ABSTRACT
Experiments were conducted under greenhouse conditions to study I) the dynamics over time of mineral N, mineralizable N and humus N during decomposition in soil of leguminous and non-leguminous plant material and ii) the impact of these changes on growth and N nutrition of wheat fertilized with 15N-labelled ammonium sulphate. Finely ground straw of wheat, maize and sesbania was allowed to decompose in soil for 0, 2, 4 and 8 weeks. Sub-samples of soil were analyzed for the content of mineral N, mineralizable N and humus N. The bulk soil was sown to wheat and the plants harvested at maturity. Nitrogen as 15N-labelled ammonium sulphate was applied in two split doses of 40 and 60 mg kg-1 soil. Accumulation of mineral N in soil during 8 weeks of residue decomposition was found to depend on the chemistry of plant residues, more mineral N being released in soil amended with plant residues with narrow C/N ratio i.e., maize and sesbania. These residues also contributed more to humus N and maintained a higher content of potentially mineralizable N. Wheat straw not only caused a net immobilization of N during 8 weeks of aerobic incubation but a substantially higher loss of NO3¯+NO2¯ -N during subsequent incubation under submerged conditions. The loss of NO3¯+NO2¯ -N was more where residues were given less time for decomposition. Plant residues applied immediately before sowing wheat or those allowed shorter period of decomposition had a negative effect on plant growth. Of the three residue types, wheat straw caused a higher reduction in plant growth. This was attributed mainly to significantly reduced availability to plants of soil-N, while uptake of fertilizer N was affected almost similarly by the three types of residues. Residues of maize and sesbania had a positive effect on grain yield and total biomass of wheat. As a whole, ca 30% of the fertilizer N applied was taken up by the plants and >40% was unaccounted in the soil-plant system, fertilizer N remaining in soil being <30% of the applied. Relatively higher proportion of the applied N remained in amended than unamended soil after crop harvest.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2005.392.400
URL: https://scialert.net/abstract/?doi=ajps.2005.392.400
INTRODUCTION
Chemistry of plant residues is an important factor governing the release and availability to plants of nutrient elements from the native and applied sources. Easily oxidizable plant constituents like glucose lead to a rapid immobilization-remineralization turnover of nutrients e.g., N, while the process slows down with the complexity of the constituents[1,2]. The chemistry of plant residues affects nutrient dynamics mainly because of the differences in the build-up and degradation of microbial population or biomass; the process being rapid when easily oxidizable plant constituents are being utilized. Nevertheless, residues high in lignin content contribute directly and substantially to the stable humus fractions[1,3] that have proven beneficial effects on growth and nutrient acquisition of plants.
Freshly applied plant residues are reported to have a negative effect on plant growth mainly because of the release of phenolic compounds during the degradation process and the changes in microbial processes, especially the immobilization of essential nutrient elements. Being the nutrient element required more abundantly by the plants, nitrogen has been of common interest in most studies. Release and accumulation of N has been reported during decomposition of plant residues rich in total N[4,5]. Similarly, incorporation of non-leguminous plant residues obtained at green stage and having a C/N ratio of Ca 25 may also release substantial proportion of their N during decomposition[4,6]. However, the time period elapsed between incorporation of plant residues and introduction of plants may have significant influence on the availability of N originating from soil as well as from fertilizer and thus the growth performance of plants. The objectives of the experiment were to study I) the release of N (NH4+ and NO3¯ + NO2¯ -N) during the decomposition of some commonly available plant materials i.e., wheat straw (at maturity) and tops of sesbania (a leguminous green manuring crop) and maize (generally used as animal fodder but can be used as a soil amendment because of 2-3% N), ii) changes in mineralizable- and humus-N content of soil and iii) growth and N acquisition of wheat from 15N-labelled fertilizer and soil that contained plant residues decomposed for different length of time.
MATERIALS AND METHODS
Soil: The studies were conducted in 2003 on a sandy-loam soil collected from the surface 0-15 cm of the experimental fields of Nuclear Institute for Agriculture and Biology (NIAB), Faisalabad, Pakistan. Air-dried and sieved (<2 mm) soil had the following physico-chemical characteristics: organic C, 0.6%; total N, 0.09%; NH4+-N, 4.2 mg kg-1 soil; NO3¯+NO2¯ -N, 67.9 mg kg-1 soil; pH (1:2.5, soil:water suspension), 7.4; EC, 0.8 dS m-1 (1:2.5, soil:water suspension); water holding capacity, 25%; sand, 30%; silt, 36%; clay, 34%.
