Research Article
The Lectin Content of Cotyledonary Callus from Canavalia brasiliensis (Mart. ex. Benth)
Not Available
Renato A. Moreira
Not Available
Ana C.G. Horta
Not Available
Andre Luis C. Silva
Not Available
INTRODUCTION
The plants belonging to the genus Canavalia are widely distributed in the tropical regions and are important for animal nutrition due to their high protein content. Canavalia brasiliensis seeds contain a lectin which shares many properties with the other lectins already isolated from members of the tribe Dioclineae, especially those belonging to the genus Canavalia[1]. Studies have revealed that all Canavalia lectins belong to the Glc/Man-type lectins, with a high degree of structural homology as well as very similar physicochemical properties[2,3].
The techniques of plant tissue culture possess an enormous potential for the plant improvement and can also be used as a tool for study of protein synthesis[4]. Tissue culture, inclusive of in vitro cells, tissues and organs, is a convenient and effective means in breeding genetics, physiology and biochemistry of legumes and it can also contribute to production of biomass and metabolites[5].
Studies investigating lectin biosynthesis have been performed using callus and suspension cultures derived from embryos[6], cotyledons[7], stem and leaf[8] and shoot[9]. Although the in vitro tissue culture has been reports for different dicotyledonous and monocotyledonous species, the number of reports approaching protein synthesis in callus culture is still low[10,11]. Callus and cell culture could not only be an alternate continuous source of proteins, but could also be a useful and important model system to study their regulation and biosynthesis[12,13] and in biochemistry studies[10,14,15].
The aim of this study was to find differences in protein contents in callus cotyledonary from C. brasiliensis calli and to follow the lectin and other protein synthesis during its growth.
Plant material: Canavalia brasiliensis plants were grown in the campus of the Federal University of Ceará (Brazil) and the mature seeds were collected from heterogenous plant groups.
Preparation of explants: The mature seeds were submitted to the treatment of chemical scarification for 50 min with sulfuric acid (95-97%) to break the latency. After that, they were washed exhaustingly with sterile distilled water and left to germinate in petri plates, under dark conditions, at 28°C in the germination chamber. After 5 days, the seedlings were moved and sterilized with NaClO 2.5% for 7 min and then washed with sterile distilled water. Cotyledon segments were use as explants for callus induction.
Medium for callus induction and maintenance: The medium containing MS salts[16] (mg L-1) and 1.0 thiamine-HCl, 1.0 pyridoxine-HCl, 1.0 nicotinic acid, 2.0 glycine and 100.0 myo-inositol was used as the basal medium. The medium also was supplemented with different concentrations of the growth regulators (2,4-D and kinetin) and of sucrose according to the treatments: 1) medium with 0.46 μM kinetin and 2% sucrose; 2) medium with 0.46 μM kinetin, 0.45 μM 2, 4-D and 2% sucrose; 3) medium with 0.46 μM kinetin, 2.26 μM 2,4-D and 2% sucrose; 4) medium with 0.46 μM kinetin, 4.52 μM 2, 4-D and 2% sucrose; 5) medium with 0.46 μM kinetin, 4.52 μM 2,4-D and 3% sucrose. The culture media were solidified with 0.8 g L-1 Agar (Merk), the final pH being adjusted to 5.8. The sterilization was performed in autoclave, at 121°C for 15 min.
Callus induction and maintenance: Segments (16±2 mm) were taken aseptically from cotyledons of the mature seeds and placed on different treatments tested. The cultures were incubated at 28±2°C in the dark. After 28 days, the calli obtained from mature seeds were separated from explants and transferred to fresh MS medium of the same composition used for induction. The calli were subcultured every 4th week.
Growth determination of callus cultures: The calli stocks established previously were used to follow the growth pattern. The growth and development of the callus were followed through growth curve in interval of 4 days (0, 4, 8, 12, 16, 20 and 24 days after) for the coming of mature seeds. The cultures were incubated at 28±2°C in the dark. The growth curves were determined based on fresh and dry weight of the calli.
