ABSTRACT
The present study was designated to evaluate the antimicrobial and antifungal activities of essential oils of some medicinal plants against four plant pathogens at concentrations of 5, 10 and 15 μg and incubation times of 24, 48 and 72 h. A total of 7 medicinal plants traditionally used in medicines were subjected to preliminary screening against several pathogens. Aqueous, hexane extracts of each plant were tested for their antibacterial activity by using agar disk diffusion method. The results indicated that all plants exhibit antibacterial activity against Clavibacter michiganense subsp. michiganense, Pseduomonas syringae pv. tomato and Macrophomina phaseoli, except Xanthomonas campestris pv. malvacearum. In addition, coriander and hyssop essential oils were exhibit antibacterial activity against Xanthomonas campestris pv. malvacearum.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2005.225.228
URL: https://scialert.net/abstract/?doi=ajps.2005.225.228
INTRODUCTION
Essential oil of aromatic plants and spices are used in industries for production of soap, perfumes and toiletries. Many of them are also used in traditional medicine for various purposes[1]. Plants contain numerous biologically active compounds, many of which have been shown to have antimicrobial properties. Plant-derived medicines have been part of traditional healthcare in most parts of the world for thousands of years and there is increasing interest in plants as sources of agents to control microbial diseases.
The exploitation of natural substances such as the essential oils, safer to consumers and the environment, for the control of post harvest diseases and their effective usage against both wild type and strains resistant to pesticides, is urgently needed[2].
Işıkber and Karcı[3], pointed that essential oils were showed fumigant and repellent affects against Tribolium confusum. Some essential oil components such as carvone, anethole, carvacrol, menthol and thymol have antifeeding and oviposition deterent effect against carmine spider mite (Tetranychus cinnabarinus)[4]. Karaca et al.[5] have been reported that Thymus leucostomus, Mentha spicata and Origanum onites essential oils have antifungal activity against some Fusarium species.
Essential oils from various plants have shown also promise as potential sources for new nematicides. Most of these plants are aromatic and culinary herbs that contain the nematicidal compounds carvacrol and thymol.
Many pest-control agents are very dangerous for environment and soil, methyl bromide, a pest control agent that is commonly used on high value crops such as tomato. Methyl bromide is a soil fumigant when added to soil, produces a gas that permeates the soil and kills microorganisms and weeds. The essential oils as nematocidal, antimicrobial and antifungal agents present two main characters: the first is their natural origin which means more safety to the people and the environment and the second is that they have been considered at low risk for resistance development by pathogenic microorganisms.
The goal of this research was to search the effect of some medicinal plants essential oils against some pathogens (bacteria and fungi) that damaged importantly field crops such as tomato, cotton and sesame.
MATERIALS AND METHODS
Plant materials: Plant materials used in the study were cumin (Cuminum cyminum), dill (Anethum gravenoles), coriander (Coriandrum sativum), anise (Pimpinella anisum), mint (Mentha spicata), hyssop (Hyssopus officinalis) and fennel (Foneicum vulgare). These plants were cultivated at the Department of Field Crops, Faculty of Agriculture, Dicle University.
Mentha seedlings were collected from local producers. Hyssopus officinalis seeds were collected from the district of Dicle, Diyarbakır, Turkey in September 2001. Seeds of Mentha and Hyssopus were first sown in greenhouse; later when seedlings reached at 10-15 cm; they were transferred to the field in April 2002. Cumin, anise, fennel, dill and coriander seeds were harvested when the fruits were fully mature and mint and hyssop were harvested at fully flowering period. In the first year leaves were not harvested, but the second year the harvest was performed at the flowering period in June 2003. Fresh leaves were dried in a shadow and airy place for one week. Voucher specimens were stored in the warehouse until the analyses were performed. In the study, plant materials were ground in a spice mil before used.
Micro-organisms: Plant pathogens used in the study were obtained from the Department of Plant Production, Faculty of Agriculture, Adnan Menderes University, Isparta, Turkey. Clavibacter michiganense subsp. michiganense (G+), Pseduomonas syringae pv. tomato (G-), Xanthomonas campestris pv. malvacearum (G-) and Macrophomina phaseoli were used in the study.
Essential oils and antibiotic discs: The essential oils of plants were extracted by steam distillation. In the study, the disk diffusion method was used to determine the antimicrobial activity[6]. The essential oil solutions were prepared in hexane. By pipetting 5, 10 and 15 μL volumes of stock essential oils, 10 μg discs were prepared. The discs used as a control were absorbed with 10 μL hexane.
Antibacterial activity: Test organisms were inoculated onto the specific LB (Lauria Broth) media and incubated at 37°C for 2-6 h. The suspension was adjusted to a 0.5 McFalland turbidity standard (1x108 CFU mL-1) by using a spectrophotometer. Of bacteria suspension prepared from LB, 15 mL was suspended on agar (Mueller Hinton).
Paper discs with a diameter of 6 mm containing 5, 10 and 15 μg of essential oil samples to be assayed were put onto agar. Plates were incubated at 37°C for 24, 48 and 72 h and the inhibition zones of the microbial produced by different essential oils were measured as mm. All the tests used against pathogens were conducted three times and the results were expressed as average values.