Plant residues: Finely powdered straw of wheat, maize and sesbania were used for incorporation in potted soil. Sub-samples of plant material were analyzed for total carbon (C), total nitrogen (N), 2 N H2SO4 hydrolysable C and hydrolysable N (Table 1). The three types of residues generally differed in most of the characteristics. However, wheat material was entirely different from the other two (especially in N concentration), which among themselves were similar in several respects including C and N content and hydrolysable N.
Pre-incubation of soil with different plant residues: Six-kilogram portions of air-dried and sieved soil were placed in 64 plastic pots. Soil in sets of 16 each were amended as follows: I) no amendment (control), ii) wheat straw at 0.5%, iii) maize straw at 0.5% and iv) sesbania straw at 0.5%. Moisture content of the soil was adjusted to 15% (w/w) with tap water and maintained at this level throughout the incubation by making up the weight loss. Incubation was carried out at 22-26°C for 0, 2, 4 or 8 weeks and managed in a way that all the samples were obtained on the same date. Portions of soil were analyzed for NH4+-N, NO3¯- + NO2¯ -N, humus N and total Kjeldahl N before sowing wheat.
Pot experiment using wheat as indicator crop: Replicate soil samples obtained after incubation were pooled and thoroughly mixed. Five and a half kg portions (on dry weight basis) of the soil were placed in plastic pots using three replicates for each treatment and sown to wheat (Triticum aestivum L., var. Inqilab). Five seeds were planted per pot and upon germination the stand was thinned to three seedlings. Moisture content of the soil was adjusted to 15% with a solution of 15N-labelled ammonium sulphate (2 atom % 15N excess) and potassium dihydrogen phosphate such that 220 mg N and 150 mg P were delivered per pot. A second dose of 330 mg ammonium sulphate N was given when maximum tillering was achieved. The plants were grown to maturity and received irrigation as required. Harvested plants were partitioned into root, straw and grain portions and data on dry matter content was recorded. Aliquots of finely ground plant components were analyzed for total N and 15N content as described later.
Analytical methods: For the determination of pH, ECe, texture and water holding capacity, methods described by Richards[7] were used. Total N and mineral N (NH4+-N and NO3¯+NO2¯-N) were determined by micro-Kjeldahl method[8,9]. Plant residues were also subjected to hydrolysis in order to determine the proportion of easily mineralizable C and N. For the determination of hydrolysable C and N, 1 g portions of the plant material were refluxed with 50 mL 2 N H2SO4 for 1 h; the contents of the flask filtered through a scintered funnel and the filtrate analyzed for total C and total N. For the determination of potentially mineralizable N, 20 g soil portions were weighed in capped plastic bottles and incubated under submerged conditions with 25 mL water for 2 weeks. The contents of the bottle were washed into distillation flasks with 2 N KCl. The amount of NH4-N released following distillation with Devarda’s alloy was taken as potentially mineralizable N, while changes in NO3¯ + NO2¯ - N during incubation were also followed. Total humic substances from KCl-extracted and freeze-dried soil samples were extracted with a mixed solution of 0.1 M NaOH and 0.1 M Na4P2O7. For this, 50 g portions of the soil samples were shaken with 150 mL of the extractant for 12 h followed by 1 h centrifugation of the suspension at 4000 rpm. Aliquots of the supernatant were acidified to pH 1.5 with concentrated H2SO4 and kept at 90°C for 30 min to precipitate Humic Acid (HA) fraction. The precipitate obtained through centrifugation was dissolved in 100 mL of 0.1 M NaOH solution. Aliquots of humic acid and fulvic acid solutions and root, straw and grain portions of wheat plants were analyzed for total N by steam distillation method[9]. The distillates were acidified with 1M H2SO4 (0.1 μL μg N-1), concentrated to 1 mL at 90°C on a hot plate and appropriate volumes subjected to N-isotope analyses on a VG Isolgas mass spectrometer using KOBr as alkalizer[10]. The mass spectrometer used was fitted with a double inlet system and had a sensitivity of 0.1%.
Microsoft Excel software was used to determine standard error and coefficient of correlations. Data on chemistry of plant residues was subjected to analysis of variance and determination of least significant difference using Duncan’s Multiple Range Test.