Callus crude extract preparation: Cotyledon calli (obtained from subculture of treatment 1) were macerated and submitted to extraction with 0.15 M NaCl, 0.1 M pH 2.6 glycine-HCl, 0.1 M pH 4.0 Na-acetate, 0.1 M pH 6.0 Na-phosphate and 0.1 M pH 8.0 Tris-HCl buffers, all containing 0.15 M NaCl. The mixtures were sonicated for 3 h and clarified by centrifugation at 12.000 g for 20 min at 4°C. The clear supernatants were used for determination of soluble protein content. The measurement of protein concentration in the different fractions was performed according to Bradford[17], using Bovine Serum Albumin (BSA) as standard.
Callus protein synthesis: The protein synthesis in cotyledonary calli during to growth curve was followed by sodium dodecyl sulphate polyacrylamide gel electrophoresis (PAGE-SDS) as described by Laemmli[18]. This was carried out in a 2 mm thick vertical slab gel consisting of stacking gel, 3.95% total acrylamide and 12.5% (m/v) in gel separation. Samples (10 mg of callus powder) were dissolved in 1 mL of 0.0625 M Tris-HCl pH 6.8, 2% SDS buffer with 1.0% β-mercaptoetanol and incubated at 100°C for 10 min. A few crystals of sucrose were dissolved in the samples, which were then applied to the gel. The electrophoresis was conducted at a constant current of 25 mA. Silver staining was performed as described by Blum et al.[19].
Western blot analysis: The calli proteins were ellectroblotted onto nitrocellulose membranes (Protan, Keene, N.H.), using a Bio-Rad electrotransfer cell, for 1 h at 150 V (constant voltage). Membranes were developed with a secondary antibody-alkaline phosphatase detection system (Promega, Wis.), using rabbit polyclonal antibodies produced against crude extract and lectin from C. brasiliensis seeds. The antiserum was obtained using a moodification of a procedure described by Tijsen[20]. The antiserum anti-ConBr titer of 1:2000 (v/v) was used for all experiments.
Amino acid composition: Amino acid analysis was performed after hydrolysis at 110°C of callus samples (in sealed glass tubes under N2) for 20 h in 6 M HCl. After hydrolysis, HCl was removed by evaporation and the residue was analyzed, in a BIOCHROM 20 (Pharmacia) amino acid analyzer. Tryptophan was determined spectrophotometrically[21].
RESULTS AND DISCUSSION
The callus induction of C. brasiliensis was found in all media supplemented with 2,4-D, kinetin and sucrose (Table 1). The medium containing only kinetin and sucrose was unable of induce calli. Among the regulators added the to medium, the 2,4-D is known as the main synthetic auxin used to induce the callogenesis, because one of its main characteristics is the capacity to efficiently stimulate the cell division in tissues of several plants[14]. After the first subculture, the treatment 4 (with 0.46 μM kinetin, 4.52 μM 2,4-D and 2% sucrose) showed calli more developed that the other media tested (Fig. 1A). It was also observed that, in this medium, the calli presented whitish coloration and friable consistency, compared with the calli obtained from the medium 2, 3 and 5, which presented dark brown coloration and hardened consistency. Ozaki[22] reported that the majority of callus from Canavalia gladiata leafs were soft and yellowish-brown, but in a part of the callus, powdery white tissues were formed on the soft yellowish-brown callus.
