2003. Micropropagation of venus fly trap by shoot culture. Plant Cell Tissue Org. Cult., 72: 95-98.
CrossRefDirect Link - Murashige, T. and F. Skoog, 1962. A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol. Plant., 15: 473-497.
CrossRefDirect Link