RESULTS
Plant residues used in this study differed significantly in their chemical characteristics (Table 1). Carbon content varied between 35 and 39%, the maximum being in wheat while maize and sesbania showed almost similar values. As expected, wheat straw had low N content of 0.5% resulting in a wider C/N ratio of 80. Maximum N concentration of 2.6% was determined in sesbania resulting in a narrow C/N ratio of 13.9. Maize also had characteristics mostly similar to that of sesbania because of being harvested at a relatively tender stage i.e., 6 weeks after seed germination. It had 2.1% N and a C/N ratio of 16.8, which can be considered optimum for net mineralization of N. In wheat, 30.2% of the total C was in acid hydrolysable forms, while in maize and sesbania this fraction was 42%. Compared to C, much lower amounts of residue N were hydrolysable being 3.0, 12.8 and 19.2 mg g-1 material of wheat, maize and sesbania, respectively. In terms of percentage, however, 62.1, 61.3 and 75.1% of the total N was hydrolysable in wheat, maize and sesbania, respectively.
Chemistry of the plant residues added to the soil had a significant bearing on the accumulation of mineral N in soil during 8 weeks of incubation.
| Table 1: | Chemical characteristics of plant residues |
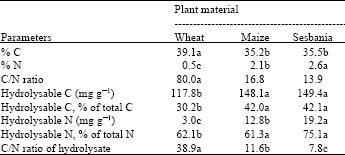 | |
| *, Figures in a row sharing a similar letter are not significantly different from each other at 5% probability according to Duncan’s Multiple Ranges Test | |
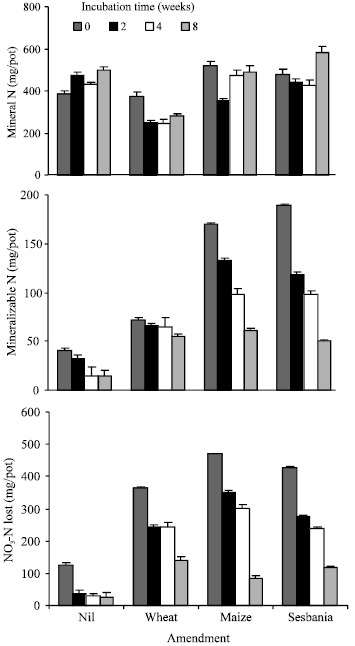 | |
| Fig. 1: | Changes in mineral N, mineralizable N and NO3¯ -N during incubation of soil samples under submerged conditions |
In general, the mineral N content of the soil samples increased with the time of incubation following an initial decrease after 2 weeks (Fig. 1). At zero incubation, where small amounts of NH4+-N were determined in soil amended with maize and sesbania, mineral N during incubation was almost entirely in the form of NO3¯+NO2¯ -N. Except for unamended soil where an increase in mineral N content was observed with time, in other treatments no consistent trends were observed. Net accumulation of mineral N was not observed in soil amended with wheat straw.
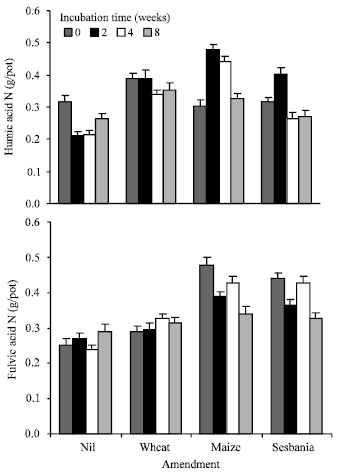 | |
| Fig. 2: | Nitrogen contained in Humic Acid (HA) and Fulvic Acid (FA) following incubation of unamended and amended soil samples |
Instead, mineral N content was lower as compared to that in unamended soil (average of all incubation intervals being 288 and 450 mg/pot, respectively).
Significant changes occurred in mineralizable N and NO3¯+NO2¯ -N content during incubation under submerged conditions of soil samples obtained after 0, 2, 4 and 8 weeks (Fig. 1). Mineralizable N (NH4+-N) in differently amended soils decreased with time of incubation and was almost similar in soil amended with maize and sesbania. In soil amended with wheat straw, mineralizable N decreased by 25% during incubation compared to about 60% in other two amendments. A decrease in mineralizable N suggested that a good part of the organic N was already mineralized during aerobic incubation for different time intervals. A significant positive correlation (r = 0.9, n = 16) was observed between mineralizable N and NO3-N unaccounted. The decrease in NO3-N +NO2¯ N was more from soil samples containing freshly added plant residues i.e., at 0-day of incubation. Higher losses of NO3+NO2¯ -N from soil samples containing less decomposed plant residues (i.e., incubated for a shorter time period) suggested a greater availability of easily decomposable plant residues.