| Table 1: | Percent of calli obtained in the treatments used for induction of callogenesis in cotyledons of C. brasiliensis in relation to different combinations of growth regulators and sucrose |
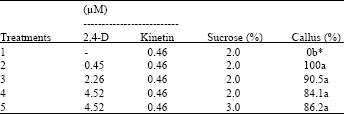 | |
| *Means of four repeat experiments with about 20 explants used in each treatment. Mean values followed by the same letter do not differ significantly by the Tukey’s test (p<0.05). | |
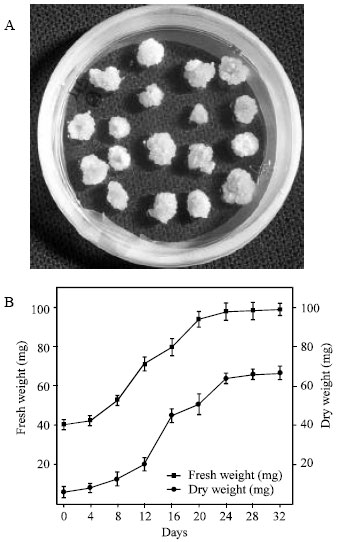 | |
| Fig. 1: | (A) Callus culture of C. brasiliensis cotyledon induced and subcultured in the treatment 4 (containing 0.46 μM kinetin, 4.52 μM 2,4-D and 2% sucrose). (B) Growth curves of the calli based on fresh and dry weight |
Growth curves of cotyledon calli were plot based on fresh and dry weights (Fig. 1B). Cotyledon calli (obtained of the medium 4) gave rise the typical growth curves with lag, exponential, linear and stationary phases. The adaptive period (lag phase), in which the fresh and dry weight of calli increases slowly, occurred up to 8th day. According to Scragg and Allan[23], the lag phase is considered an energy producer period. The fast growth phases (exponential and linear phases) occurred between 8th and 24th day. The exponential or biosynthetic phase is the period of maximum cellular division and greatest growth rate of the calli, while the linear phase is the period in which the cells grow but the cellular division decreases[23]. The stationary phase of cotyledon calli occurred between the 24th and 32nd days. In this phase, the growth rate decreases and then remain essentially constant. According to Smith[24], in the stationary phase occurs depravement of nutrients in culture medium and a reduction of the O2 amounts inside the cells.
The calli obtained from medium 4 were submitted to extraction with different pH values (Fig. 2). It was found maximum soluble protein content at pH 8.0 (11.6 mg g-1 calli flour) and a minimum content at pH 2.6 (3.7 mg g-1 calli flour). The result obtained with the C. brasiliensis seeds flour showed that the pH 8 also was the most efficient in the soluble proteins extraction[2]. Gupta and Srivastava[25] also observed a high protein content (28.7 mg g-1 calli flour) in the cotyledonary calli from Zizyphus mauritiana.
The SDS-PAGE assay of cotyledon calli obtained from treatment 4 (first, second and third subculture) revealed that there was one protein band in the calli samples similar to protein band from C. brasiliensis seeds lectin (ConBr). The 30 kDa lectin subunit of native ConBr was always included in these experiments as an control (Fig. 3A). Using antibodies produced against native ConBr, we detected a strong reaction of the antibody with proteins obtained from callus.
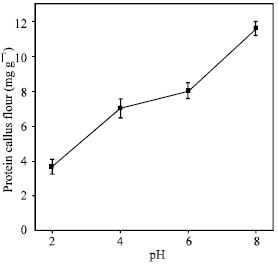 | |
| Fig. 2: | Influence of pH on the solubility of C. brasiliensis cotyledon callus proteins obtained from treatment 4 (containing 0.46 μM kinetin, 4.52 μM 2,4-D and 2% sucrose) |
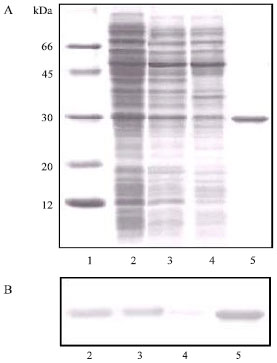 | |
| Fig. 3: | (A) SDS-PAGE of the calli proteins (obtained from treatment 4). Lanes: 1) Molecular weight standard; 2) Crude extract of the calli from first subculture; 3) Crude extract of the calli from second subculture; 4) Crude extract of the calli from third subculture and 5) C. brasiliensis seeds lectin (ConBr). (B) Western blot using antibody produced against ConBr. Lanes: 2) Crude extract of the calli from first subculture; 3) Crude extract of the calli from second subculture; 4) Crude extract of the calli from third subculture and 5) ConBr |
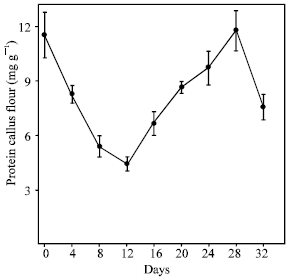 | |
| Fig. 4: | Soluble proteins content of C. brasiliensis cotyledon calli obtained during the growth curve |
It is evident from the Western blot data that a protein band with an apparent mass around 30 kDa that cross-react with anti-ConBr (Fig. 3B) antibodies are present in calli samples.