RESULTS AND DISCUSSION
The results showed that essential oils from different plants possessed a wide antibacterial spectrum because they inhibited the growth a zone diameter of inhibition from 8 to 30 mm depending upon the susceptibility of the tested organism. In the study, among bacteria the most resistant is X. malvacearum. In addition, essential oils of cumin, dill, fennel, mint and anise were ineffective against X. malvacearum, on the other hand, coriander and hyssop essential oils were active against these bacteria. However, hyssop essential oil was not effected P. syringae pv. tomato (Table 1).
Except cumin oil, all essential oils at 5 μg were decrease inhibition zone against bacteria than 10 and 15 μg. Cumin oil was showed inactive effect at 10 and 15 μg against Clavibacter michiganense. Lower essential oil amount produced lower inhibition zone than high one.
At 10 μg, the highest inhibition zones are obtained from mint and hyssop oils against C. michiganense as 22 and 16 mm, respectively and at 15 μg, the highest inhibition zones obtained from mint essential oils against C. michiganense and M. phaseoli as 24 and 30 mm, respectively (Table 1). Inhibition zones were found gradually increased considerably when the concentration rate increased. Kızıl and Söğüt[7] have been reported that, the amount of inhibited bacteria is more at low concentrations than high concentrations for cumin and coriander essential oils. Therefore, it can be said that the quantity of oil was important for inhibition effects. Low concentration of essential oils were quite sufficient to prevent the growth of tested C. michiganense (Gram-positive), P. syringae pv. tomato (Gram-negative) and M. phaseoli.
The antibacterial and antifungal activity was observed after 24, 48 and 72 h of incubation. Inhibition zones of 24, 48 and 72 h incubation times were similar each other (Table 1). It can be said that 24 h incubation time is sufficient for microorganisms. Similar results were also reported by Cimanga et al.[1].
Some researchers reported that there is a relationship between the chemical structures of the most abundant compounds in the tested essential oils and the antimicrobial activity. Essential oils rich in phenolic compounds such as cumin aldehyde and linalool are widely reported to possess high levels of antimicrobial activity[8,9]. The major substances for cumin was cumin aldehyde (55.7%), anise oil was characterized by anethole, (80-90%) and fennel contain trans anethole (75.7-86.5%), The most abundant substances in mint is carvon, in coriander is linalool (60-80%), in dill is d-carvone (40-60%) and in hyssop is isopinocamphone (44.7%), respectively[9-11]. The variations of antibacterial activities of tested spice extracts may be due to their essential oil content, such as linalool, carvon, anethole etc. and the probably non-volatile compounds of plants.
It appears that there is a relationship between the chemical structures of the most abundant compounds in the tested essential oil and antibacterial and antifungal activity[1].
| Table 1: | Inhibition zones of essential oils at different concentrations (5, 10 and 15 μg) and incubation times (24, 48, 72 h) |
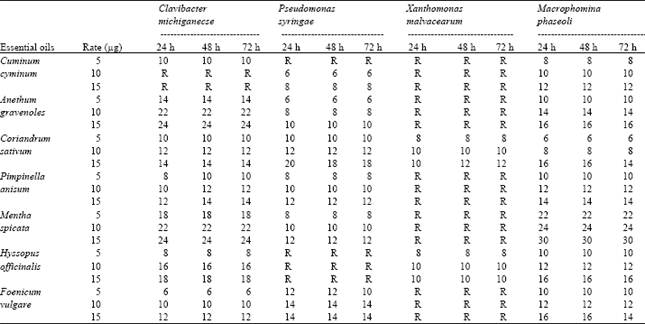 | |
| R.: Resistant | |
Further research is needed in order to obtain information regarding the practical effectiveness of essential oils to protect the plants, but present study gives support for the application of certain essential oils to control plant pathogens such as P. syringae pv. tomato, X. campestris pv. malvacearum and M. phaseoli or to eliminate the C. michiganense subsp. michiganense under specific application conditions.
In order to control the contamination of seeds with C. michiganense and other seed borne diseases and the growth of survival during storage, it is necessary to have an understanding of the controlling factors for this organism in seed.
In conclusion, present results suggest that the extracts of all tested essential oils might be a source of antibacterial activity against plant pathogens. In the study, the data show that Gram-negative bacteria were more resistant to various essential oils than Gram-positive. Present results suggest that the use of some spice and aromatic plants essential oils as antimicrobial agents may be exploitable to prevent the deterioration of seeds by bacteria and fungi.
ACKNOWLEDGEMENT
We would like to thank Prof. Dr. Kemal Benlıoğlu Department of Plant Protection, Faculty of Agriculture, Adnan Menderes University, Isparta-Turkey for providing plant pathogens.
REFERENCES
- Cimanga, K., K. Kambu, L. Tona, S. Apers and T. De-Bruyne et al., 2002. Correlation between chemical composition and antibacterial activity of essential oils of some aromatic medicinal plants growing in the Democratic Republic of Congo. J. Ethnopharmacol., 79: 213-220.
PubMedDirect Link - Farag, R.S., Z.Y. Daw, F.M. Hewedi and G.S. El-Baroty, 1989. Antimicrobial activity of some Egyptian spice essential oils. J. Food Prot., 52: 665-667.
Direct Link