Figure 2 presents the data on N contained in soluble humus fractions i.e., humic acid (HA) and fulvic acid (FA) or humates. Amended soils contained higher amounts of N in humates. Wheat and maize straw made a higher contribution to the humates, particularly to the humic acid fraction. However, average of different incubation intervals showed less N in fulvic acid fraction of wheat straw amended soil compared to the other two amendments. Incubation time had no consistent effect on the amount of N in the two humus fractions, although average values of different treatments showed a decrease in humates N (Fig. 3). Humic acid N decreased substantially during the first 2 weeks in unamended soil followed by a consistent increase, small changes with time were also noted in fulvic acid N. Humic acid N increased initially in amended soils followed by a decrease.
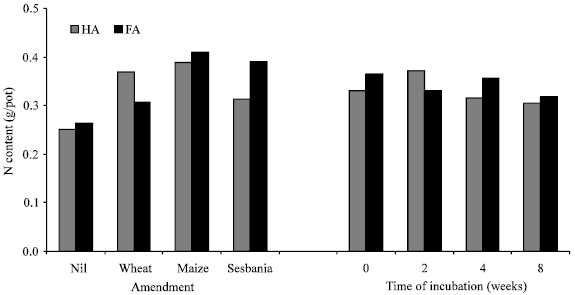 | |
| Fig. 3: | Effect of amendment (average of different incubation intervals) and incubation time (average of unamended and amended soil samples) on content of Humic Acid (HA) and Fulvic Acid (FA) following incubation of unamended and amended soil samples |
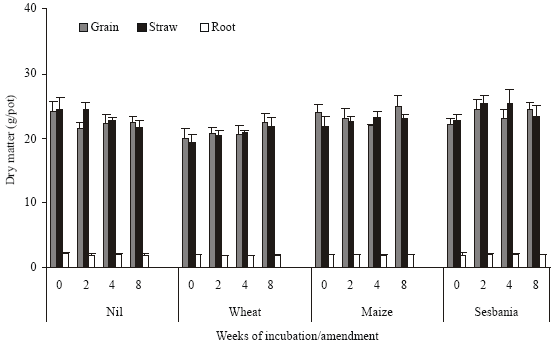 | |
| Fig. 4: | Dry matter distribution in root, straw and grain of wheat grown in amended and unamended soil incubated for different time periods |
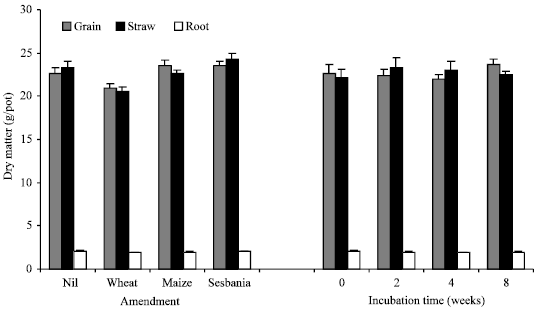 | |
| Fig. 5: | Effect of amendment (average of different incubation intervals) and incubation time (average of unamended and amended soil samples) on dry matter distribution in root, straw and grain of wheat |
Fulvic acid N increased with time in wheat straw amended soil, while it decreased in case of maize and sesbania amendments.
Incubation of unamended soil for extended period of time prior to wheat sowing had a negative but non-significant effect on dry matter yield (Fig. 4). Wheat straw amendment caused a significant reduction in dry matter yield of different plant components (15% in case of grain); the negative effect was relieved to some extent with extended time of decomposition. The two other amendments i.e., maize and sesbania, had a negligible effect on biomass yield as compared to unamended control. In case of sesbania, decomposition time had a positive effect. Average values for different amendments did not show a significant change due to the time elapsed prior to planting wheat (Fig. 5). Number of spikes increased due to maize and sesbania amendment but decreased in case of wheat straw amendment, period of decomposition had apparently no significant effect (data not shown).
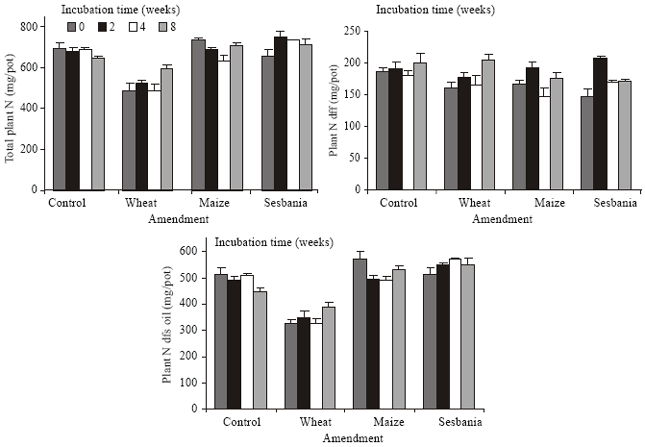 | |
| Fig. 6: | Total plant N and its origin as affected by soil treatments (incubation time and amendment); N dfs, N derived from soil; N dff, N derived from fertilizer |
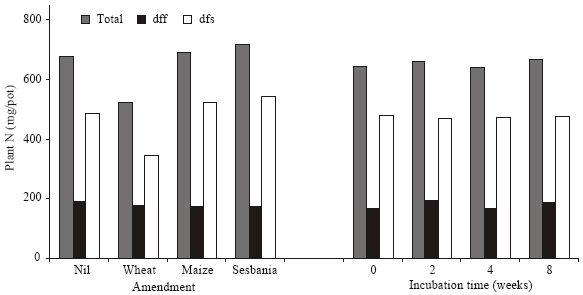 | |
| Fig. 7: | Effect of amendment (average of different incubation intervals) and incubation time (average of unamended and amended soil samples) on total plant N, N derived from soil (N dfs) and N derived from fertilizer (N dff) |
A close relationship was obtained between dry matter yield and N content of root, straw and grain portions; coefficient of correlation being 0.93, 0.92 and 0.94, respectively (n = 16). In unamended soil, length of incubation prior to sowing wheat had a negative effect on the uptake of total N and N derived from soil, while amended soils showed a positive effect of incubation time (Fig. 6).
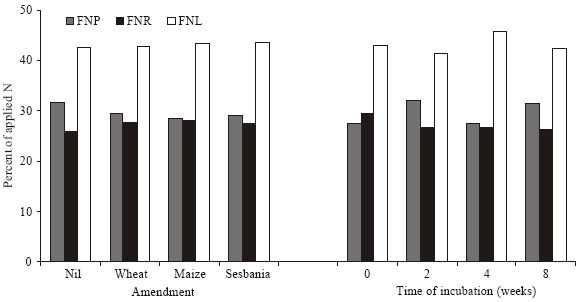 | |
| Fig. 8: | Percent of applied Fertilizer N in Plant (FNP), remaining in soil after harvesting plants (FNR) and lost from the soil-plant system (FNL) |
Wheat straw amendment had a negative effect on the N content of plants, while maize and sesbania had a negligible effect. The negative effect was relieved to some extent by prolonging the time of decomposition prior to sowing wheat. Uptake of fertilizer N was affected relatively positively by the length of incubation of both unamended and amended soil prior to wheat sowing. As a whole, incubation time and amendment had no significant bearing on the uptake of fertilizer N or soil N (Fig. 7). However, significant differences due to the type of amendment were observed in the uptake of soil N. There was a close correlation between total N in biomass and the N derived from soil suggesting greater dependence of plants on soil N.
A balance of fertilizer N after harvesting wheat (Fig. 8) showed a slight but non-significant decrease in the percentage of fertilizer N determined in wheat plants as a result of organic amendment; 28-32% of the fertilizer N being found in plants. The decrease was commensurate with a higher recovery of fertilizer N in soil (26-29%) and that unaccounted in the soil-plant system (41-46%). As a whole, the effect of organic amendment on the recovery of fertilizer N in plants was non-significant.
DISCUSSION
Plant residues decomposing in soil play a key role in determining the net release and availability to plants of N from native organic matter and/or applied sources i.e., organic matter and fertilizer. In general, immobilization-mineralization turnover of N and the further fate of mineralized N (e.g., losses through denitrification and NO3¯ leaching) are regulated by the chemistry of the organic inputs, the process being slow with the increased complexity of the latter[1]. In the present study, unamended soil showed a gradual increase in the mineral content of the soil with time, while a net immobilization prevailed in wheat straw amended soil (Fig. 1, top). Such results are frequently reported and have been attributed to net immobilization rather than net mineralization of N in soil amended with plant residues low in N content and having a wider C/N ratio[1,5]. In the other two amendments, net accumulation of mineral N was observed following an initial immobilization. However, the mineral N status of soil amended with maize and sesbania was roughly similar to that of unamended soil when average values for different incubation intervals were compared.
An accumulation of mineral N would be expected in relatively stabilized soil[11] with low level of easily oxidizable C. Soils rich in easily oxidizable C will not only show a net immobilization of N, but the mineralizable N will also be susceptible to losses through denitrification. Unamended soil in the present study will contain less amounts of easily oxidizable C and thus lower loss of N through denitrification and a higher accumulation of mineral N. Addition of organic matter not only results in increased microbial activity leading to higher mineralization of organic N, but it may also cause greater loss of mineralized N through denitrification at the expense of easily oxidizable C[12-14]. The results obtained in this study suggested that organic amendment led to the loss/immobilization of mineral N already present in the soil giving the impression of no net accumulation of mineral N. In general, higher mineralization of N is recorded in soils amended with N-rich plant residues[4,15]. Simultaneously, however, the mineralized N will be susceptible to losses in soil amended with plant residues like sesbania and maize containing significant amounts of easily oxidizable C and mineralizable N (acid hydrolysable C and N, respectively). Higher proportion of hydrolysable N in sesbania and other legumes may indeed be the reason for a rapid mineralization as well as loss of N. The C/N ratio of the hydrolysate was lowest (7.78) in case of sesbania and highest in case of wheat (38.85), with maize showing a value of 11.55 (Table 1). A narrow C/N ratio of the hydrolysate will help in a net mineralization of N, while wider C/N ratio could lead to a net immobilization of N and thus its restricted availability to plants as was the case in wheat straw. Immobilization and remineralization of N is reported as governed by the labile C component of the plant residues[1,4] which was roughly quantified in the present by acid hydrolysis.
Mineral N that accumulated (mainly as NO3¯) in soil was highly susceptible to losses following rise in moisture content. Soil samples incubated under anaerobic (submerged) conditions for the determination of potentially mineralizable N showed an almost complete loss of NO3¯+NO2¯ -N (Fig. 1, bottom). Higher losses of NO3¯+NO2¯ -N from soil samples containing less decomposed plant residues (i.e., incubated for a shorter time period) suggests the availability of easily oxidizable C for denitrifiers. The available C decreased with time and so was the loss of NO3¯+NO2¯ -N under anaerobic conditions. The three types of plant residues had almost a similar effect on the loss of NO3¯+NO2¯-N suggesting that C availability was not very different. Because of the substantial loss of NO3¯+NO2¯ -N, a net decrease in mineral N content of the soil samples was observed during anaerobic incubation. The loss of mineral N would also be expected upon irrigation of soil prior to planting wheat (a common practice) and hence any benefit in terms of available N will be lost.
A close relationship between easily oxidizable C and loss of NO3 through denitrification has frequently been observed[12,13]. Since under anaerobic conditions, organic matter is decomposed/fermented mainly by anaerobic microorganisms, more C is available for denitrifying population that is also encouraged by reduced oxygen supply. However, while determining potentially mineralizable N, generally no attention is given to the fate of mineral N (NO3¯+NO2¯ -N) already present in soil. In the present study, the three amendments differed considerably in affecting the decrease in mineral N, maximum loss being observed for wheat straw. The loss of mineralized N under high moisture conditions generally encountered following irrigation and land preparation for cropping may have a significant bearing on the N economy of agroecosystems. The difference in the nature of crop residues being incorporated will also affect the subsequent crop vis-à-vis the loss and availability of N from native and applied sources.
Potentially mineralizable N (NH4+-N) in differently amended soils decreased with the time of incubation and was almost similar to that in soil amended with maize and sesbania (Fig. 1, middle). In soil amended with wheat straw, mineralizable N decreased by 25% during incubation compared to about 60% in other two amendments. A decrease in mineralizable N also suggested that a good part of the organic N was already mineralized during aerobic incubation for different time intervals. A significant positive correlation (r = 0.9, n = 16) was observed between NH4+-N released and NO3¯+NO2¯ -N lost suggesting that the same C pool was driving the two processes.
The decrease in potentially mineralizable N was commensurate with an increase in N contained in humates i.e., humic acid + fulvic acid (Fig. 2 and 3) leading to a highly significant coefficient of correlations between the two (r = 0.89, n = 16). Soil amendments caused a significant increase in N contained in stable humus fractions that decreased with the time of incubation prior to sowing wheat. This decrease is possible as a significant part of the non-humified material also gets extracted and counted towards humates. This non-humified component is easily susceptible to further transformations and hence exhausted with the passage of time. Increased incorporation of N in relatively stable humus fractions and its non-availability to plants may indeed be one reason for no to negligible effect of organic amendments on plant N and dry matter yield at least on the short term.
In the present study, wheat straw had a negative effect on growth and N uptake of wheat (Fig. 4 and 5). This was expected in view of the potential of wheat residues to immobilize and thus decrease the plant availability of N[1]. The negative effect was, however, mitigated to some extent by allowing more time for the decomposition of wheat straw in soil. Incidentally, the soil used in the present study had high mineral N content of Ca 65 mg kg-1 (400 mg/pot) that was more susceptible to loss in amended than unamended soil following irrigation for sowing wheat. Hence, a good part of the available N of soil origin would have been lost from the soil-plant system thereby masking any differences in N supply expected from organic amendment. This contention was further supported by no to negligible effect of sesbania and maize straw on wheat yield and N uptake in spite of the positive effects reported for low C:N ratio plant residues[6,16,17]. A reduction in N supplying potential of amended soils was further augmented by higher incorporation of N in stable humus fractions (Fig. 2 and 3).
The plant residues, especially wheat straw, affected the plant growth more by reducing the availability of soil N (includes N derived from added plant residues) rather than that of 15N-labelled fertilizer N applied to the soil during the pot experiment. Indeed, availability of soil N was more closely linked to dry matter yield of wheat (r = 0.94, n = 16) than that observed for fertilizer N (r = 0.55, n = 16). This is justified as ca 70% of the plant N is derived from soil and any factor affecting the availability N from the majority source will have more effect on the yield as reported previously[18]. The residues affected the availability of soil N by affecting its losses as well as through immobilization. The impact of fertilizer N could therefore be enhanced by increasing the amounts applied. The observation that uptake, immobilization or loss of fertilizer N was not significantly affected by the form or decomposition status of the residues (Fig. 8) could well be the time of its application when the crop was at fairly advanced stage of growth and the soil relatively stabilized in terms of microbial activities.
In conclusion, the time period elapsed between incorporation of plant residues and introduction of crop plants will have a significant bearing on N nutrition and yield of the latter. Negative effects of plant residues (with a wider C/N ratio) on N nutrition of plant can be mitigated by giving more time for microbial action. However, a major effect of plant residues is on the availability of soil N rather than that of fertilizer N.
ACKNOWLEDGMENTS
The authors gratefully acknowledge the financial support of Pakistan Council for Science and Technology and Pakistan Ministry of Science and Technology, for partial financial support to carry out the reported work.
REFERENCES
- Azam, F., K. Haider and K.A. Malik, 1985. Transformation of 14C labelled plant components in soil in relation to immobilization-remineralization of N fertilizer. Plant Soil, 86: 15-25.
CrossRefPubMedDirect Link - Azam, F., F.W. Simmons and R.L. Mulvancy, 1993. Mineralization of N from plant residues and its interaction with native soil N. Soil Biol. Biochem., 25: 1787-1792.
Direct Link - Ibewiro, B., N. Sanginga, B. Vanlauwe and R. Merckx, 2000. Nitrogen contributions from decomposing cover crops residues to maize in a tropical derived savanna. Nutr. Cycl. Agroecosyst., 57: 131-140.
CrossRefDirect Link - Azam, F., 1990. Comparative effects of organic and inorganic nitrogen sources applied to a flooded soil on rice yield and availability of N. Plant Soil, 125: 255-262.
CrossRefDirect Link - Azam, F., A. Lodhi and M. Ashraf, 1991. Availability of soil and fertilizer nitrogen to wetland rice following wheat straw amendment. Biol. Fert. Soils, 11: 97-100.
CrossRefPubMedDirect Link