 | |
| Fig. 5: | (A) SDS-PAGE patterns of C. brasiliensis cotyledon calli proteins during the growth curve. Lanes: 1) Molecular weight standard; 2) 4 day; 3) 8 day; 4) 12 day; 5) 16 day; 6) 20 day; 7) 24 day; 8) 28 day; 9) 32 day and 10) Seeds lectin (ConBr). (B) Western blot using antibody anti-ConBr. Lanes: 2) 4 day; 3) 8 day; 4) 12 day; 5) 16 day; 6) 20 day; 7) 24 day; 8) 28 day; 9) 32 day and 10) Seeds lectin (ConBr) |
This result indicated to presence of the lectin in the callus culture. During subsequent subcultures, this protein did not appear or was not clearly defined. Sircar et al.[26] reported that callus obtained from hypocotyl of Canavalia gladiata, after several passages of subculturing showed the absence of lectin (ConA). From this initial finding, it showed that callus had no totipotency for the production of mRNA ConA.
Figure 4 show soluble protein content found in callus during the growth curve. The results showed that between the 0 and 12th day of both culture there was a decrease in the protein content. This reduction possibly occurred due to an adaptation period of calli in the culture medium. In addition, the calli also showed an increase in the protein levels at 28th day and a decreasing at 32nd day.
Electrophoretic patterns of proteins were also analyzed during the growth curve and revealed that there was a small reduction in the number of protein bands at 12th day (Fig. 5A). Between the 12th and 28th day, it was observed that some protein bands (between 30 and 66 kDa) reappeared, suggesting a process of de novo synthesis.
| Table 2: | Amino acid composition of the calli and seeds flour from C. brasiliensis |
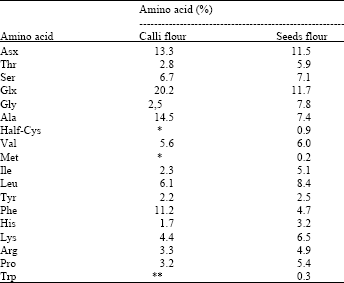 | |
| *Not detected, **Not determined. | |
In addition, the Western blot showed that the ConBr band (30 kDa) was preserved during all period of callus development (Fig. 5B).
Proteins are valuable indicators of differentiation and have been used in taxonomy, as genetic markers and could be practical to identity specific stages of development of somatic embryos[27]. A biochemical marker might be useful for early detection of embryogenic cultures before any morphogenic changes. Its use would help to optimize culture conditions and discriminate cultures to follow the multiplication process. Among these are proteins[28].
The data on amino acid content of the cotyledon calli flour obtained from treatment 4 (callus with 28 days old) revealed that the Asx and Glx to be the two most abundant hydrophilic amino acids, in agreement with the results obtained with seeds flour (Table 2). Together they constituted 33.5% of the total amino acid residues. The calli flour also contain large amounts of hydrophobic amino acids, such as Ala and Phe. Met and Cys residues were not detected in calli flour. The low or null concentrations of half-Cys, Met and His are very common in vegetable proteins[29]. Others amino acids showed similar values to the seeds flour.
Present results suggest that callus culture provides an interesting model system to investigate the lectin and others proteins biosynthesis, thus providing a clue to their physiological functions.
ACKNOWLEDGMENTS
This study was supported by grants from the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq).